Q: Use the given standard enthalpies of formation to determine the heat of reaction of the following…
A: Dear student , since you have posted multiple parts questions we will allow to solve only first…
Q: Explain the difference between: a strong acid or base and a weak acid or base
A: pH scale helps us in measuring acidity or basicity of a solution. If a solution is neither acidic…
Q: ☆ Ma-N=Ma: Ma-N-Ma: :Ma=N=Ma: :Ma: #1 :Ma: #2 Ma: #3 c. Put a star or next to the number of any…
A: D) In I and III Octet rule is not followed by N so these are not best option of Lewis structure
Q: How many milliliters of 1.5% alcoholic dimethylglyoxime should be used to provide a 36.0% excess…
A:
Q: [Report answer to correct number of significant figures] Convert 25.9 lb/in to g/cm [1kg = 2.205 lb;…
A: Here we have to convert 25.9 lb/in into g/cm . This can be done by unit dimension analysis.
Q: 17. Calculate the de Broglie wavelength of a proton (in nm) whose mass is 1.673 x 10-27 kg and is…
A:
Q: 53.62 x 10-3 O 5.362 x 10-4 O 5.362 x 104 53.62 x 103
A: The scientific notation form of 53620 is
Q: 60. Column- (Molecular species) (A) SO₂ (8) PO (C) CO₂² (D) CIO CHE Column-II (Characteristics of…
A: A question based on general chemistry that is to be accomplished.
Q: Which substrate will go through E1 reaction fastest? Br Br A) B) ㅇ BY BV DJ ㅇ
A: Dear student, As you have asked multiple questions but according to guidelines we will solve first…
Q: A 4.94-g sample of gas exerts a pressure of 8.78 atm in a 883-mL container at 45°C. Determine the…
A:
Q: Wouldnt the volume go up if gas is being added. It is now less than, in the answer given. I do not…
A:
Q: Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of…
A: We need to calculate the molecular formula and empirical formula of the compound
Q: Rewrite the equation as thermochemical equations in two different ways to include the heat…
A:
Q: I H¬C-C=N ││ Η Η
A: The given structure is lewis dash structure in which bond is shown with line and lone pair of…
Q: 4. A chemist wants to determine the concentration of an acidic solution. They start by placing 25.0…
A: Given: The molarity of the NaOH solution = 0.12 M The volume of NaOH = 7.5 mL The volume of acid…
Q: a molar mass of 598 g/mol. If 33.7 mg of the compound is dissolved in enough water to make 175 mL of…
A:
Q: A balloon is filled to a volume of 1.50 L with 2.50 moles of gas at 25 °C. With pressure and…
A:
Q: What are the oxidation number of hydrogen in H₂O and H₂O₂ molecules respectively? A. + 1 B. -1 C. -1…
A: Oxygen has -2 charge.
Q: Calculate the wavelength in meters of light absorbed by an electron in an atom of hydrogen as it…
A:
Q: What is the IUPAC name of the following substance? ball & stick labels Retry Entire Group 5 more…
A:
Q: How many grams of Ag2CO3 will precipitate when excess (NH4)2CO3 solution is added to 74.0 mL of…
A:
Q: Fill in the blanks: Ethers may be prepared by involves the alkyl halides. 7 mechanism for which
A: Please see explanation.
Q: Calculate the pH at the equivalence point for the titration of 0.240 M methylamine (CH₂NH₂) with…
A:
Q: 2- If the electronic structure of polonium Po is '6s 4f 5d1° 6p [1], then the number of electrons in…
A: Electronic configuration is defined as arrangement of electrons in shell, subshell and orbitals.
Q: Name Period Date Unit 4 Review Worksheet #2 Empirical and Molecular Formulas Directions: Please…
A:
Q: Titanium-51 decays with a half life of 6.00 minutes. You have a bottle of titanium-51 that reads 452…
A:
Q: 2. Evaluate each of the following expressions. State the answer to the proper number of significant…
A: Rules for writing significant figures in addition subtraction multiplication and division; In case…
Q: ) Identify the calibration method used. b) Calculate the molar concentration of morphine in the…
A: a) The calibration method used is standard addition where different amount of standard is being…
Q: Shown here is a d-orbital. Label it accordingly.
A: The value of the azimuthal quantum number for the d-subshell is 2, hence the value of the magnetic…
Q: What mass of silver nitrate (molar mass- 169.9 g/mol) is dissolved in 40.0 mL of a 0.400 M aqueous…
A: We have to calculate mass of silver nitrate in the solution
Q: 4) Structure: the appropriate box. CHO H-OH HO-H HO -H Write the requested cyclized form of the…
A:
Q: One dietary supplement capsule weighing 4.90 was ground into a fine powder. Two portions of the…
A: given 4.90 g fine powder W= 0.200g V=50 ml Mn 5.00 Ml of 40.0 ppm absorption 0.300 and 0.650 find…
Q: For each row in the table below, decide whether the pair of elements will form a molecular con…
A: There are two types of bonding to form molecule- 1- covalent bonding 2- ionic bonding For more…
Q: 4. Use the bond energy from the Data Sheet to calculate AH for burning of Imole of ethyl alcohol,…
A: Given reaction: C2H5OH(l) + _O2(g) → _CO2(g) + _H2O(l) We have to calculate the ∆H for the burning…
Q: Normality (N) of Na2S2O3 = 0.025 ml Volume of the sample = 203 ml Volume (V) of Na2S2O3 = 0.98 ml…
A: Given: Normality of Na2S2O3 = 0.025 N Volume of sample = 203 ml Volume of Na2S2O3 = 0.98 ml For…
Q: True or False Considering that two carbon chains have equal number of carbons, but one has Fluorine…
A: We jave to tell wheather the statement is true or false.
Q: 16 14 12 10 Detector response 8 9 4 2 tm= 4min I 10 20 M A 30 Time, min 40 B 50 60 70
A: Capacity factor is rate of migration of a solute in a coloumn or relative indication of time spent…
Q: what happens if you measure the absorbance without diluting the chromium oxide that reacts with…
A: The reaction of chromium oxide and alcohol is given below.
Q: q. pV = nRT r. sin 0c = n₁ n₂ 1 s. qV= = 2/27 == my ² ‚T= ‚0c = C , V =
A: Rearrange the given formulas of the following--
Q: The illustration to the left represents a mixture of hydrogen (light blue ) and oxygen (red)…
A:
Q: What is the plate height if the length of the column is 10cm?
A: The plate height is calculated as: H=LN where, L is the length of column and N is the number of…
Q: Calculate the mass defect and the binding energy (in kilojoules per nucleons) for nickel-62. (The…
A: Here we are required to find the mass defect and binding energy for Ni62
Q: Directions: Balance the following chemical equation: 1. Al + O2 → Al2O3
A: In this question, we will balanced the given chemical reaction equation means both side same atoms…
Q: Fill in the blanks: The reaction involved is called occurs Use arrow formalisms to show the reaction…
A: Friedel-Craft alkylation reaction is the electrophilic substitution recation. This reaction is used…
Q: Determine the freezing point depression of a solution that contains 30.7 g glycerin (C3H8O3, molar…
A:
Q: The acidity of the alcohol is increased by efficient stabilization of the alkoxide ion through…
A: The acidity of the alcohol is increased by efficient stabilization of the alkoxide ion through…
Q: Identify the relationship between these compounds. он H NH2 HC H HN но H CH3 Diastereomers Identical…
A: The molecules with the same molecular formula but different structures are known as isomers. If the…
Q: Experiment 1: Effect of substrate concentration at a constant pH (pH = 7) and temperature Plot the…
A: A question based on kinetics of reaction in solution that is to be accomplished.
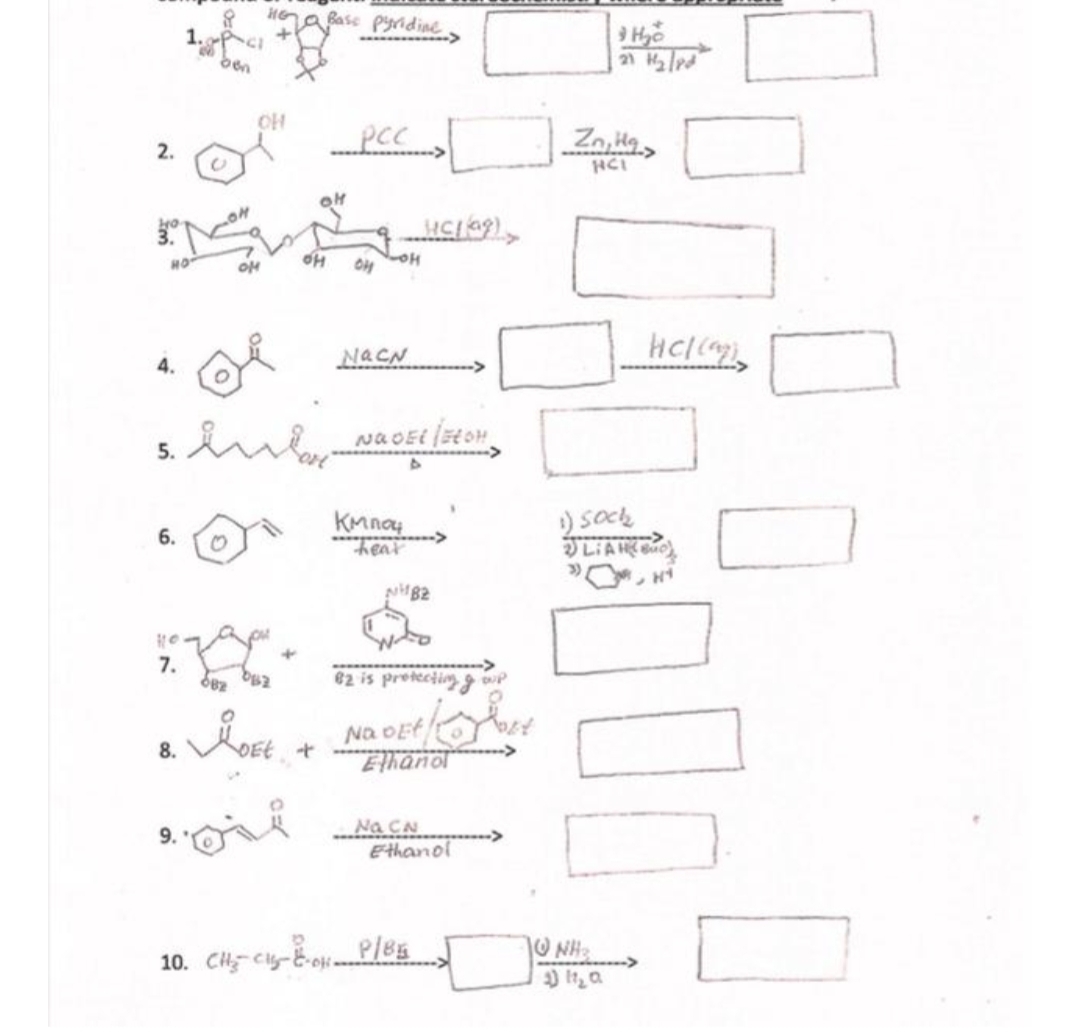
Q: Synthesize the given compound by Claisen condensation. Show all the required steps and reagents.…
A:
Q: Naturally-occurring amino acids have the L-configuration. All amino acids are chiral except for…
A: Since you have posted a question with multiple sub-parts,we will solve first three sub-parts for…
Step by step
Solved in 2 steps with 2 images

- Hexanoic acid was added to an immiscible biphasic solvent sysem, water and CCl4 at 20.0OC and the equilibrium concentrations of hexanoic acid were determined to be 3.66 g/L in H2O and 67.0 g/L in CCl4. Caluclate the distrubution coeffiecent (D1) of hexanoic acid in CCl4 with respect to water.a) Succinimide is an organic compound used in manysyntheses as a building block and is related to a class of drugs known asanticonvulsants. Given the following solubility of succinimide in water andmethanol, which would be a better solvent for recrystallizing succinimide?Solubility data: 1 gram of succinimide is soluble in 5 mL of cold water, 3 mL ofwater at room temperature and 0.7 mL of boiling water. 