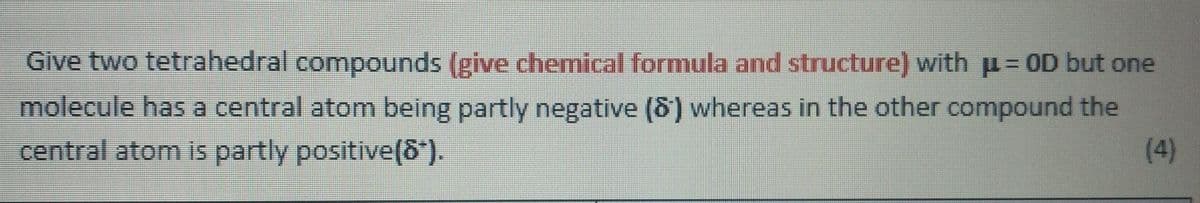
Give two tetrahedral compounds (give chemical formula and structure) with μ = OD but one molecule has a central atom being partly negative (8) whereas in the other compound the central atom is partly positive(8*). (4)
Q: isomers with the formula C510. A с Answer: + Give the correct IUPAC names for both isomers selected…
A: Molecular formula = C5H10
Q: What do you call the high-energy complex that forms from the collision of reactants, and which…
A: Reactant particles collide with each other and a high energy complex formed first of all. ->This…
Q: 1. What is the energy in Cal unit of one photon of microwave radiation with a wavelength of 550nm?…
A: Dear student , since you have posted multiple questions we will allow to solve only first question…
Q: Consider the coordination compound [FeCl₂(NH3)4][FeBr4]. Which of the following is TRUE regarding…
A: Suitably substituted square planar and octahedral complexes can show the cis-trans isomerism.…
Q: Consider the reaction: NH3(g) + Cl2(g) → NH₂Cl(g) + HCl(g) Compute AH*rxn for the reaction of 100…
A: ΔH0rxn = Σ ΔHbonds broken - Σ ΔHbonds formed
Q: Predict the sign, + − or 0, of ASº for each of the following reactions a) Oxygen gas and nitrogen…
A:
Q: there is an enzyme is catalyzed reaction that is at equilibrium and it produces 0.1 M of substrate…
A:
Q: Calculate the total number of oxygen atoms in 65.0 g of KClO3. Express your answer in scientific…
A:
Q: 1s²2s²2p63s²3p64s²3d104p65s²4d4
A: Here we have to predict the symbol of divalent cation of the following given electron configuration.
Q: 6. 45.2 g of Iron metal reacts with excess oxygen gas to produce 58.1 g of Fe3O4. a) What is the…
A: We need to calculate Theoretical yield Percentage yield Actual yield
Q: Question 24 Which of the following is the electron configuration of Cu O [Ar]4s²3d8 O [Ar]4s²3d64p²…
A: This question belong to d-block of element. Electronic configuration of d- block element. generally…
Q: What is the IUPAC name for each of the following compounds? a.C6H5CH2(CH2)3COOH b. Br(CH2)3COOH D-CH…
A: In this question, we will give the IUPAC name of the given three compounds. You can see the…
Q: Protein synthesis begins when a ribosome assembles at an initiation codon (AUG) in the mRNA. The…
A: a. Both of the statements are TRUE Protiens are synthesised (made) by the ribosomes using messenger…
Q: What type of isomerism is exhibited by the following pair? NH₂ NH₂- -NH₂ H₂N- "NH₂ NH₂ O optical…
A: The given pair exhibits optical isomerism as they form non-superimposable mirror images and no plane…
Q: What isotopic element results when 157Gd emits emits a beta particle? 64 Periodic table element…
A: Nuclear reaction: The given reaction is a nuclear reaction. In the nuclear reaction, one nuclide is…
Q: Calculate the nuclear binding energy of U-238 in J/mole
A: The correct answer is given below
Q: 2.37 L solution containing 1.30 mol L-1 potassium hydroxide (KOH) has been left on your bench. How…
A: Given-> Volume of KOH = 2.37L Molarity of KOH = 1.30 mole/L
Q: Draw the structure of 2-phenylpropan-1-ol. Draw the molecule on the canvas by choosing buttons from…
A:
Q: What is the systematic name of Fe3[Fe(C2O4)3]2? (Do not use capital letters aside from the Roman…
A: This question belongs to co-ordination compounds and the naming is based on the name of…
Q: For the compound with the chemical formula MnS2, what is the charge from one Mn cation? No charge,…
A: Let the oxidation number of Mn= x Now , We know the oxidation number of sulfide ion = -2
Q: Consider the coordination compound [FeCl₂(NH3)4][FeBr4]. Which of the following is a coordination…
A: we have to select the coordination isomer of the given complex
Q: Calculate the solubility of MgF2 (MW = 62.3 g/mol) in water at 25 °C if its Ksp is 7.4 x 10-11.? No…
A:
Q: Consider a voltaic cell with the cell notation shown below. Which of the following statements is…
A: Here we have to predict the incorrect statement for the following given Galvanic cell reaction.
