Q: Determine the value of Ksp for Zn (PO4)2 by constructing an ICE table, writing the solubility…
A: Step 1: Recall that pure substances (solid and liquid) are not included in an ICE table. Let x be…
Q: 25 26 27 28 29 3 8 3.5 4 4.5 8 288 401 8 201 20 10 5 4. Molecular weight = 86; with 10 MICRONS 5.5 6…
A: Rule of thirteen is a simple mathematical calculation which can be used to determine the molecular…
Q: None
A: The objective of the question is to calculate the mass of ethane gas in a cylinder using the ideal…
Q: A 54.0 L container holds 6.27 moles of oxygen gas, O2, and 4.19 moles of neon gas, Ne, when the…
A: The objective of this question is to calculate the pressure inside a cylinder containing oxygen and…
Q: 10. (3 pts) Draw the most likely mechanism for each of the following transformations: a. NO₂ b. Br…
A: 1: This is an aromatic nucleophilic substitution. rate of nucleophilic aromatic substitution the…
Q: Highlight each of the positions on the benzene ring that you expect will be most likely to react by…
A: In electrophilic aromatic substitution, substituents direct the position of the other…
Q: The reduction of iron(III) oxide (Fe2O3) to pure iron during the first step of steelmaking, 2…
A: Step 1:Given chemical equations are;2Fe2O3 (s) → 4Fe(s) + 3O2(g)C(s) + O2(g) → CO2 (g) On combining…
Q: Compare and contrast the amount of caffeine obtained from the single vs multipleextractions with Red…
A: Overall, the multiple extractions with Red Bull and Coca-Cola beverages result in higher…
Q: 2. How many moles of HOCI should be present together with 0.50 mole of Ca(OCI)2 in a 1.00 L solution…
A: Step 1:Step 2:Step 3:Step 4:
Q: A solution is prepared at 25 °C that is initially 0.41 M in chloroacetic acid (HCH2CICO2), a weak…
A:
Q: Will Fe, Fe 2+, or Fe 3+ react more favorably with CO? Explain your reasoning.
A: •CO as a Ligand: CO (carbon monoxide) is known as a strong ligand. In chemistry, ligands are…
Q: A 23.0-liter gas cylinder contains 8.6 moles of nitrogen gas and 1.6 moles of oxygen gas. If the…
A: We can find the total pressure inside the cylinder using the ideal gas law. Ideal Gas LawThe ideal…
Q: None
A: Approach to solving the question: Detailed explanation:a) Markownikov reaction.b) Addition reaction…
Q: A 10.0 L sample of carbon dioxide gas has a temperature of 500.0 K. If the volume of the gas is…
A: The objective of this question is to find the final temperature of the carbon dioxide gas when the…
Q: C H- -OH H- -OH H- -OH CH₂OH H -OH -OH CH₂OH OD CHO H- -OH H- -OH HO- H CH₂OH ○ E CHO -OH HO- -H…
A: Step 2: Step 3: Step 4:
Q: Olympic cyclists fill their tires with helium to make them lighter. Calculate the mass of air in an…
A: Volume of the tire (V) = 855 mLTotal pressure of the tire (P) = 125 psiTemperature (T) = 25Molar…
Q: Refer to the attached compound and draw/represent the Haworth cyclic structure projection of the…
A: To draw the Haworth cyclic structure projection of β-Mannose between C1 (carbon 1) and C5 (carbon…
Q: 1. A wet gas from a field in Iran is processed through two stages of separation; first stage…
A: The objective of the question is to calculate the specific gravity of the reservoir gas, the average…
Q: b) DRAW THE product of each of the following reactions? DRAW expected product stereochemistry, where…
A: b) Heck Reaction: Stepwise Explanation The Heck reaction is a palladium-catalyzed C-C coupling…
Q: Given the information A+B 2D C D ΔΗ° = 739.8 ΚΙ AH° = 453.0 kJ AS 314.0 J/K AS-167.0 J/K calculate…
A: The objective of the question is to calculate the Gibbs free energy change (ΔG°) for the reaction…
Q: hello, can you please this according to chemical kinetics?
A: t(min)CA (mol/L)02.00101.00300.50400.40700.25Step 1: Plot this CA vs. time relationship. This will…
Q: Payalben
A: The objective of this question is to calculate the volume of gas inside a balloon given the…
Q: Part II: 1. Draw the mechanism and the energy diagram for the reaction shown below. Include any…
A: Here in this question H3O+ given which is a source of H+ and H2O.
