Give the appropriate label for each of the following VBT prbitals for Note the standardized colour coding for atoms in these diagrams: oxygen (red), sulfur The orientation of the molecule is as shown in the perspective diagram on do the right. nitrogen (blue), carbon (grey), and hydrogen white) 1. SO 6, (S-O sigma b) 2. SO o" (S-O sigma star) SỐ n,(S-O pi b) 4. SO n (S-O pi star) 5. S sp (S sp) 6. O sp' (O sp) 7. SC o, (S-C sigma b) 8. SC a* (S-C sigma star) 9. CO 0h (C-O sigma b) 10. CO o* (C-O sigma star) 11. none of the above 3.
Give the appropriate label for each of the following VBT prbitals for Note the standardized colour coding for atoms in these diagrams: oxygen (red), sulfur The orientation of the molecule is as shown in the perspective diagram on do the right. nitrogen (blue), carbon (grey), and hydrogen white) 1. SO 6, (S-O sigma b) 2. SO o" (S-O sigma star) SỐ n,(S-O pi b) 4. SO n (S-O pi star) 5. S sp (S sp) 6. O sp' (O sp) 7. SC o, (S-C sigma b) 8. SC a* (S-C sigma star) 9. CO 0h (C-O sigma b) 10. CO o* (C-O sigma star) 11. none of the above 3.
Principles of Modern Chemistry
8th Edition
ISBN:9781305079113
Author:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Chapter5: Quantum Mechanics And Atomic Structure
Section: Chapter Questions
Problem 46AP: For the Li atom, the energy difference between the ground state and the first excited state, in...
Related questions
Question
100%
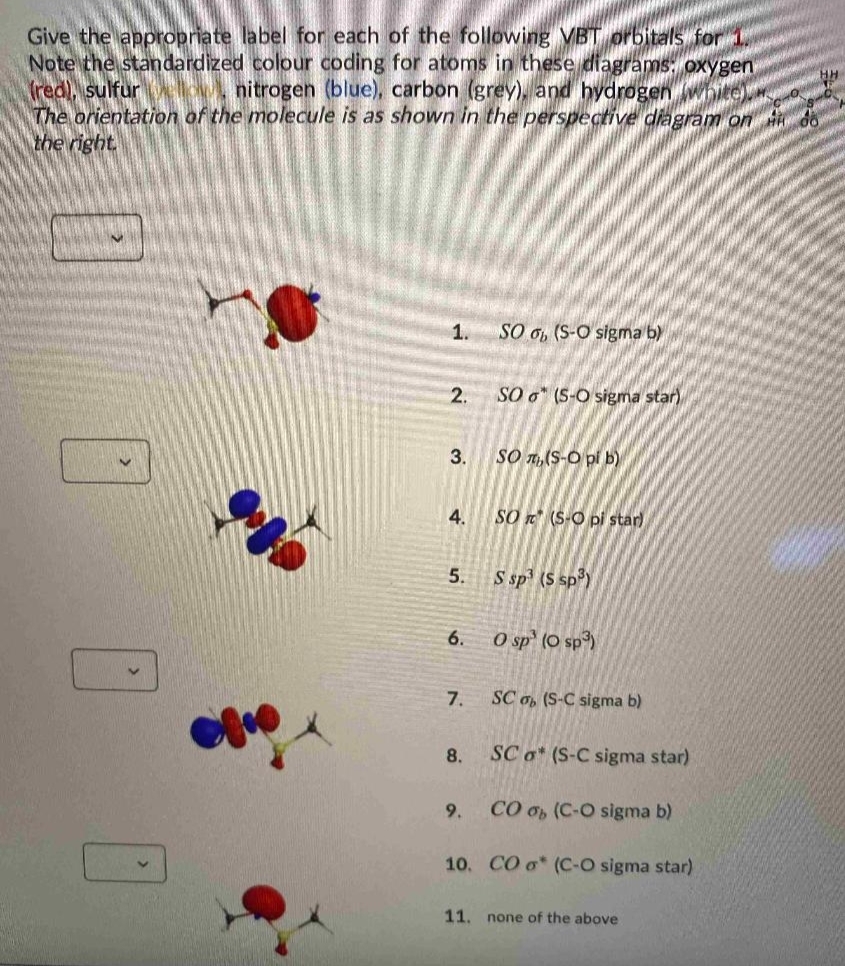
Transcribed Image Text:Give the appropriate label for each of the following VBT orbitals for
Note the standardized colour coding for atoms in these diagrams: oxygen
Tred), sulfur O, nitrogen (blue), carbon (grey), and hydrogen w
The orientation of the molecule is as shown in the perspective diagram on
the right.
1.
SO o, (S-O sigma b)
2.
SO o" (5-O sigma star)
3.
SO T1,(S-O pi b)
4.
SO (S-O pi star)
5.
S sp (S sp)
6.
O sp" (O sp³)
7.
SC o, (S-C sigma b)
8.
SC o* (S-C sigma star)
9.
CO 0h (C-O sigma b)
10. CO o* (C-O sigma star)
11.
none of the above
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781133949640
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Introductory Chemistry: An Active Learning Approa…
Chemistry
ISBN:
9781305079250
Author:
Mark S. Cracolice, Ed Peters
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning