Q: For µ=0.1, the activity coefficient (gamma) of M2+ is 0.41 while gamma of X- is 0.75. Calculate how…
A: At ionic strength of 0.1, the activity coefficient values are: γM2+ = 0.41γX− = 0.75 Ksp = 3.8 x…
Q: 1) PhN=C=S; 2) HF CF3CO2H HF 1) DCC; 2) HF NaOH, H2O 1) DCC; 2) CF3CO2H 1) DCC; 2) NaOH PhN=C=S DCC
A:
Q: The graph below shows the distribution of molecular speeds in a sample of fluorine gas at…
A: The distribution of the speed of gas molecules at various temperatures can be understood by the…
Q: In what volume ratio should you mix 1.0M solutions of NH4CI and NH3 to produce a buffer solution…
A:
Q: Draw the major product(s) of the following reaction. O, initiator • You do not have to consider…
A: Note : Alkene reaction with oxygen gives carbonyl compounds.
Q: For which of these pairs of species is the first species an acid AND the second species is its…
A:
Q: Substance Xis known to exist at 1 atm in the solid, liquid, or vapor phase, depending on the…
A: The temperature of substance X is needed to be determined if it was initially present at 0°C and a…
Q: Solution: Kso=1.41-10-11 The resistance of a saturated solution of PbSO4 (very insoluble salt) at…
A:
Q: Enter your answer in scientific notation. Be sure to answer all parts. Calculate the nuclear binding…
A: Proton is a positively charged particle and neutrons are neutral.Protons and neutrons are present in…
Q: 1) 03, -78 °C H. ? 2) CH3SCH3
A: ->Ozonolysis of alkene produces carbony compound. In cinnamaldehyde there is present alkene which…
Q: QUESTION 2 Match the compound names with their proper formulas: v ammonium nitride a. Au2S v iron…
A: Name and given its chemical formula of the compound ---
Q: Calculate the heat capacity of your calorimeter, using Eq. 9d. (Enter your answer to three…
A: The heat capacity of calorimeter is needed to be calculated using the following equation. ∆H1 +…
Q: According to the following reaction, how many moles of lithium sulfide should be needed to produce…
A: Given :- 3Li2S + Mg3P2 → 2Li3P + 3MgS No.of moles of Li3P produced = 9.206 mol To calculate :-…
Q: The usual concentration of the agarose gel used is 1% (w/v) agarose. If you were to make 50 ml of…
A: Given ,%w/v=1 We have to calculate mass of agarose.
Q: Suggest a synthesis for the molecule shown below. Only starting materials containing six (6) carbons…
A:
Q: 7)) Sucrose does not undergo mutarotation because A) it contains no hemiacetal linkages. B) it is a…
A:
Q: What is the pH of a solution made by mixing 100.mL of 0.0500 M HCIO4lan) and 50.0mL 0.0400 M KOH(an?…
A:
Q: Chemistry Name the compound using mass spec, NMR, and IR.
A: From the mass spectra, it is given that the mass of the unknown compound is 245.32 According to the…
Q: Please give the the VB partial :C : CI Te CI CI: orbital diaaram for the following:
A:
Q: For u=0.1, the activity coefficient (gamma) of M2+ is 0.41 while gamma of X- is 0.75. Calculate how…
A: At ionic strength of 0.1, the activity coefficient values are: Ksp = 1.2 x 10-9
Q: One of the buffers that can be used for running an agarose gel is 1x TBE (tris borate EDTA) buffer.…
A: On dilution concentration changes but no of moles remain same. So, M1V1 = M2V2 M1: Concentration of…
Q: Draw and label a structure of: A diglyceride of oleic acid
A: Diglyceride of oleic acid have the structural formula
Q: Calculate the ΔG°' for the following reaction: NAD:H + FAD → NAD+ + FAD:H2
A: Given Reaction: NAD:H + FAD → NAD+ +…
Q: Calculate Q for the following: 125.0 mL of 0.0500 M Pb(NO3)2 is mixed with 75.0 mL of 0.0200 M NaCl…
A: Answer: After the mixing of given two solutions, precipitate will only form if ionic product of lead…
Q: What is the molar solubility of calcium hydroxide Ksp (Ca(OH)2) = 5.02 × 10-6J, when it is added to…
A: Given : Ksp of Ca(OH)2 = 5.02 x 10-6
Q: QUESTION 10 Which compound is best represented by the following 13C NMR data? 21 6 (ppm) O enthanoic…
A:
Q: How to prepare 0.10M of potassium iodate
A: Let us discuss the molarity and then solve the problem.