Q: Which of the following molecules, when viewed in three dimensions, is nonpolar, with zero net…
A:
Q: As your undergraduate thesis, you developed a colorimetric method for determining a certain metal…
A: A question based on absorption that is to be accomplished.
Q: the acidity function when adding 5 ml of NaOH Calculate the pH value when titrated between 25 ml of…
A:
Q: What is the reactant in this reaction? 1. PPh3 2. BuLi ? 3. a. (CH3)2CHCH₂Br b. CH3CHBr2 C. CH3CH₂Br…
A: ->When phosphorus ylide reacts with carbonyl compound there is formation of Alkene.
Q: Chemistry A 25g sample of Strontium Acetate reacts wuth Sodium Chloride in a double replacement…
A: In the balanced chemical equation, the no of each atom should be equal on both the side of the…
Q: The isotope 166-Yb decays by electron capture with a half-life of 57 hrs. What percentage of the…
A: We have to calculate the percentage of isotope left
Q: How long would it take to deposit 50.0 mg Cu at the cathode of an electrolysis cell containing 6.0 M…
A: Weight of Cu = 50.0 mg = 50.0 × 10-3 gm = 0.050 gm (1gm = 1000 mg) Current (i) = 150 mA = 0.150 A…
Q: A brewed tea brand sample was subjected to solvent extraction to extract caffeine. The cup of tea…
A:
Q: 45. Assume that one unit of life is composed of the following chemical formula: C,H,O,P,N. If an…
A: Given-> C4H8O4P2N is chemical formula for 1 unit life. Carbon atom = 24 Hydrogen atom = 80…
Q: The nucleus of U-238 has a mass of 238.0003 amu. The mass of a proton is 1.0073 amu. The mass of a…
A:
Q: Consider the Ellingham diagram shown below. Choose the INCORRECT statement among the following…
A: Ellingham diagram is the graph plotted between ∆G° and Temperature for the formation of oxide for…
Q: Consider the combustion of 1.0 mol propane gas (C3H₂) in excess oxygen in open atmosphere at 25 °C.…
A:
Q: Consider the Lewis structure for the polyatomic oxyanion shown here, where X is an element from the…
A: Solution - According to the question - Given - (a) The central atom X is = PO43- The element…
Q: Name the three particles of the atom and their respective charges
A: Detail description is given below
Q: AH° = -1670 kJ/mol 2. For this unbalanced chemical reaction: X(s) + 3/2 Y2(g) → X₂Y3 (Sh 1.…
A: Recall the given reaction, X s + 3/2 Y2 g → X2Y3 s…
Q: Hi, could you please help calculate the rest of the table and the question. thank you.
A: The question is based on titration between Ca ion and HCl. The calculations based on your…
Q: Arrange the atoms in order of increasing atomic size. W, Ag, Fr, Te Arrange the atoms in order of…
A: (1) Given elements are - W , Ag, Fr , Te The atomic radius of different elements are - W -…
Q: 9. Which of the following statements are correct for ceramic materials? i. They are inorganic…
A: 9.Ceramic materials are inorganic materials consisting of a metal/semimetal and a non metal.…
Q: Please describe one regulatory mechanism involved in amino acid synthesis, with an example
A:
Q: HOW
A: EON is an extremely long period of time.
Q: Draw the Lewis Dot Structures of the following ionic compound. I 1. KBr 3. A1,0, 2. CaF₂ 4. Na,O the
A: Given-> ->KBr , CaF2 , Al2O3 , Na2O ->Ionic compounds are in form of ions .
