Q: Based on your khowiedge liquid CCI4 has a higher surface tension than
A: it is true or false that Intermolecular forces a container of liquid CCl4 Has Higher surface tension…
Q: A liquid property responsible for its resistance to flow is called what? a. viscosity b. density…
A:
Q: Does solid state stearic acid has a viscosity at room temperature
A: Viscosity is a measure of a fluids resistance to flow. It is a frictional forces resulting due to…
Q: Please do surface tension, polar molecules, shape structure and transparent
A: Surface tension,polar molecule,shape and transparent property explain below.
Q: Surface tension measures which of the following? a. a liquid's resistance to flow b.…
A: Surface tension is the force acting per unit length. It depends on the intermolecular forces of…
Q: L flask containing in the flask after the stopcock is
A: In flask 1. V1 = 4.2 L P1 = 2.2 atm V2 = 8.4 L P2 = 1.9 atm At the both flasks are at same…
Q: Identify which of the following molecules can exhibit hydrogen bonding as a pure liquid. Check all…
A: Note: According to our guidelines we are supposed to answer only one question. Kindly repost other…
Q: If instead of water it rained a non polar substance, how might the drop size and shape differ? would…
A:
Q: In getting/testing the surface tension of a liquid, what is the importance of collecting more than…
A: Interpretation - What is importance of collecting more than one drop of the liquid sample from the…
Q: Discuss briefly factors that influencing plastic properties.
A: Plastics are semisynthetic materials with polymer as an active ingredient. Thus the properties of…
Q: Critical point Melting. Liquid Freezing Vaporization Solid Condensation SublimationTriple point Gas…
A: A phase diagram represents the conditions such as temperature, pressure and volume at which…
Q: A liquid property responsible for its resistance to flow is called what?
A:
Q: Less is the surface area of a liquid, high will be its rate of evaporation. Select one: True False
A: Evaporation is a surface phenomenon. A surface of a liquid is the interface between the liquid and…
Q: The liquid was nonpolar gasoline, would it be able to support a paper clip ?
A: The given concept is explained by concept of surface tension. Surface tension is responsible for the…
Q: Define oil viscosity.
A: Viscosity is a physical property of liquid that depends on various factors like intermolecular force…
Q: The table below lists factors that may affect the rate at which solids and gases dissolve. Fill in…
A:
Q: Why does diffusion in solids important?
A: The correct answer about diffusion in solids is given below
Q: draw an atomic/molecular level representation of what you think the solvent (H2O) looks like before…
A: Water is a polar molecule, which is liquid at standard temperature and pressure. Water molecules can…
Q: 1. Which of these models show the process of boiling (fast evaporation)? Circle the correct letter.
A: The left diagram A. showing the fast evaporation than the another. Evaporation is the process in…
Q: explain what advantage the technique of reflux has over conventional boiling
A: Refluxing is a technique that is used to boil a mixture over a long period of time. In this…
Q: TRUE OR FALSE Melting Point should be taken on dry solids only.
A: Yes it is true melting point should be taken of dry solid only . because if it contains moisture…
Q: Identify which of the following molecules can exhibit hydrogen bonding as a pure liquid. Check all…
A: Hydrogen bonding is a type of dipole-dipole interaction of molecules when the hydrogen is bonded to…
Q: Which substamf below is expected to give the highest surface tension in liquid phase CH2F2…
A: We have find out the Which substance have expected to give the highest surface tension in liquid…
Q: An unknown substance was subjected to boiling point determination. Temperature is recorded at 75℃…
A: In the method of finding boiling point of substance using capillary tube, when the bubbles escape…
Q: Describe the process inside the test tube during boiling point determination
A: Boiling Point is the temperature at which the vapour pressure of the liquid is equal to the…
Q: Can you interpret this graph. Normally on a temperature-composition phase diagram, there is a bubble…
A: Liquids that are miscible to each other to a limited extent are termed partially miscible liquids…
Q: Match the slope of the solid-to-liquid boundary with the information that can be drawn. negative…
A: The phase diagram explains the different phases of any given substance wrt. its pressure and…
Q: Komal found that her vial of isopropyl alcohol showed a much better surface tension bubble shape (a…
A: Answer Options A, B and C are the three experimental errors that could have most likely occurred.…
Q: normal boiling point (in
A:
Q: Less is the surface area of a liquid, high will be its rate of evaporation. Select one: True False
A: The surface area of a liquid is the portion of liquid that has been revealed to or touches the air.
Q: Properties of Liquids and IMF example andbrief discussion
A: Liquids can be defined as fluids which do not have fixed shape but volume. For example: Water, Oil,…
Q: How does the ramp rate affect the actual AND observed melting point?
A: Thermal rate effect the actual melting point of subtance, higher the ramp rate higher the observed…
Q: Will solid sodium chloride be harder (that is, more resistant to indentation) or softer than solid…
A: Is sodium chloride be harder or softer than solid carbon tetrachloride has to be explain,
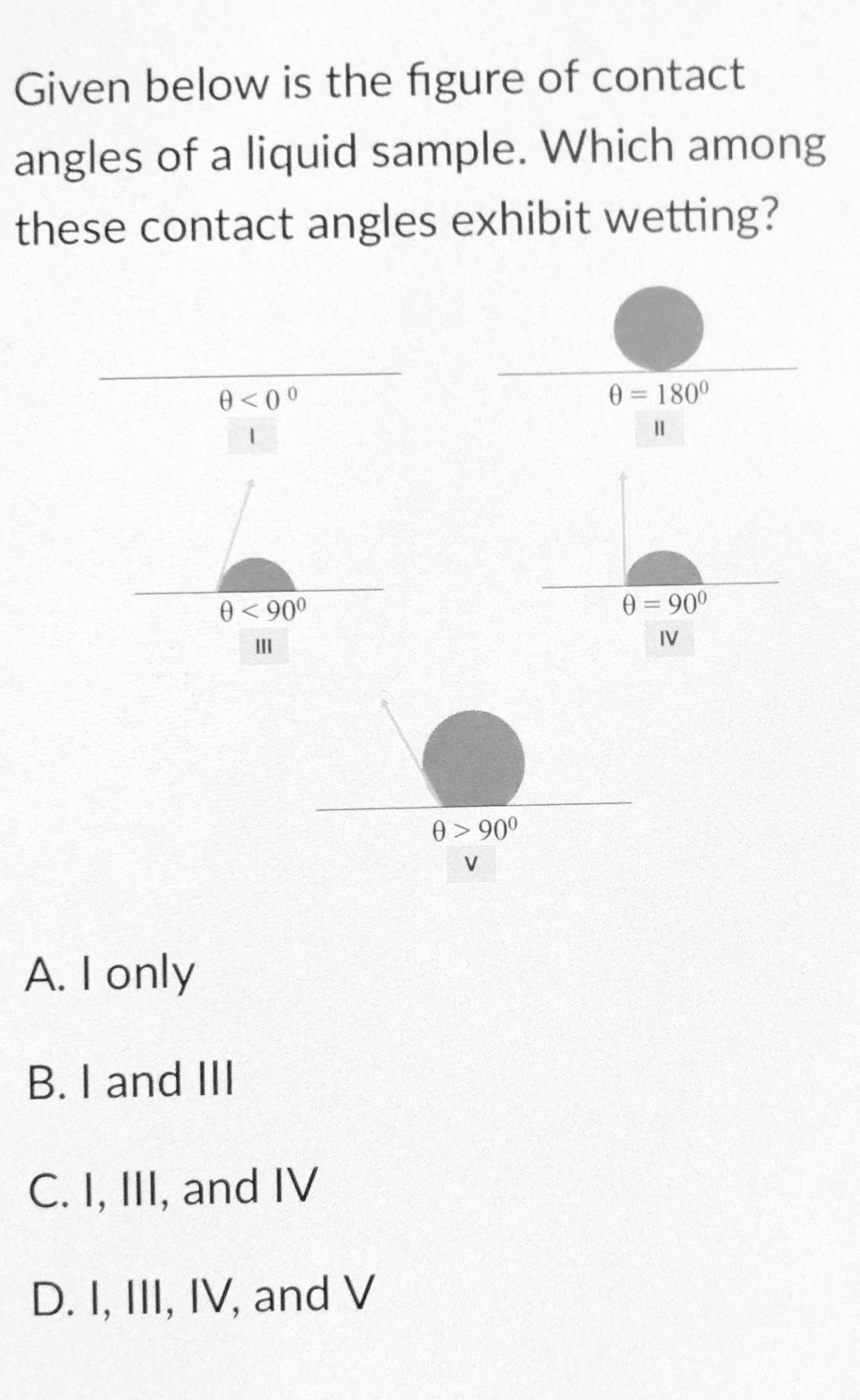
Q: Can you please paraphrase the text below for me? with your own words please Wettability is the…
A:
Q: Are there any alternatives for a rotary evaporator?
A: Rotary evaporator is an equipment used in chemical laboratories to separate the solvents and the…
Q: Which liquid is expected to have the higher surface tensio
A: It is a property of liquid that allows it to resist an external force , it is due to cohesive…
Q: What is the formability?
A: In this question we have to explain and also determine which component of MSE was considered in…
Q: 5.Measures to observe when measuring viscous liquids include the following, with exception of a.Use…
A: To answer the given questions
Q: Why Clausius–Clapeyron equation imporant?
