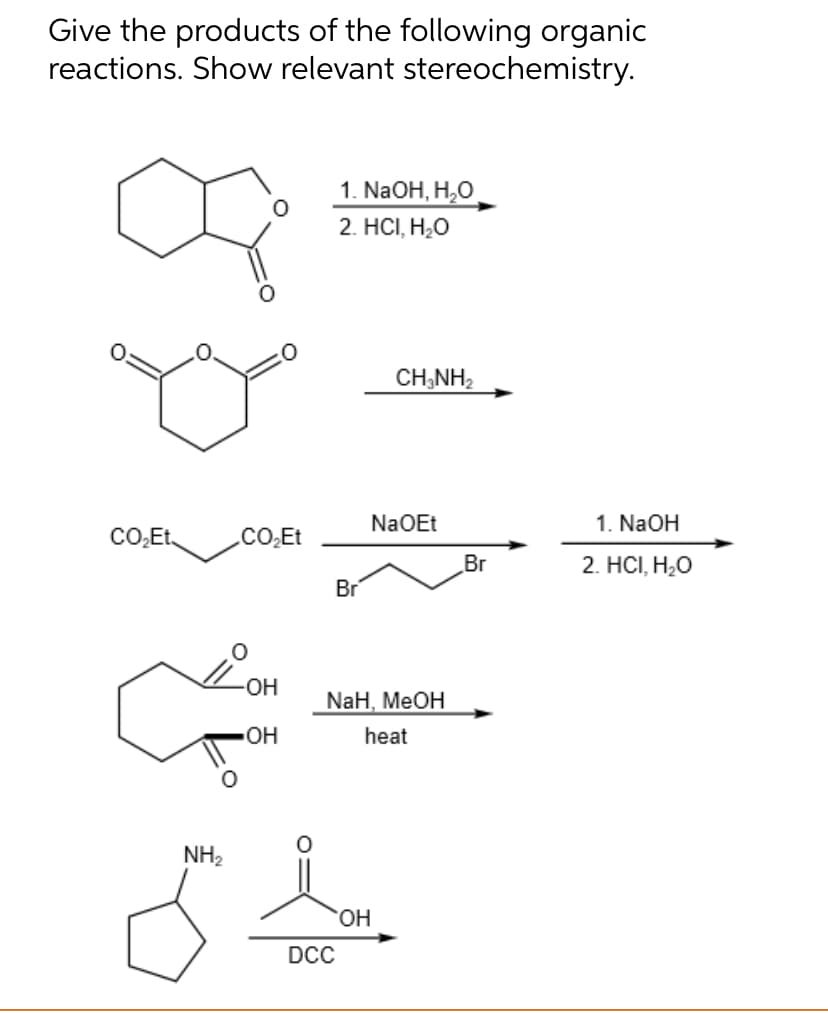
Give the products of the following organic reactions. Show relevant stereochemistry. 1. NaOH, H₂O 2. HCI, H₂O CHÍNH, CO₂Et CO₂Et Br NaOEt Br 1. NaOH 2. HCI, H₂O
Q: 9. Which one of the following groups has the Lowest rank as assigned by the Cahn-Ingold-Prelog…
A:
Q: Which of the two chair conformations is lower in energy? CH3 H3C CH3 The structure on the left is…
A:
Q: Choose the following set of elements that correctly shows a DECREASING trend for atomic radius.
A: As we know atomic radius along the period from left to right in the periodic table decreases because…
Q: Identify the conjugate acid-base pair in the reaction H2O + Cl- ↔HCl + OH-
A: Compound loss H+ ion form conjugate base, and gain H+ ion form conjugate acid. Here HCl is a…
Q: Write the balance equation for the ionization of HNO3 and H3BO3. Identify the conjugate-base pair in…
A: Bronsted-Lowry acid-base theory: The Bronsted-Lowry acid-base theory states that the acid is a…
Q: Chemistry What is the molarity of a solution formed when 37.79 g of copper II nitrate is dissolvedin…
A: Given that - Mass of Copper II nitrate = 37.79 g Volume of water used = 170 mL Then, from…
Q: Consider the reaction of 2-bromo-2-methylpropane with water, shown below, to answer the following…
A:
Q: Assign R, S configurations to each indicated chirality center in the molecules below. OH B. HO COOH…
A:
Q: Write the mechanism of the reaction of trans-2-butene with hydrogen bromide.
A: Given, Trans-2-butene + hydrogen bromide ------>
Q: The solution has [H+] = 2.2x10^-7M. The solution is A. acidic B. basic
A: pH of solution is defined as negative logarithm of hydrogen ions present in the solution
Q: A. When one of the enantiomers of 2-butanol is placed in a polarimeter, the observed rotation is…
A:
Q: 6. Which statement(s) is/are true? I The solubility of most ionic compounds in water increases with…
A:
Q: 2. 3. HO 8 ben ОН OH Bass Pyridine Рсс OH OH OH HG142), OH H₂ 21 H/₂/pd Zn, на, HCI
A:
Q: 4. How much energy is needed to heat a 35.5g sample of ice at -17.5°C to liquid water at 77.3°C?