Q: Balance the equation and calculate the Eocell of the following reaction: Zn(s) + Ag+(ag) --------->…
A: We have to calculate E0cell for reaction Zn(s) + Ag+(ag) ---------> Ag(s) + Zn2+(aq)
Q: Choose the correct order based on the given criterion per line for comparative 1.5 M aqueous…
A: ->Acids has pH less than 7 , stronger acid has less pH. -> Base has pH greater than 7 ,…
Q: 33-35. Predict the product of the reaction sequence shown below. MgBr H,SO, (conc.) Bra -COM light…
A:
Q: ctron reorganization.
A:
Q: Sometimes in lab we collect the gas formed by a chemical reaction over water (see sketch at right).…
A:
Q: A sample of an unknown gas is placed in a vessel with a volume of 1,638 mL at a temperature of 48.7…
A: Given: The volume of the gas = 1638 mL The temperature of the gas = 48.7 ∘C The pressure of the gas…
Q: 19. What set of ketone (1) and aldehyde (2) will provide the same alcohol product (3) when submitted…
A: Lithium aluminum hydride reduces the carbonyl compounds into alcohols. In this reagent, a hydrogen…
Q: Please complete the reaction Br LIAIH, Br2 Br FeBra excess Mg
A: Detail solution is given below
Q: in mind that HCN is a weak acid. acids: O bases: O ? other: O acids: O bases: O O other: O
A: Acid base reaction takes +place between KOH and HCN. HCN + KOH --> KCN + H2O As both have same…
Q: Concept Check No. 5. (Problem 7.30) Chemical degradation of chlorophyll gives a number of substances…
A:
Q: One of the molecular orbitals of carbon disulfide, CS, (S=C=S) is shown below. Identify the type of…
A: CS2 is a linear molecule having double bond between C and S i.e.there are two single bonds and two…
Q: Consider these two entries from a fictional table of standard reduction potentials.…
A: We have to calculate the standard potential of cell.
Q: Trial 3: Final buret reading (mL) Buret reading 36 37 38 39
A: what is the reading of this buret?
Q: QUESTION 4 According to the following reaction, how many moles of water will be produced from 1.84…
A:
Q: 24-26. Predict the product of the reaction sequence shown below. 1.KMNO,KOH; 2. H H,O(excess) H,SO,…
A: Gabriel phthalimide synthesis: In the Gabriel synthesis an alkyl halide is converted into a primary…
Q: Oxygen is transported around the body by the globular protein hemoglobin which contains four Fe2*…
A: 1. The equilibrium constant, K, is much stronger in haemoglobin-carbon monoxide reaction compared to…
Q: Assign oxidation numbers to each element and balance the equation in an acidic solution. HSO3- +…
A:
Q: Predict the major product of the following reaction. HBr peroxides • You do not have to consider…
A: Alkene gives an electrophilic addition reaction with hydrogen halide to form an alkyl halide.
