Q: Part A (2 points): Which of these species has the largest ionic radius? Part B (2 points): Explain w...
A:
Q: le answer for each question and fill in the corresponding bubble on your Scantron form. 1. A solutio...
A: Polar compound are those compounds which have polar bond which arises due to difference in electrone...
Q: e moles o e correct
A:
Q: Sulfur dioxide and oxygen react to form sulfur trioxide during one of the key steps in sulfuric acid...
A: Volume of tank = 75.0 L Initial number of moles ...
Q: Can the combination of NH3 and (NH4)2SO4 form a buffer? Use appropriate reactions to explain why or ...
A:
Q: A 25.00 mL sample of phosphoric acid (H3PO4) is titrated to the equivalence using 19.71 mL of 0.2056...
A: Answer: In acid-base reaction, neutralization takes place where hydrogen ions and hydroxide ions com...
Q: Suppose 1.53 g of barium nitrate is dissolved in 250. mL of a 42.0 m M aqueous solution of ammonium ...
A:
Q: in which of the following diagrams, I-II, is a hydrogen bond correctly shown between molecules? ö: c...
A: Intermolecular forces of attraction are the force of attraction takes place between two molecules.
Q: Predict the major product for the following reaction. Na,Cr207 H2SO4, H2O ?
A:
Q: Consider the structure of but-2-yne, if it undergoes halogenation with Cl2, which of the following f...
A: The but-2-yne undergo halogenation with Cl2 gives
Q: The boiling point of helium is 4 K. The boiling point of xenon is 170 K. Both of these are in the no...
A: London dispersion forces: The unequal distribution of electrons around the nucleus in an atom can in...
Q: Ionic compound A has both the cation and the anion in FCC. Ionic compound B has the cation in BCC an...
A: density of unit cells:
Q: How many grams of magnesium are required to produce 3.000 kg of Si? (At mass: Mg=24.31; Si=28.09) Si...
A:
Q: 1. Show that the atomic packing factor for FCC is 0.74.
A: For FCC lattice Z = 4 a = 2√2r To find the packing fraction (PF) Packing fraction is percentage o...
Q: 2. Determine the Miller indices for the planes shown in the given unit cell.
A: The Miller indices of the above plane can be calculated as follows
Q: Br O,N
A: 1. In aromatic electrophilic substitution reaction, substituent attached with benzene ring controls...
Q: Which of the following is true? a. Both alkane and alkenes can undergo radical mechanized reaction...
A: Alkane are saturated hydrocarbon whereas alkene and alkynes are unsaturated hydrocarbon.
Q: What reagent would you use to distinguish be
A: Ketones are having carbonyl funtional group , to identify this functional group 2,4-DNP test is p...
Q: An analytical chemist is titrating 139.8 mL of a 0.1600M solution of aniline (C,H,NH,) with a 0.6500...
A: Given that, 139.8 mL of 0.1600 M of aniline was titrated with 0.6500 M solution of HNO3. The pKb of...
Q: Determine the Miller indices for the planes shown in the given unit cell.
A: Determine the Miller indices for the planes shown in the given unit cell.
Q: 3/4 1/2 +y +x
A:
Q: 7. A molecular compound contains 92.3% carbon and 7.7% hydrogen by mass. If 0.592 mol of the compoun...
A: The ratio of the given mass to that of molar mass of the substance gives the no of moles of that sub...
Q: Hannah needs to make a solution of KMnO4 to be used in a chemistry experiment. On the shelf in her s...
A: Basically when we need to make solution of another lower concentration from a higher concentrations ...
Q: Draw the major product of this reaction. Ignore inorganic byproducts. Na, NH3 CHCH3ОН
A: It is an example of birch reduction.
Q: of TT electrons:
A:
Q: Determine the element which is oxidized, element which is reduced, oxidizing agent, and reducing age...
A:
Q: Knowing that atomic radius and ionization energy are two important properties of an atom that are pe...
A: Atomic radius : It is the distance measured from the centre of nucleus to the outermost electron .
Q: Describe the relationship between each of the pairs of isomers shown by recording Constitutional, Co...