A: “Since you have asked multiple question, we will solve the first question for you. If youwant any…
Q: Which of the following arranges the groups in order of decreasing priority according to the sequence…
A: Given are some groups and we will have to arrange them in decreasing priority. We will be using the…
Q: A sample containing 1.00 g CaCO3 (100.09 g/mol) was allowed to react with 50.0 mL 0.720 M HCl. If…
A: The percentage yield is ratio of practical yield to that of theoretical yield, whole multiplied by…
Q: Question 24 Which functional group is not found in the structure shown below? H₂N OH O ether alcohol…
A:
Q: Indicate the following on your diagram; A)Anode and cathode B) Flow of electrons C) Flow of ions…
A: Electrochemical cell diagram and representation.
Q: Given below are the ¹H NMR spectra of two isomeric compounds with the molecular formula C4H₂O₂.…
A:
Q: VOLUMENTRIC ANALYSIS Instructions: Answer each of the problems below by showing your solutions…
A: If a 0.2800 g sample of NaHCO3 (96.5% NaHCO3 ) is titrated with 0.9165N sulfuric acid, The…
Q: Explain if it is possible to combine different types of mass analyzer.
A: Mass Analyzer: It is the center of the mass spectrometer, which takes ionized loads and disparate…
Q: Select to Draw mCPBA Select to Draw OH PBr3 DMF H₂ Lindlar's catalyst HBr Select to Draw HCCNa THF…
A: Given reactions are : Predict all the products = ?
Q: An Erlenmeyer flask contains 41.40 mL of a hydrochloric acid. The solution is titrated and reaches…
A: Here we are required to find the concentration of HCl solution .
Q: Draw the product of the E2 reaction shown below. Include all lone pairs. Ignore byproducts. -:O-CH3…
A: Elimination reaction is a type of chemical reaction which is mainly used to convert saturated…
Q: Which of the following statements is INCORRECT? a. In the Bohr model, electrons travel in circular…
A: All the given statements are related to atomic structure and various models and experiments…
Q: 5. How much energy is needed to heat a 68.9g sample of water at 88.5°C to steam at 103.7°C? 6. How…
A:
Q: In balancing redox reactions using half-reactions method, why do we use H2O when balancing Oxygen…
A: In balancing redox reactions using half-reactions method , why do we use H2O when balancing Oxygen…
Q: Choose the appropriate starting material for the products seen in the reaction below: A + B + Br
A:
Q: 51. The adsorption of CO on mica at 90K gave the following results: Pressure 1.05 4.55 5.46 7.91…
A: To show whether the given data fit the Langmuir adsorption isotherm or not. “Since you have…
Q: Airbags used in automobiles are inflated with nitrogen gas produced from the reaction of sodium…
A:
Q: 4. A 0.1475-M solution of Ba(OH)2 was used to titrate the acetic acid (60.05 g/mol) in a dilute…
A:
Q: Question 13 What is the name of the structure shown below? win O 5-sec-butyldecane O…
A:
Q: D-Pinitol is an interesting hexahydroxy cyclohexane, whose structure is shown below. HO OH HO/4 HO…
A:
Q: Common System: 1. C4H9NHC2H5 (sec.) || 2. CH3(CH₂)2C(CH3)2COC(CH3)3 3. C₂H5CON(C2H5)2
A: 1. Thus compound is an anime , which has common name as alkylamine, and IUPAC name is alkanamine ,…
Q: Electron-donating groups increase the acidity of phenol. Select the correct response: 11 True False
A:
Q: Br A Br B 1. AICI 3, 2. Zn(Hg), HCI 3. Br2, FeBr3 Br ? D Br E Br
A: Here the starting material is benzene. Benzene undergoes Friedel-Crafts acylation reaction in…
Q: Calculate the pH of a 0.050 M aqueous solution of CH3CH2COOH, Ka = 1.80 x 10^-5
A:
Q: Which of the following is the most stable carbanion? Select one: H NH₂ H O H H :Z:I NH₂ NH₂ NH₂
A:
Q: The mass of one methane molecule is 2.7×10−23 gram. Find the mass of 90,000 molecules of methane.…
A:
Q: The half-life of radium 226 is 1602 years. If you have 500 grams of radium today how many grams…
A: This is a first order reaction where the radioactive substance disintegrates. The half life time of…
Q: . Which of the following has the correct assignment of oxidation numbers? a. H₂O₂ (H: 1+, O: 2-) b.…
A:
Q: Based on the reaction, Sn + Cu²+ → Sn²+ + Cu* (in acidic medium) 2+ 2+ Calculate the following…
A: Given cell reaction is : Sn + Cu+2 -----> Sn+2 + Cu+ At anode oxidation takes place while at…
Q: 4. . Which is NOT TRUE about the Law of Conservation of mass? O The number of atoms before and after…
A:
Q: Identify the Acid in the reaction H2O + Cl- → HCl + OH-
A: Acid has pH value less than 7 and base has pH greater than 7. If pH=7 , then species is neither…
Q: Which bond has the largest dipole moment? O C-F O C-CI O C-Br SO C-1
A: Dipole moment is the product of the charge on the atoms and the seperation between the charges…
Q: Lab Activity: Isolation of Lycopene from Tomato Paste What is the drying agent used? What is the…
A: The lycopene extract is washed with aqueous NaCl and K2CO3 to remove any impurity.
