General Chemistry 4th Edition University Science Books presented by Macmillan Learning Two 20.0 g ice cubes at -12.0 °C are placed into 295 g of water at 25.0 C. Assuning no energy is transferred to or from McQuarrie - Roc the surroundings, calculate the final temperature, Tf, of the water after all the ice melts. heat capacity of H,O(s) 37.7 J/(mol-K) °C Tf = heat capacity of H,O(1) 75.3 J/(mol-K) 6.01 kJ/mol enthalpy of fusion of H,0
General Chemistry 4th Edition University Science Books presented by Macmillan Learning Two 20.0 g ice cubes at -12.0 °C are placed into 295 g of water at 25.0 C. Assuning no energy is transferred to or from McQuarrie - Roc the surroundings, calculate the final temperature, Tf, of the water after all the ice melts. heat capacity of H,O(s) 37.7 J/(mol-K) °C Tf = heat capacity of H,O(1) 75.3 J/(mol-K) 6.01 kJ/mol enthalpy of fusion of H,0
Chapter10: Liquids And Solids
Section: Chapter Questions
Problem 102E: A 0.250-g chunk of sodium metal is cautiously dropped into a mixture of 50.0 g water and 50.0 g ice,...
Related questions
Question
I need full and accurate answers.
Transcribed Image Text:Iswer
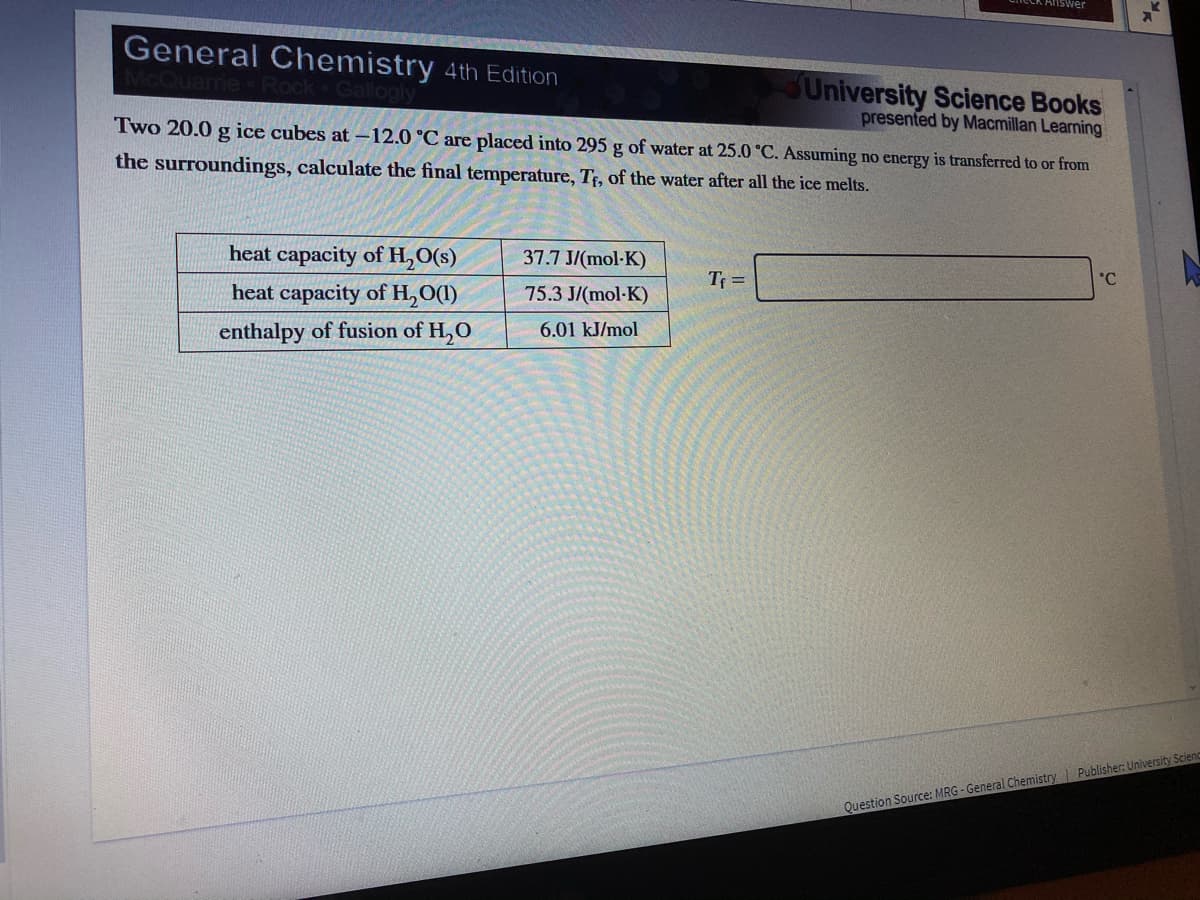
General Chemistry 4th Edition
University Science Books
presented by Macmillan Learning
Two 20.0 g ice cubes at -12.0 °C are placed into 295 g of water at 25.0 °C. Assuming no energy is transferred to or from
CQuarrie Rock
Sallogly
the surroundings, calculate the final temperature, Tf, of the water after all the ice melts.
heat capacity of H,O(s)
37.7 J/(mol-K)
°C
Tf =
heat capacity of H,O(1)
75.3 J/(mol-K)
6.01 kJ/mol
enthalpy of fusion of H,O
Question Source: MRG - General Chemistry Publisher: University Sciend
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Physical Chemistry
Chemistry
ISBN:
9781133958437
Author:
Ball, David W. (david Warren), BAER, Tomas
Publisher:
Wadsworth Cengage Learning,

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning