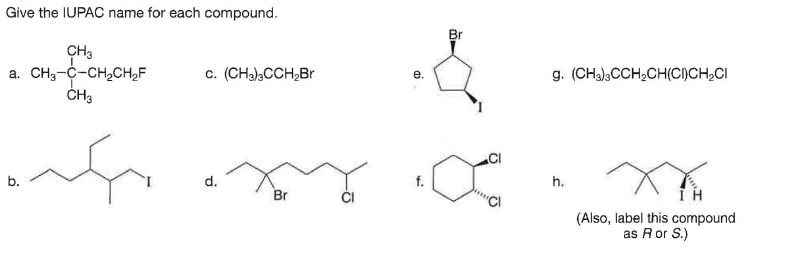
Give the IUPAC name for each compound. Br CH3 a. CH3-C-CH2CH,F ČH3 c. (CH),CCH,Br g. (CHa),CCH,CH(CI)CH,CI е. b. d. h. Br (Also, label this compound as Ror S.)
Q: Give the IUPAC name for each compound. a. (CHa)3CCH,CH(CHCH3)2 b. CH3(CH2)3CH(CH,CH2CH3)CH(CH32 d.
A: The names of the following compounds are as follows:
Q: 2. Give the IUPAC name for each compound. CH, CH,CH2CH3 CH3 CH3 CH3 CH2CH3 CH,CH2. CHCH2CH3 a. b. CH…
A: The IUPAC is a system that contains some rule that helps in the naming of the organic compounds. The…
Q: Give the IUPAC name for each compound. a. H-C=C-CHC(CH,CH;CH3)3 b. CH3C= CC(CH3)CICH,CH3 c. CH2=…
A: Since we only answer up to 3 sub-parts, we’ll answer the first 3. Please resubmit the question and…
Q: но NBS HBr ether CCI, a. b. Mg, ether SOCI, Br H,0 d.
A: ->HBr give substitution reaction through formation of carbocation. ->NBS give allylic…
Q: Rank these alkenes according to stability:
A: Alkenes stability can be explained by "hyperconjugation" Displacement of alpha C-H sigma…
Q: H₂N- -Br H₂C Provide the locant and substituent in the order that they will be written in the name.…
A: Interpretation- As here we have to tell the locant and substituent present in the given organic…
Q: Give the IUPAC name of each alkyne. (a) CH3 CH3 (b) CH3 CH3CHC=CCHCH3 HC=CCCH3 CH3 (c) CH3 (d) CH3…
A:
Q: Circle and the functional group (5) n each compound. name キ a. CHy CH3 Br b. CH3 -CH CH = CH-CHz- NH…
A: The groups which are responsible for the chemical reactivity are known as functional groups.…
Q: Give the IUPAC name for each compound. Br F а. (CHa)2CHCH(CI)CH;CH3 b. С. d. Br
A:
Q: 1) Write the IUPAC name for each molecule below. b. a. HN. N'
A: To find: The IUPAC name of the given compounds.
Q: Arrangement of more stable ? and why? CH3 Cニ Br Arrangement of more boiling point ? CH3 cHs CHs…
A:
Q: OH 2 points 10. 11. HO CH2
A: We have to give the IUPAC name of the following compounds :
Q: 1. Give the IUPAC name for each compound. a. e. b. 2. Give the IUPAC name each compound. CH3 CH3 CH3…
A: First question is based on IUPAC of alkane Rule: 1)select the longest carbon chain Rule:2) follow…
Q: (a) excess NH3 + Ph-CH₂ CH₂ CH₂ Br (b) 1-bromopentane (1) NaN3 (2) LiAlH4 (3) H3O+
A:
Q: Give the IUPAC name for each alkyne. a. CH;CH,CH(CH,)c=cCH,CH3 d. HC=C-CH(CH,CH3)CH,CH,CH3 g. CH3 e.…
A: IUPAC name is as follows
Q: Which compound will give a positive test with Br2/CH3Cl, a positive test with HCl/ZnCl2, and a…
A:
Q: Give the IUPAC name for each alkyne. a. CH,CH,CH(CH3)C=CCH,CH3 d. HC=C-CH(CH2CH3)CH,CH,CH3 g. CH3 e.…
A:
Q: HCI CH, но. CHs Solvent: Toluene H;C CH3 .CI KOC(CH 3)3 Solvent: Toluene -CH3 CH3
A:
Q: the reaction of 1-butyne with the four hydrogen atoms result .A CH3-CH2-CH2-CH3 .B CH2=CH-CH2-CH3 .C…
A:
Q: 3. What is the IUPAC name for a,b,c and the common na CH2-CH3 CH3-CH2-CH-CH2-CH-CH3 Br a. b.…
A: Rule of IUPAC- 1) Longest chain as parent chain. 2) Numbering start from those side where more prior…
Q: Give IUPAC names for the following substances: (b) (a) CH3 CH3CHCH₂CH₂CCI (9) H₂C=CHCH₂CH₂CNHCH3 (e)…
A: Esters are alkyl derivatives of carboxylic acids. IUPAC name of an ester is alkyl alkanoate as it…
Q: a.) Classify the following carbocations (1°, 2°, or 3°). b.) Encircle the carbocation/s that can…
A:
Q: 3.) Butanedionic Why completely soluble in water? Why insoluble in HCl? Why completely soluble in…
A: Solubility :- It is the property of the compound which have tendency to soluble in a…
Q: Write down the IUPAC name and structure of the followings (i) HOH,C-CH = CH - CH,OH (ii) H.C…
A:
Q: Mant The atcohol! ) CHg CHy OH 2) CH, CH CH, CH, OH 3) CHg CHs CH CH, CH, OH 4) CHgC=C CH2 CH_CH3 5)…
A: During the IUPAC naming of an alcohol first select the principal chain i.e longest possible carbon…
Q: Oxidation of a 1° alcohol with K2CRO4 yields
A: oxidation of primary alchol converted into an CARBOXYLIC ACIDS in presence of K2Cr2O4 Eg;n- propyl…
Q: f) g) h) i) j) Br of CH3 H3C CH3 Br 1. NaOEt 2. H₂, Pd/C Hg(OAc) 2 H₂O, THF -C=C-CH3 -MgBr (xs) Na…
A:
Q: O Give the IUPAC names of the following alkyl halides: Br (b) çI ÇH3 (c) CH3 CH;CHCH;CH;CI…
A: Detail description is given below
Q: DIBAH toluene H.O 1. H,O* HGSO 2. он HO- 3. CH,CHMgBr 4.
A: 1) The ethyl phenyl acetet reacting with the Di-isobutyl Aluminum Hydride (DIBAL) and…
Q: H;C „CH3 ? CH3 starting material final product
A:
Q: Is the reaction of 2-butene with HBr regioselective? a. Is it stereoselective? b. Is it…
A: Please go through the below pic for the reaction mechanism.
Q: Show the reagents and experimental conditions necessary to bring conversion in good yield.
A: The given reaction is, CH3CH=CHCH3→CH3CH=CHCH2COOH The reactant is But-2-ene and the product is…
Q: 1. Give the IUPAC name of the following: CH3 | a. CH3CHCHCH2CH2COOH CH3 b. CH3CHCHCH2COOH Cl H2…
A: a. 4,5-dimethylhexanoic acid b. 4-chloropentanoic acid c. N-ethylpropanamide
Q: 4.23 Which alkenes can exist as pairs of E/Z isomers? For each alkene that does, draw both isomers.…
A: E/Z are the stereoisomers of alkene.
Q: Name each compound. OCH, a. CH,-C-CHCH, b. CH,CH,CH,CH,CHCHO Br CH,CHCH,CH,-C-H C.…
A: a. The name of the given compound is given below along with its structure.
Q: 8. CHOH Dess Martin Periodinane CHC 9. CH pH 4.5 H. 10. KCN HCN 11. (CHP-CHCH, THF 12. ether 2. H0
A:
Q: Draw the products of combustion of each alkane.
A: Combustion is a process where the alkanes are react with oxygen to give coarbondioxide and water…
Q: -CH3 KOC(CH 3)3 H3C CH3 Solvent: Ethanol HCI Hc-OH Solvent: Ethanol CH3 KOCCH 3)3 Solvent: Toluene…
A: Tertiary butoxide is a base and it will only abstracts the proton.
Q: 15 45 Circle and name the functional group(s) in each compound: (a) CH,-CH=CH-CH2-OH (b) CI-CH, C-OH…
A: Functional group are the group of molecules or atoms in the compound with distinguishable properties…
Q: Thiols Molar mass a. CH3-SH in oyster and cheddar cheese C- H- b. CH3-CH2-CH2-SH in freshly chopped…
A: Percentage of any component may be obtained by dividing the mass of that component by the total mass…
Q: What reactions are needed to convert alcohol A into either alkyne B or alkyne C?