Q: A reaction vessel is charged with 0.50 atm of A and 0.190 atm of B. Once the reaction reaches…
A: Given, A (g) ⇌ 2 B (g) Equilibrium constant (Kp) = 67.2 Initial partial pressure of the species:…
Q: What is the solubility (in g/L) of lead(II) chromate, PbCrO4 in (2.600x10^-1)M potassium chromate?…
A: Given: The Ksp for lead(II) chromate = 1.8x10-14\ We have to calculate the solubility (in g/L) of…
Q: 1. Show how would you convert aniline to the following compounds: A. Fluorobenzene B. Chlorobenzene…
A: Since you have asked multiple questions with multiple sub-parts, we will solve first question for…
Q: he nuclear binding energy
A:
Q: You obtain a cell count of 4 x 105 cells/mL. Before counting, your cell suspension was diluted 1:5…
A: Given that, the cell count is 4×105 cells/mL. Also, given that, before counting the cell suspension…
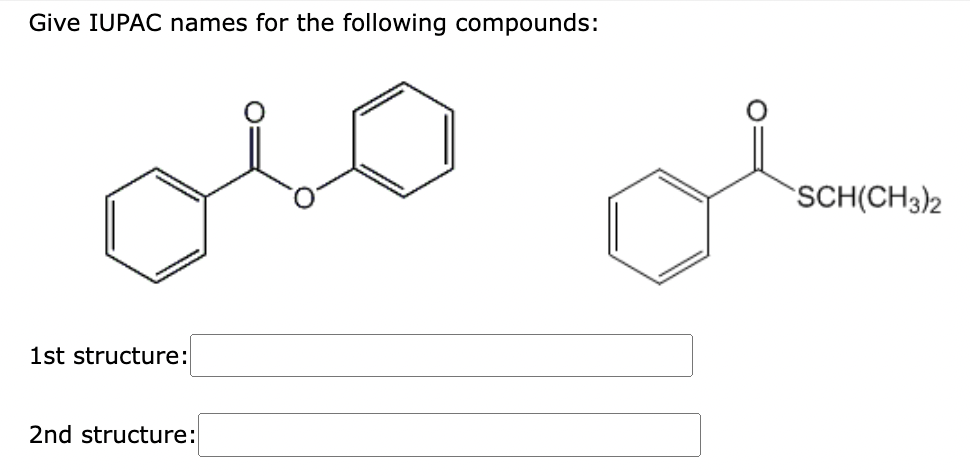
Need help, please.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- a. Draw the structures and give the common and systematic names for alkynes with molecular formula C7H12. Ignore stereosiomers. (Hint: There are 14.) b. How many would there be if stereoisomers are included?Choose one answer for the following. Refer to the diagram below. 1. Hybridization of atom labelled A (sp, sp2, or sp3) 2. Hybridization of atom labelled B (sp, sp2, or sp3) 3. Hybridization of atom labelled C (sp, sp2, sp3) 4.Class compound for functional group D (phenols, aryl halide, alcohols, aromatics, or carboxylic acids) 5. Class compound for functional group E (phenols, aryl halide, alcohols, aromatics, or carboxylic acids)Draw and name all of the structural isomers possiblefor an alkyl halide with no branches and the molecularformula C 5 H 10 B r 2
- Complete the following noting structures for A, B, and C. If more than one step is required to list them as 1, 2, 3, and so on.Briefly, but clearly, explain why the –OH hydrogen in acetic acid (CH3CO2H) is more acidic than in ethanol (C2H5OH).1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. What is the theoretical yield of 2,4-pentanediol expressed in grams? Show calculations. What is the percentage yield of pure 2,4-pentanediol?
- 1,4-Pentadiene (CH2=CH-CH2-CH=CH2) is a liquid at room temperature and has a density of 0.66 g/mL and molar mass of 68.12 g/mol. In a laboratory experiment, 3.80 mL of this compound was treated with 4.80 mL of conc. H2SO4 (100% w/w; molar mass 98.08 g/mol). Note that the density of conc. H2SO4 is 1.84 g/mL. The resulting sulfate ester was then treated with 1.20 mL of water (molar mass 18.02 g/mol) affording, after work- up, 2,4-pentanediol (molar mass 104.15 g/mol) as the crude product. The crude product was then purified by simple distillation, which yielded 2.00 g of pure product. a. Provide a balanced chemical equation to show the reaction between 1,4-pentadiene and sulfuric acid. Do not use molecular formulas in the chemical equation except for sulfuric acid. b. What reactant is the limiting reagent in this chemical equation? Show calculations to support your answer.Place the following compunds in order from lowest to highest boling points. Explain why you have ranked each compound C6H14 CH3OH H2O C3H81. (d) Provide a name for each molecule (I narrowed it down to three subparts)