Q: A solution is prepared at 25 °C that is initially 0.53M in acetic acid (HCH, CO₂), a weak acid with…
A:
Q: Draw the most likely conjugate base resulting from this acid-base reaction. Include all lone pairs.…
A: Step 1: When a proton is removed from hexanenitrile, it becomes its conjugate base (also known as…
Q: Which statement is TRUE for a precipitation reaction? The driving force is the formation of a…
A: The objective of the question is to identify the correct statement about a precipitation reaction. A…
Q: None
A: Approach to solving the question:1. Calculate the moles of NaH2PO4 and Na2HPO4.2. Determine the…
Q: If you spike the sample you collected with a known amount of the analyte of interest, you are…
A: The objective of the question is to identify the calibration method used when a known amount of the…
Q: For the reaction shown, draw the product of one mole of reagent adding across the triple bond, and…
A:
Q: Select the correct product for the following SN2 reaction. Br NaSH A SH HS H B H C O A ○ B C D Br HS…
A: Step 1: Step 2: Step 3: Step 4:
Q: None
A: Step 1:- First calculate number of moles of oxygen atoms in 12.34 mg of aluminum chlorate.Mass…
Q: www-awu.aleks.com/alekscgi/x/Isl.exe/10_u-lgNslkr7j8P3JH-IQUHIQg6bJxmeSyVPHOEB1plef9xyC5Ca9QIn2eRVQ-…
A: Step 1: Step 2: Step 3: Step 4:
Q: 7. Which of the following species are likely to be nucleophiles and which electrophiles? ( explain…
A: Step 1: Step 2: Step 3: Step 4:
Q: 40.0 mL sample of a 0.431 M aqueous nitrous acid solution is titrated with a 0.282 M aqueous barium…
A: Step 1: Step 2: Step 3: Step 4:
Q: Give proper explanation of the answer please.
A: Approach to solving the question: Detailed explanation: Examples: Key references:
Q: 26. MATHEMATICAL Determine the values of Ky and Vmax for the de- carboxylation of a ẞ-keto acid…
A: The objective of the question is to determine the values of Ky and Vmax for the decarboxylation of a…
Q: The following data were recorded from an experiment to measure the activity of a peptidase enzyme. A…
A: To determine the maximum velocity (Vmax) and Michaelis constant (KM) for an enzyme-catalyzed…
Q: need help with the major product
A: Step 1: Step 2: Step 3: Step 4:
Q: I need your solution...
A: The objective of the question is to calculate the equilibrium molarity of aqueous Al3+ ion when…
Q: PROBLEM How might you carry out the following transformation? More than one 20-12 step is needed.…
A: The objective of the question is to understand the chemical transformation process of converting a…
Q: The abundance of ozone is typically inferred from measurements of UV absorption and is often…
A:
Q: None
A:
Q: None
A: Step 1:
Q: Show reaekson and don't use hend raiting and step by step solutions please
A: Step 1:Step 2:The reaction simply follows the SN2 mechanism which is a most favoured solvent that is…
Q: Sally obtains a standard calibration curve for their assigned food dye by plotting absorbance versus…
A: The objective of the question is to determine the concentration of the food dye in the original…
Q: Please make sure answer is correct a lot of controversial answer on this I will trust you and take…
A: The sequence of reactions is: Double elimination from vicinal dihaloalkanes ⟶ alkyne ⟶cis-alkene ⟶…
Q: Draw the structural formula of the product of the reaction shown below. • • Na OCH2CH3 + CH3CH2OH…
A: Step 1: Step 2: Step 3: Step 4:
Q: What is the name of this structure?
A: Step 1: Step 2: Step 3: Step 4:
Q: 5 attempts left Check my work Click the "draw structure" button to launch the drawing utility. What…
A: Benedict's reagent is commonly used to test for the presence of reducing sugars, which include both…
Q: Problem 19.36e-f Draw the starting materials needed to synthesize each of the following compounds…
A:
Unlock instant AI solutions
Tap the button
to generate a solution
Click the button to generate
a solution
- Draw structural formulas for the alkene that gives each alcohol upon hydroboration-oxidation. (a) (b)From the given two compounds in the image, which reagents would differentiate the pair? a. Br2 in CH2Cl2 b. CrO3, H2SO4, acetone c. concentrated HCl with ZnCl2 d. aqueous FeCl3 e. aqueous NAHCO3 f. ammoniacal AgNo38. Give an example of a carbene and give a reason why CHCl3 readily forms a carbene in NaOH.
- What is the major monobromination product formed by heating each alkane with Br2?Organic Chemistry Acid catalyzed dehydration reaction of 2-methyl-1-butanol produces 2-methyl-2-butene as the major product. Also acid catalyzed dehydration reaction of 3-methyl-1-butanol give the same product as major product. Explain the reason why both of the reaction produce the same product as the major product.Draw the structure of the predominant form of CF3CH2OH (pK a = 12.4) at pH = 6.
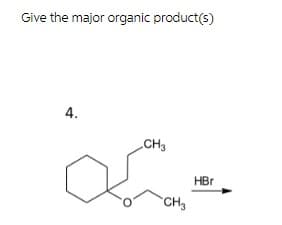
- Chemistry what is the major organic product? show sterochemistry when needed.Explain why the addition of HBr to alkenes A and C is regioselective, forming addition products B and D, respectively.Explain why the addition of HBr to alkenes A and C is regioselective,forming addition products B and D, respectively.