1 gram of succinimide issoluble in 30 mL of cold methanol, 24 mL of room temperature methanol and 5mL of hot methanol. Show calculations.b) A researcher wanted to purify 0.4 grams of crude succinimide viarecrystallization using only 2 mL of solvent. Decide the best solvent to use (water or methanol) to show the maximum % recovery of the researcher. The best solvent chosen here, may differ from the one chosen in part a. PLEASE show calculations for both parts as proof.1. Which of the following compounds can form hydrogen bonds with water?a. octaneb. HClc. NaCld. acetic acid2. Maria wants to determine the percent water content of an unknown hydrate of AB. Given the following data:mass of empty crucible - 12. 2784gmass of AB · xH20 + crucible - 17.4960gmass of AB + crucible - 15.1314gCalculate the % water content of the hydrate3. From the above data, give the correct formula for the hydrate. (AB₂ Molar mass = 129.84 g/mol)
- Aleks data for PbCO3 is 7.40 x 10^-14.Hexanoic acid was added to an immiscible biphasic solvent system, water and CCl4 at 20.0OC and the equilibrium concentrations of hexanoic acid were determined to be 3.66 g/L in H2O and 67.0 g/L in CCl4. Caluclate the distrubution coeffiecent (D2) of hexanoic acid in water with respect to CCl4.I need help on figuring out which are the four data points that can be considered outliers, below is my graph following the intructions but, not I dont know which points are outliners.
- pls help asp and provide all the explanation of how you got to the answer ( I am lost of what values to use when)Please give clear handwritten answer calculated logp of the following two drugs & show workings..?What is Ibuprofin Medication Production in a chemical Industrial Process? A) Explain what is produced, B) What chemical reaction(s) are involved. C) Why this process/product is important? D) Finally show how an understanding of limiting reagents is important in optimizing this process. E) provide at least Iwo reference URL legitimate sources Note; Please explain clearly, With a full sentence free of plagiarism.
- What is ΔHsys for a reaction at 16.9 °C with ΔSsurr = -159 J mol-1 K-1 ? Express your answer in kJ mol-1 to at least two significant figures. (Please type answer no write by hend)Gypsum (CaSO4) is one of common precipitants in brackish water desalination processes. CaSO4 ⇋ Ca2+ + SO42- Ksp = 10-4.6Assume a water containing: [Ca2+] = 10-2.1 M; [SO42-] = 10-3.2 M.(a) Find Qsp or IAP for the given water. (b) Find the solubility index (SI) and determine whether CaSO4 is under, super, or at saturation in the water.a pharmacist added 15 g of 20% w/w calamine cream to 300g of aquaphor. what is the percent stregth w/w of the final calamine product? Show work of how to get the answer. Answer should be 0.95%



