Q: 2. The dipole moment of an individual peptide group is approximately 3.46 D. Assuming that the…
A:
Q: 7. The enthalpy of formation of Frenkel defects (kJ mol) is AgCI, 140, AgBr, 109, AgI, 58. The…
A: Frenkel effect is caused if some of the ions of an ionic compound is missing from its crystal…
Q: Aqueous hydrobromic acid HBr will react with solid sodium hydroxide NaOH to produce aqueous sodium…
A: Mass of HBr = 3.24g Mass of NaOH = 2.5 g
Q: Enumerate atleast three examples each for a spotaneous reaction and a nonspontaneous reaction
A: Spontaneous reaction : These are the reactions which occur on their own that means no external…
Q: Calculate the change in entropy that occurs in the system when 23.0 g of isopropyl alcohol condenses…
A:
Q: 32. Which of the following statements about tetramethylsilane is incorrect? A. it produces a single…
A: The reference compound or internal standard compound used in NMR is tetramethylsilane (TMS).
Q: e. Calculate the pH of a solution containing 50.0 mL of 0.150 M of benzoic acid after the following…
A: First we would Calculate moles of both acid and base. Then we would draw an ICE table to calculate…
Q: Select from the following which steps are involved in the natural production of ozone in the…
A: In the stratosphere, the concentration of ozone is the maximum. The ozone gas is produced naturally…
Q: grams of HF the limiting reactant is? SiO2+4HF=SiF4+2H2O
A: Answer: SiO2 + HF = Weight 3 g…
Q: (Calculate the number of grams in one mole of CaS04.7H20, (Ca = 40, S=32, 0=16, H=1 .A g/mol 136 .B…
A:
Q: CIve an accēptable name for each amine. a. CH;CH,CHCH3 NH2 C. -N(CH)2 H. b. CH;CH,CH,NHCH3 d. f.…
A: We would use IUPAC nomenclature rules to name the given animes.
Q: Give a clear handwritten answer and explain
A: We would check boiling point to check the strength of intermolecular forces.
Q: Activity 6. Working with pH, pOH, [H30°), and (OH] A solution has a pH of 3.45. You may answer the…
A: pH = - log [H3O+] [H3O+] =10-pH pOH =- log [OH-] [OH-] = 10-pOH pH + pOH = 14 If the…
Q: Gaseous butane will react with gaseous oxygen to produce gaseous carbon dioxide and gaseous water…
A: Ethane reacts with oxygen to form carbon dioxide and water. The equation for the balanced chemical…
Q: HO, H* HO, SOCI2 SO2CI Рyridine NaH HO NABH4 МеОН Br
A: In a given question , we have to do mechanism of different reactions in different condition
Q: Consider the reaction below. If you start with 2.00 moles of C3H3 (propane) and 2.00 moles of 02 ,…
A: Given :- C3H8(g) + 5O2(g) → 3CO2(g) + 4H2O(g) No.of moles of C3H8 = 2.00 mol No.of moles of O2 =…
Q: Give a concise svnthesis for the molecules if they start from a benzene. a. F- -Br b. Но. OH C.
A:
Q: 8. What organic starting materials are needed to synthesise the compound below by reductive…
A: In a reductive amination reaction, carbonyl compound reacts with ammonia or amine to form an imine…
Q: emical apparatus te easure potassium?
A: Answer: to measure the potassium the method are used atomic absorption or inductivity coupled…
Q: HCI C,H,CI OH CH,O Na,Cr,O, C,H,O Compound A H20 Compound B Compound C
A:
Q: If 3.0 grams of KClO3 are reacted and 1.5 gram of KCl is actually collected, what is the percent…
A:
Q: Determine the mass of nitrogen that can be formed from the reaction of 50.0 g N2O4 and 45.0 g N2H4.…
A:
Q: Question 5 Listen A 28.5 inch long AISI 1040 steel bar is heated by 61.5°F. Calculate the change in…
A:
Q: Bromine is used in the manufacture of disinfectant products, it has a very strong oxidizing…
A:
Q: For an ideal gas: 1. Assuming that all quantities are held constant except for pressure and…
A: #1. Given all the quantities are constant except P and T.
Q: How many grams of O2 are required to completely react with 100.0 grams of Fe in the following…
A:
Q: Practice ools Add-ons Help Accessibility Last edit xt Arial 18 BI (ANSWER reure raised by 21a, How…
A:
Q: half-life
A:
Q: Carbon Nuclear Magnetic Resonance Spectroscopy (C-NMR, or "C-NMR) Experience of chemists for C-NMR…
A: The correct answer about C-13 NMR spectroscopy is given below
Q: ball & stick + labels The molecule has a carbonyl group. A general functional group representation…
A: Given, Based on ball and stick model the structure of the molecule is:
Q: The reaction between ammonia and oxygen is given below: 4 NH3(g) + 5 02(9) - 4 NO(g) + 6 H20(g) We…
A:
Q: HCO3- + HCl ==== H2CO3 + Cl- . Which one of reactants is an acid?
A:
Q: Calculate the entropy change of the surroundings in J K-1.
A: The entropy change of the surrounding is needed to be calculated.
Q: 2.) Indicate whether each statement is true or false. (a) a reaction that is spontaneous in one…
A: If the change in free energy is negative the reaction will be spontaneous in the forward direction…
Q: The reaction between nitrogen monoxide and oxygen is given below: 2 NO(g) + 02(g) 2 NO2(9) We…
A:
Q: 2. Complete the following equation by drawing the most plausible structure for compound A, compound…
A: When a protic acid (HX) is added to an asymmetric alkene, the acidic hydrogen attaches itself to the…
Q: What is the source of energy for the heat of reaction in the neutralization reaction? Hint: Consider…
A:
Q: For p=0.1, the activity coefficient (gamma) of M2+ is 0.45 while gamma of X- is 0.80. Calculate how…
A: At ionic strength of 0.1, the activity coefficient values are: γM2+ = 0.45γX- = 0.80 Ksp = 3.2 x…
Q: 231.25 g/mol) is an Isocarboxazid (MW organic monoamine oxidase inhibitor used to treat depression…
A: The pressure created by the flow of solvent molecules from a less concentrated solution to a more…
Q: वाह क्रण्ला उपलणणर जला कषट अच्पलिपट the For ळा्चनीयवं हवौवै- Phिवगर अभतजीच्य ह, what is the function…
A: We have to tell the function of the di-tert-butyl-dicarbonate in the Merrifield solid-phase peptide…
Q: What is the frequency of light having a wavelength of 456nm?
A: Given :- wavelength of light = 456 nm To calculate :- frequency of light
Q: a. Propose a plausible mechanism for the following Heck reaction TBSO Pd(PPH3)4 K2CO3 H. OTBS 2.
A:
Q: 18.26 Synthesize each of the following compounds from diethyl malonate or ethyl acetoacetate and any…
A:
Q: Select which molecule is the better nucleophile in the following pair: a). Br− or Cl− in H2O b).…
A:
Q: CO, (aq) OH (aq) CI (aq) | (aq) NO3 (aq) , (aq) 5o, (aq)
A: Answer: unknown bottles (solution)in set 1 are bottles A- E . NaOH ( aqueous ) was kept in…
Q: consider the reaction below 3 Mg (s) + AI203 (s) > 2 AI (S) + 3 MgO(s) if 68.9 g of AI2O3 (S) are…
A: Given mass & molar mass--> mass of Al2O3=68.9 g molar mass of Al2O3=102 g mol-1 molar mass of…
Q: List the following from lowest to highest first ionization energy. Cl, Na, K
A: Ionization energy : The energy required to remove the highest energy electron i.e.valence electron…
Q: The conjugated diene 1,4-diphenyl-1,3- butadiene has a network length of 723 nm. Using the particle…
A:
Q: What effect would you expect the following changes to have on the rate of the Sy2 reaction of…
A: Here we are required to predict the effect on the rate of reaction when the concentration are varied…
Q: The combustion of isoctane reaction produces -5099.5 kJ of heat per mole of C8H18 (g) consumed,…
A: Given data , □Hcomb = -5099.5 kJ of heat per mole of C8H18 (g) (□Hf) for CO2(g) = -393.5 kJ/mol…
Explain with arrows and Give Hand written answer
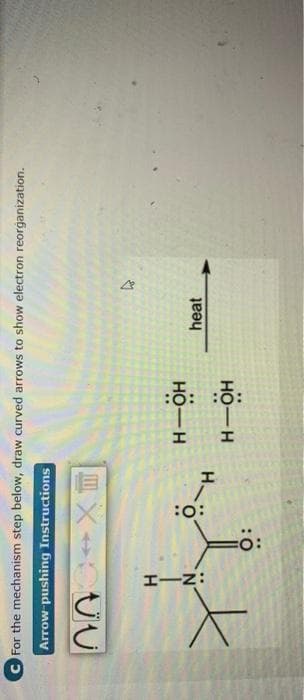
Step by step
Solved in 2 steps with 2 images

- Orgonic Chemistry II: The answer is writtten as followed. But I Need Explanation. My question is that: Can I siwtch reagent 2 to reagent 1? for example reagent 1 is Cl2,Fecl3, can I changed it to reagent 2??? Why and why not???Step 4c: Draw the resulting structure or structures indicated by the curved‑arrow mechanism shown. Include formal charges, but not lone pairs.kindly answer from C to E
- How do I get to my target molecule (1st image) with the following uncomplete reaction (2nd image)Carbocations often rearrange, as shown below. Draw in the hydrogens on the two carbons involved in the rearrangement, and show formal arrow-pushing to illustrate the transformation.The reactants, intermediates, final products, and all curved arrows showing bonds forming andbreaking are collectively referred to as the mechanism of a reaction. For the following reactants: a. Explain why the original statement of Markovnikov’s rule does not help in this case, but themodern restatement of Markovnikov’s rule tells you which carbon will get the X (Cl). b. Show the mechanism of the most likely addition reaction between the reactants.
- 1. Which absorbs at higher frequencies: a C-H or C-D bond? Explain. 2. Predict which alkene will predominate at equilibrium. Explain.#6). Second picture shows instructions on how the arrows must be shown as.Can you draw the products that form when 1-butene reacts with; O3, then Me2S OsO4, then NaHSO3/H2O Br2 in H2O Please analyze in detail for each. Also can you please reply fastly?