A: The enantiomers are non super imposable mirror images of each other. The Diastereomers are not mirr...
Q: Relating Density to Crystal Structure 1. Show that the atomic packing factor for FCC is 0.74. 2. Sho...
A:
Q: Given a solution of 0.1 M Ag+ and 0.2M of Pb2+, (Ksp for silver oxalate = 3.5x10^-11 and ksp for lea...
A: More soluble precipitate is the one having higher solubility product value. Here lead oxalate is the...
Q: A reaction occurs according to the following balanced chemical equation: AgB (aq) + 6 CD (aq) → CB (...
A:
Q: 15 EtgSiOTf (16) CH;CN OTf EtO EtO Show the mechanism for this reaction
A:
Q: Calculate Kb for a 0.1 M solution of acetate ion, given that Ka for acetic acid is 1.8 x 10−5.
A: Given : Ka = 1.8 x 10-5
Q: The potential energy curve below is for two nuclei undergoing fusion. As the nuclei approach, the po...
A: Answer: Movement in the direction of attraction force decreases the potential energy and movement in...
Q: Liquid hexane (CH,(CH,) CH, will react with gaseous oxygen (0,) to produce gaseous carbon dioxide (C...
A: The complete combustion reaction of hexane + O2 is 2C6H14 + 19O2 --------> 12CO2 + 14H2O
Q: cell 5. Copper crystallizes in a face-centered cubic structure. What is the mass of one unit? What i...
A: Given : copper crystallizes in FCC
Q: Calculate the pH of a 7.51 M solution of HBRO. The K, of HBRO is 2.50 x 10. Express your answer usin...
A: The PH of HBrO can be calculated as follows
Q: What is the empirical formula for a compound containing 59.72% Tin 16.10% sulfur and the remainder o...
A:
Q: Define each stereocenter as either (R) or (S). If the molecule does not have a stereocenter select "...
A: Assign priorities to the groups attached to the chiral centre as per CIP rule(Cahn-Ingold-Prelog) If...
Q: 3. From the given compound below, calculate the heat of reaction at constant pressure. Use the stand...
A: From the given compound below, calculate the heat of reaction at constant pressure. Use the standard...
Q: N. DN H-N F E OF
A: When loan pair of nitrogen not involved in delocalization (resonance) then it shows more basic chara...
Q: What is the maximum concentration of Ni²⁺ that can be added to a 0.00150 M solution of Na₂CO₃ before...
A:
Q: Which of the letters (a-c) represent a chiral carbon in the molecule? CH3 a CH;CHCH2CHCH3 | b CI O b...
A: Chiral carbon ia a carbon which has all four different group attached.
Q: Calculate the pH of a 7.51 M solution of HBrO. The K, of HBRO is 2.50 x 10.
A: pH = -log[H+] Ka for an acid is the ratio of product of concentration of products raised to their ...
Q: Give the calomel electrode half cell diagram. What equailibria control the electrod potential of cal...
A: Answer: Electrode potential of a cell can be calculated by the formula shown below: Ecell=E°cell-0.0...
Q: What is the process of naming molecules?
A: We have to correct the IUPAC NAME of the following compound. Some points necessary while writing IUP...
Q: Explain the reason behind why oils leave a translucent spot on paper even after drying, while ethano...
A:
Q: What is the name of the following apparatus? O a. Crucible Tongs O b. Wire gauze Oc Erlenmeyer flask
A: Laboratories require a wide range of equipment and instruments in order to conduct testing and resea...
Q: Spectrophotometric of a Cobalt Chloride Solution Analysis 1. explain in detail the results o...
A: The wavelength at which maximum absorbance of a compound is obtained is known as lambda max. And as ...
Q: 8. Pure copper can be produced by the reaction of copper(II) sulfide with oxygen gas as follows: CuS...
A: Given, Chemical reaction : CuS(s) + O2(g) -------> Cu(s) + SO2(g) 0.640 g of Cu metal produced...
