Q: A biochemist who is studying the properties of certain sulfur(S)–containing compounds in the body…
A: Sulphur belongs to group 16 of the periodic table , i.e, chalcogen family . Its atomic number and…
Q: Give a Carrect formula between Ca (9) Ca₂1 (b) Calz (e) C92 13 C9312 and I A
A:
Q: what is the difference between fe(s) and fe3+ at the atomic level?
A: When the neutral atom losses electron then it forms cation. When the neutral atom accepts electron…
Q: 23Na,88Sr2+,Se,Br-,35CI. Which of the following are ions ? Provide reason
A: Given, 23Na, 88Sr2+, Se, Br-, 35CI
Q: Qu) Fill the blanks 1) The analysis that quantifies analytes called. 2) The most abundant element in…
A: Fill in the blanks _____ 1) Quantitative Analysis 2) Hydrogen is the most abundant element in…
Q: Provide the molecular formulas for the following compounds Aluminum bromide Gallium phosphide…
A: Molecular formula of ionic compounds can be written by following electrical neutrality principle…
Q: I. Provide a systematic name for each of the following compounds O,N. OH O,N. H он (d) "Br (n)
A: For these given compounds, the determination of position of the substitutions either in the aromatic…
Q: What is the Chenical Formula of the 1. Aurous selenide 2. Yttrium following: Phosphide 3. Hydronitic…
A: We will solve only first three subparts from given problem according to Bartleby guidelines. Write…
Q: Give systematic names for the following compounds: ОН HO, NH2 Br
A: Select the principle carbon chain Numbering Naming as per alphabetical rule (prefix + word root +…
Q: 111 5. Explain the reasoning behind the following properties of ionic compounds. a. brittle b. high…
A: When the two elements with high electronegativity difference combined forms the ionic compound .
Q: 2 6 3 5 9 The erroneous part in the illustrated set up is 4 O 9 O 1 O 6 7 8
A:
Q: MeetLeam (b) The organic compound P, named , 2-hyd roxylpropanenitrile exhibits isomerism, (i) Draw…
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Rank the following acid in order of increasing strength (weakest to strongest) and explain you…
A:
Q: PKA o
A: Amino acid: An organic compound containing both ammonia and carboxylic group is called amino acid.…
Q: 3. Write the equations for the following reactions: a) potassium oxide reacts with water b) cesium…
A: a) Formula of potassium oxide = K2O Formula of water = H2O
Q: Đ1. Docs the charge of the material remain the same when paired up with other material? 02. What is…
A:
Q: Write the chemical formula of the compounds involved in the following reactionsbelow. Indicate…
A: Chemical formula is a representation of a compound by denoting fixed proportion of elements in…
Q: n hydrogen and CO-?
A: An acid is defined as a chemical substance which contains hydrogen and can react with bases in order…
Q: II + Ca(OH), - D
A:
Q: Describe the formation of positiveand negative ions.
A: To find: Description about the formation of positive and negative ions
Q: Briefly explain the following observation. S can have an expanded octet and thus SF6 exists (~$1.00…
A:
Q: Why does maleic acid react with mg metal?
A: Maleic acid is a dicarboxylic acid. It is cis- isomer which can attain intra-molecular bonding with…
Q: Which of the following values is the overall charge of the dominant form of His at pH 11.0? O 2 O 2…
A:
Q: Sequestering agent for Benedict’s Solution:
A: In this we have to know about sequestering agent for Benedict's solution and salts of these alkaline…
Q: For each of the following, give the most specific classification according to type of matter:…
A: Generally, each and everything in the universe that has some mass and some space is known as matter.…
Q: Give formulas corresponding to the following names:(a) Tetraamminezinc sulfate (b)…
A:
Q: What is the pKa of the NH4+ ion?
A: Given, NH4+ ion
Q: What are the charges of germanium and antimony in Gd3Sb4
A: Metals take positive change after losing the electrons whereas non-metals take negative charge after…
Q: oblems: Provide the name for the following compounds. F (a) (b) (c) Br (d) (e) (f)
A: A. (3S,4R,6R,7S)-4-chloro-6-floro-3,7-dimethyl nonane. B. (3R,4R)-4-ethyl-2,3,7,7- tetramethyl…
Q: write ће тирас Name K+ [2₂ (1₂0) (1) TCCB, CN), 2 Hg (H₂)₂ OHJON Each of the Following
A: RULES FOR IUPAC NAME:- Name ligands and arrange according to alphabetical order. Add prefix for…
Q: The charge on the iron in the salt FeF3 is
A: In chemistry, different type of compounds made by different type of atoms by sharing, gaining or…
Q: What is the name for the compound NO2?