A: Alcohol reacts with TsOH to form tosylate with retention of configuration. Tosylates are good…
Q: A. Provide a reasonable method for the synthesis conversion below. You may use any organic or…
A: Please find the attachment.
Q: NaCI CH4 CH3-0-CH3 |CH3-CH2-OH Acetone (CH3COCH3) Нехane, CgH14 CH3-CH2-CH2-CH3
A: The forces of attraction or repulsion that act between neighboring particles such as atoms,…
Q: H2C=CHCH=CH2 CH;CCH,CH,CH,CH,CH3 Reagents a. Br2, CH2CI2 b. KCN, HCN LIAIH, then H3O* d. H2, Pd/C…
A:
Q: Neopentyl alcohol, (CH,),CCH,OH, cannot be dehydrated to an alkene without rearrangement. What is…
A: We have to tell which option is correct
Q: || 6. H- C-O-CH: 7. CH3-CH2-CHz-O-C-CH: CH: || 8. CH3-CH2- C-O-CH-CH3 lookat
A: IUPAC naming of the given compounds. As per our guideline we have to answer first three questions…
Q: Four alkyl groups bonded to carbon marked * in the molecules shown below are: CH3 IV CHCH3 CH3…
A: Ethyl group - C2H5-- sec - sec is used when there the group is attached at second carbon of…
Q: Give the IUPAC name for each alkyne. CH;CH,CH(CH,)C=CCH,CH3 b. (CH,),CHC=CCH(CH3)2 С.…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 3 images

- Indicate whether each statement is true or false. (a) Butanecontains carbons that are sp2 hybridized. (b) Cyclohexaneis another name for benzene. (c) The isopropyl group containsthree sp3-hybridized carbons. (d) Olefin is anothername for alkyne.Rank the following groups in order of decreasing priority. −H, −CH3, −Cl, −CH2Cla. What alkane, with molecular formula C5H12, forms only one monochlorinated product when it is heated with Cl2? b. What alkane, with molecular formula C7H16, forms seven monochlorinated products (disregarding stereoisomers) when heated with Cl2?
- Explain why the addition of HBr to alkenes A and C is regioselective, forming addition products B and D, respectively.Explain why the addition of HBr to alkenes A and C is regioselective,forming addition products B and D, respectively.Consider the attached tricyclic structure B. (a) Label each substituent on the rings as axial or equatorial. (b) Draw B using chair conformations for each sixmembered ring. (c) Label the atoms on the ring fusions (the carbons that join each set of two rings together) as cis or trans to each other.
- Consider the tricyclic structure B. (a) Label each substituent on the rings as axial or equatorial. (b) Draw B using chair conformations for each six-membered ring. (c) Label the atoms on the ring fusions (the carbons that join each set of two rings together) as cis or trans to each other.1. a) write the product 2-methyl-3-ethyl-2-hexene -----O3------> Zn, H30+ b) Name this compound according to the IUPAC system: (CH3)2CH-CH2-C≡C-CH-CH2-CH2F | CH31. Using Br2 in C2H4Br2 will result in HBr and ______. a. C2H3Cl3 b. C2H4Cl3 c. C2H2Cl3 d. none of the above 2. How many halogenation are posible in propane? a. 3 b. 8 c. 6 d. 10 3.Sulfonation of pentane will result in ________ and water. a. C5H11SO3H b. C5H12SO3H c. C5H14SO3H d. none of the above 4.Nitration of hexane will result in ________ and water. a. C6H13SO3H b. C6H15NO2 c. C6H13NO2 d. C6H14NO2 5.How many moles of O2 in heating a C12H26 (dodecane) a. 27 b. 37 c. 24 d. none of the above
- The skeletal line formula for a branched alkene is shown below. (i) What is the molecular formula of this compound? (ii) How many carbon atoms are in the longest chain, ignoring the double bond? (iii) What is the longest chain incorporating both carbons of the double bond? (iv) How many substituents are on this chain? (v) Give the IUPAC name for this compound. [6]Is the reaction of 2-butene with HBr regioselective? a. Is it stereoselective? b. Is it stereospecific?Tell the name and tell the R/S or E/S or cis/trans (IUPAC)