A: The Clausius-Clapeyron equation refers to the relationship between temperature and pressure in…
Q: More is the surface area of a liquid, high will be its rate of evaporation. Select one: True False
A: The phenomenon of change of a liquid into vapours at any temperature below its boiling point is…
Q: 11. This is the term used to describe resistance to flow of a liquid. * surface tension O vapor…
A: Surface tension - it is a tendency of fluid surface to shrink into minimum surface area possible.…
Q: Two samples have exacted the 1 point same melting points, so the samples are: If the mixture has the…
A: The temperature at which the state of the substance turns from solid to liquid is known as the…
Q: Select the statements that correctly describe the term viscosity. This quantity is low for…
A: All matter can be classified as solid, liquid, and gases mainly. In these physical states of matter,…
Q: As part of an investigation into the strength of electrostatic forces between molecules of water, a…
A: Since measuring the melting point to measure the electrostatic force is very sensitive case. (In…
Q: The temperature is 78°F and the dewpoint is 59°F. Find the ratio of vapor pressure to the saturation…
A:
Q: if we keep wet clothes in a plastic bag, they do not dry, while when we put them on the wire, the…
A: When wet clothes dry, the water from the clothes gets evaporated and the water vapour formed from it…
Q: Which physical property of liquid is measured using stalagmometer?
A: Stalagmometer is based on the principle of drop method . The weight of the drop falling from the…
Q: Describe why adding a solvent would either lower the vaper pressure of raise the boiling point.
A: For the solvent, the presence of the solute decreases its vapor pressure by dilution.This means in…
Step by step
Solved in 3 steps with 3 images

- dipole dipole interaction between aspirin and silica explanation pls and steps alsoAn ostwald viscometer was used to determine the time of flow of syrup at 20°C Data of water: Time of flow: 0.23 sec Density at 20°C: 0.998 g/cm3 Viscosity: 0.0101 poise Data of Syrup: Time of flow: 1.42 sec Density at 20°C: 1.153 g/cm3 Question: 1. What is the kinematic Viscosity of water? 2. What is the Viscosity of syrup? 3. From the computed Viscosity of syrup, what will be its fluidity? Show your complete solution!When 10 mL of water at 20oC is placed in an Ostwald viscometer, it takes 136.5 s for the liquid levelto drop from the first mark to the second. For 10 mL of hexane at 20oC in the same viscometer, thecorresponding time is 67.3 s. Find the viscosity of hexane at 20oC and 1 atm.(ηH2O=1.002 cP, dH2O= 0.998 g/cm3, dC6H14=0.659 g/cm3)
- An ostwald viscometer was used to determine the time of flow of syrup at 20°C Data of water: Time of flow: 0.23 sec Density at 20°C: 0.998 g/cm3 Viscosity: 0.0101 poise Data of Syrup: Time of flow: 1.42 sec Density at 20°C: 1.153 g/cm3 1. What is the fluidity of water? 2. What is the Viscosity of syrup? 3. From the computed Viscosity of syrup, what will be its kinematic Viscosity? Show complete solution.PLEASE ANSWER THEM ALL IN ASAP FOR AN UPVOTE. I PROMISE THAT. THANKYOU. An Ostwald viscometer was used to determine the time of flow of honey at 20 °C. Data for Water:Time of flow: 0.23 secondsDensity at 20 °C: 0.998 g/cm³Viscosity: 0.0101 poise Data for Honey:Time of flow: 20.79 secondsDensity at 20 °C: 1.426 g/cm³ a.) What is the viscosity of honey? b.)From the computed viscosity of honey, what will be its kinematic viscosity? c.) From the computed viscosity of honey, what will be its fluidity?Which organic liquid has: Molecular weight = 79.53g/mol Density = 0.7708 g/ml Viscosity = 1.39 mPa.s index of refraction = 1.420
- An Ostwald viscometer was used to determine the time of flow of syrup at 20 °C. Data for Water:Time of flow: 0.23 secondsDensity at 20 °C: 0.998 g/cm³Viscosity: 0.0101 poiseData for Syrup:Time of flow: 1.42 secondsDensity at 20 °C: 1.153 g/cm³ 1. What is the fluidity of water? 2. From the computed viscosity of syrup, what will be its kinematic viscosity? 3. From the computed viscosity of syrup, what will be its fluidity? 4. What is the viscosity of syrup? 5. What is the kinematic viscosity of water?When the temperature is 20 ° C, water flows through an ostwald viscometer at 363 s, whose density and viscosity are given as 0.9982 g / cm3 and 1,000 cP, respectively. The flow time of OF2 from the same viscometer at -145.8 ° C was measured as 68 s. Since the density of OF2 at this temperature is 1.523 g / cm3, what is the value of its viscosity?The viscosity of Ethyl acetate determined at 25°C was 4.2cp. The efflux time for water to flow from marks A to B was 3 minutes.Additional data: Viscosity of water at 25°C = 1.002cp Density of water at 25°C = 0.997g/mL Calculate for the time for a 10.0mL Ethyl acetate (equivalent to 5.0g) to flow from Marks A to B.
- Give detailed Solution asapAscetic acid with a specific gravity of 3.25 has a kinematic viscosity equal to1.35 × 10-6 m2/s , what is its dynamic viscosity?Kindly watch the experiment around 15:38 LINK: https://www.youtube.com/watch?v=oTT46EzBJ2Y Procedure: Using a pipette, allow 10ml to flow, taking the time for the pipette to empty. Repeat using 10ml each of hot water, 10% sugar, alcohol, and normal saline solution. What is the effect of heat on viscosity? The effect of non-electrolytes and highly ionized salts on viscosity?