A: In the given question, the name for the compound NO2 has to be determined. The given compound NO2…
Q: Of the following reactions below, which represents the second ionization energy of calcium? O Ca(g)…
A:
Q: What is the charge on the Cr ions in Cr2O3?
A: We have Cr2O3 , we have to calculate the charge on Cr ion.
Q: AH AS and AG° for
A:
Q: Zinc iodide will have the same appearance and properties as which of the following? zinc sulfide…
A: Zinc sulfide is coreect answer It is white hexagonal crystals which is a form of solid. Zinc…
Q: The name of P205 is_ A diphoshorous pentoxide B phoshorous dioxide diphoshorous oxide phosphate
A: Answer The name of P2O5 compound is
Q: 6)
A:
Q: give the charge of the transition metal Ag2C2O4
A: Answer : Charge of transition metal Ag is +1. Ag2C2O4 siver oxalate is a ionic compound. Which…
Q: which cation in KAlSi308 could Sr^2+ substitute for? In order for this substitution to occur, what…
A: Given : We have to tell what will happen when KAlSi3O8 reacts with Sr2+ .
Q: Calculate the bonder of O22.
A: The ion given is O22+.
Q: Give an example of ionisation isomerism.
A: Given: Explain ionisation isomerism with example.
Q: Calculate the formula of a compound having the following composition: Na = 27.38 %, H = 1.19 %, C =…
A:
Q: Explain the following observations: (i) Transition elements generally form coloured compounds. (ii)…
A: Elements which have partially or incompletely filled d-orbitals in their ground or stable oxidation…
Q: (a) What are the central metal ions in chlorophyll, heme,and vitamin B₁₂? (b) What similarity in…
A: The given problem deals with Bio-Inorganic chemistry. It is the branch of chemistry which involves…
Q: Given the following reactions: 3C =D Kc= 8.33 × 10¬4 %3D D = 3A+ 3B K,=3.32 × 10¯2
A:
Q: |NazO positive ions negatite ions b Feo
A:
Q: Predict the chemical formula for the ionic compound formed by the ions Ca²⁺ and PO₄³⁻.
A: Given ions are Ca2+ and PO43- . Here , Ca2+ is a cation and PO43- is an anion.
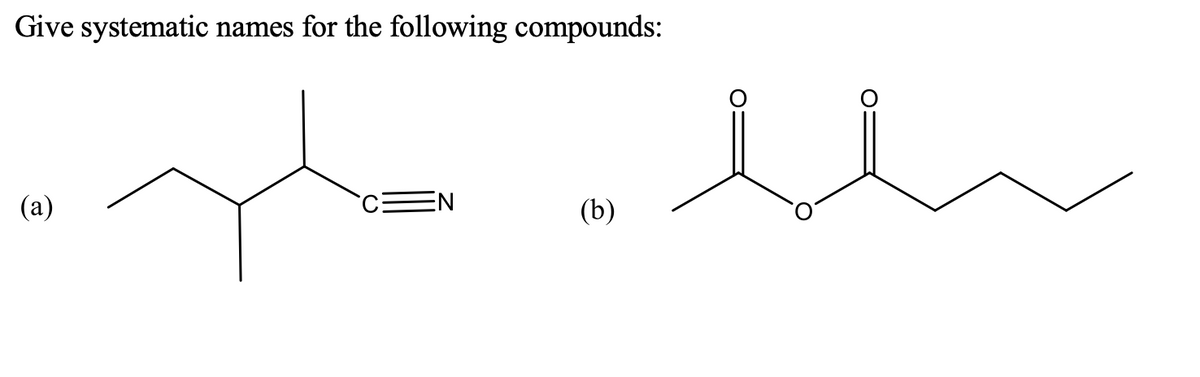
Step by step
Solved in 2 steps with 1 images

- If C2H6 was converted into C2H2 in a reaction, then was the C2H6 oxidized or reduced? Explain the answer by using oxidation states for the carbon in the two compounds. EN Values: C=2.5 ; H=2.1Give the IUAPC name of the following compounds.Give the chemical names of the corresponding structures for compounds A and B.