Q: What is the pH of a buffer solution that contains 0.2M HC2H302 and 0.35 MNa C2H3O2, if pKa= 4.74 ?…
A:
Q: The rate of a certain reaction is given by the following rate law: rate k[N₂][H₂] Use this…
A:
Q: Which of the following is NOT a Representative Element O Ni Sn Rb Sb O Pb O
A:
Q: 100 90 80 70 60 50 40 30 20 10 0 4000 3000 2000 1500 wave number, cm-1 WY 1000 500
A: The given spectra is for IR spectroscopy. IR spectroscopy useful for identifying functional groups.…
Q: For the following redox equation, complete the oxidation and reduction half equations and identify…
A: We need to fill the empty boxes correctly
Q: Q4- Three moles of monoatomic idea gas in state A is expanded to state B through the following…
A: According to bartleby guidelines we're supposed to answer only first question among these So please…
Step by step
Solved in 2 steps with 2 images

- Identify the type of hykidization isip, sp², sp³) for all N (NITROGEN) atoms of the following compound.Select one: True or False 1-The mesomeric form of succimer is the active chelating form that is used to chelate trivalent metals through its COOH groups. 2-Anions with large radius particularly those having less than two negative charge are more easily polarized by the small size cations while those of ≥1 charge like sulfide S-2 and selenium Se-2 are much harder to be polarized 3-Covalent bond is encountered in case of NaCl and KBr while ionic bond is encountered with BeCl2 and BF3 4- Besides providing the largest suitable number of valance orbitals for covalent bond formation it can determine the final shape of the molecule for example Sp hybridization leads to a diagonal planner molecule, SP2 hybridization while Sp3 hybridization leads a pyramidal (tetrahydral shape) 5-About the underlying compound ( in the images ) , it it’s a calcium complex of EDTA used to treat metal toxicities like pulmbism in order to avoid hypocalcaemia.Given: 2 H2 + O2 yields 2 H2O, Delta H = -483.6 kJ/mol What is the enthalpy for the following reaction? H2O yields H2 + 1/2 O2
- The dissociation of H3 AsO4 to H3 O+ and AsO43-Iodine azide, IN3 , adds to alkenes by an electrophilic mechanism similar to that of bromine. If a monosubstituted alkene such as 1-butene is used, only one product results: REFER IMAGE (a) Add lone-pair electrons to the structure shown for IN3 , and draw a second resonance form for the molecule.(b) Calculate formal charges for the atoms in both resonance structures you drew for IN3 in part (a).(c) In light of the result observed when ΙÎ�3 adds to 1-butene, what is the polarity of the I—N3 bond? Propose a mechanism for the reaction using curved arrows to show the electron flow in each stepIn which of these two hydrides:NH3or SbH3,would you expect a greater distortion from the normal tetrahedral bond angle of 109°? And give reason for your answer
- Rank the given compounds in increasing order based on the property. (1 = lowest)The [a] of pure quinine, an antimalarial drug, is -165.(a) Compare the bond enthalpies (Table 8.3) of the carbon–carbon single, double, and triple bonds to deduce an averageπ -bond contribution to the enthalpy. What fraction ofa single bond does this quantity represent? (b) Make a similarcomparison of nitrogen–nitrogen bonds. What do youobserve? (c) Write Lewis structures of N2H4, N2H2, and N2,and determine the hybridization around nitrogen in eachcase. (d) Propose a reason for the large difference in yourobservations of parts (a) and (b).
- Compound A, shown below, is a developmental drug used for the treatment of gastrointestinal disorders. a) List all atoms present in compound A and rank them in order of increasing electronegativity (including the electronegativity values to 2 dp). c) By drawing on the diagram of compound A below, draw the expected polarity of each non C-H and C-C bond found in compound A.C and S are always tetravalent but Ge, Sn And Pb show divalency. Why?Suppose a chloride ion and a sodium ion are separated by a center–centerdistance of 5 Å. Is the interaction energy (the energy required to pull theminfinitely far apart) predicted to be larger if the medium between them iswater, or if it is n-pentane? (as shown)If Ca2+, Na+, and F- each have ionic radii ~1.16. Which ionic bond is stronger: Ca-F or Na-F?If Ca2+ is often bound on the surface of a protein by carboxylic acid functional groups. If the pKa of a particular -COOH group is 4.2, wouldyou predict Ca2+ to be most tightly bound at pH 8, pH 4.2, or pH 3? Explain your answer.