Q: A. Determine the reduction potential for A and B B. Construct a Latimer diagram for Element X C.…
A: We are provided with the data of standard reduction potentials for a hypothetical element X under…
Q: IDENTIFY DIFFERENT CHEMICAL REACTIONS AND ITS ADVANTAGES AND DISADVANTAGE
A: A Chemical Reaction is a process that occurs when two or more molecules collide with the right…
Q: Label each pair of compounds below as: a. conformational isomers b. stereoisomers C. constitutional…
A: We have to determine the relation between the given structures
Step by step
Solved in 2 steps with 1 images

- A certain hydrocarbon, C5H6, yields the two products shown below upon ozonolysis with 1. O3/CH2Cl2 2. Zn/H3O . Deduce the structure of the hydrocarbon.Deduce the structure of each compound from the information given. All unknowns in this problem have molecularformula C8H12.(a) Upon catalytic hydrogenation, unknown W gives cyclooctane. Ozonolysis of W, followed by reduction with dimethylsulfide, gives octanedioic acid, HOOC¬(CH2)6¬COOH. Draw the structure of W.(b) Upon catalytic hydrogenation, unknown X gives cyclooctane. Ozonolysis of X, followed by reduction with dimethylsulfide, gives two equivalents of butanedial, O“CH¬CH2CH2¬CH“O. Draw the structure of X.(c) Upon catalytic hydrogenation, unknown Y gives cyclooctane. Ozonolysis of Y, followed by reduction with dimethylsulfide, gives a three-carbon dialdehyde and a five-carbon dialdehyde. Draw the structure of Y.*(d) Upon catalytic hydrogenation, unknown Z gives cis-bicyclo[4.2.0]octane. Ozonolysis of Z, followed by reductionwith dimethyl sulfide, gives a cyclobutane with a three-carbon aldehyde (¬CH2¬CH2¬CHO) group on C1 and aone-carbon aldehyde (¬CHO) group on C2. Draw the…A certain hydrocarbon, C7H10, yields the two products shown below upon oxidative cleavage with KMnO4/H3O+ . Deduce the structure of the hydrocarbon.
- Give to all parts? (a) Give the complete mechanism for the reaction of trans-2-butene with Br2 in excess H2O. (b) Name the products (including stereochemistry). (c) Give the complete mechanism for the reaction of trans-2-butene with Br2 in excess H2O. (d) Name the products (including stereochemistry).Provide the structure of the major organic product of the following reaction and? explain the stereochemistry which results in this product. 2-Pentanol reacting with 1.) PBr3, pyridine 2.) NaCNUnknown X, C5H9Br, does not react with bromine or with dilute KMnO4. Upon treatment with potassium tert-butoxide,X gives only one product, Y, C5H8. Unlike X, Y decolorizes bromine and changes KMnO4 from purple to brown.Catalytic hydrogenation of Y gives methylcyclobutane. Ozonolysis–reduction of Y gives dialdehyde Z, C5H8O2. Proposeconsistent structures for X, Y, and Z. Is there any aspect of the structure of X that is still unknown?
- Account for any stereochemistry, major/minor products in the following reactions. Provide mechanistic explanations for your product(s).Modify the structure of 3-methyl-1-pentene to show the product formed when it is reacted with: Hydrogen chloride (HCl). Show relevant stereochemistry when applicable. A dilute aqueous solution of sulfuric acid (H2SO4). Show relevant stereochemistry when applicable. Diborane (B2H6) in diglyme, followed by basic hydrogen peroxide (H2O2, OH−). Show relevant stereochemistry when applicable. Bromine (Br2) in water. Show relevant stereochemistry when applicable. Peroxyacetic acid (CH3CO3H). Do not include stereochemistry.Predict the major products of the following reactions, including stereochemistry where appropriate. octan-1-ol + DMSO + oxalyl chloride
- Following are diastereomers (A) and (B) of 3-bromo-3,4-dimethylhexane. On treatment with sodium ethoxide in ethanol, each gives 3,4-dimethyl-3-hexene as the major product. One diastereomer gives the E alkene, and the other gives the Z alkene. Which diastereomer gives which alkene? Account for the stereoselectivity of each -elimination.When 3-bromo-1-methylcyclohexene undergoes solvolysis in hot ethanol, two productsare formed. Propose a mechanism that accounts for both of these productsFor each one of the following reactions, show step by step mechanism of the reactions and indicate the stereochemistry of the products and their relationship