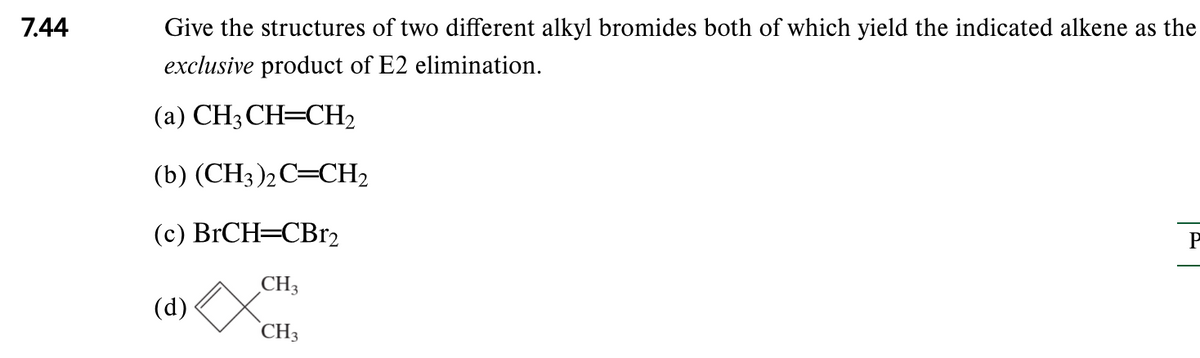
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- Friedel–Crafts alkylation of benzene with (R)-2-chlorobutane and AlCl3 affords sec-butylbenzene.a. How many stereogenic centers are present in the product?b. Would you expect the product to exhibit optical activity? Explain, with reference to the mechanism.Dehydrohalogenation of 1-chloro-1-methylcyclopropane affords two alkenes (A and B) as products. Explain why A is the major product despite the fact that it contains the less substituted double bond.Provide a reasonable arrow-pushing mechanism for Reaction 5b, and explain the the stereochemical outcome. 5d below
- Write out all the isomers of the compound with molecular formula C4H10O . Select the normal/ primaryisomer and treat it with conc. H2SO4 and heat. Identify the reaction and give the product ‘A’ from it. 2. When ‘A’ is treated with HBr in the presence of a peroxide, give the name and structure of the product.2-bromopentane, when treated with alcoholic KOH yields a mixture of three alkenes A, B and C. Identify A, B and C. Which is predominant?-Ocimene is a pleasant-smelling hydrocarbon found in the leaves of certain herbs. It has the molecular formula C10H16 and a UV absorption maximum at 232 nm. On hydrogenation with a palladium catalyst, 2,6-dimethyloctane is obtained. Ozonolysis of -ocimene, followed by treatment with zinc and acetic acid, produces the following four fragments: (a) How many double bonds does -ocimene have? (b) Is -ocimene conjugated or nonconjugated? (c) Propose a structure for -ocimene. (d) Write the reactions, showing starting material and products.
- In the following three compounds(1,2,3) arrange their relative reactivity towards the reagent CH3Cl / AlCl3. and justify it.1-Chloro-1,2-diphenylethane can undergo E2 elimination to give either cis- or trans-1,2-diphenylethylene (stilbene). Draw Newman projections of the reactive conformations leading to both possible products, and suggest a reason why the trans alkene is the major product.1. (1-bromo-1, 3-dimethylcyclopentane) an optically active pure sample reacted with water. Write the complete mechanism which includes the electron-pushing arrows. Show the expected stereochemistry for each of the case and each mechanism should give two distinct products and then describe the relationship of the products to one another.
- Consider the following chemical transformation:The transformation takes place via twosequential pericyclic reactions. Identify the two reactions and give a critical explanationwhether the reactions are allowable.When 1,2-dimethylcyclopentene undergoes hydroboration–oxidation, one diastereomerof the product predominates. Show why this addition is stereospecific, and predict thestereochemistry of the major product.1- Chloro-1,2-diphenyletane can undergo E2 elimination to give either cis- or trans-1,2-diphenylethylene (stilbene). Draw New man projections of the reactive conformations leading to both possible products, and suggest a reason why trans alkene is the major product?

