Given the element pairs and electron configuration explain how the electrons are transferred and also identify the correct ratio in which they will form the ionic bond Element Electron Element Electron Explanation of electrons transfer Cation:Anion Ionic Formula configuration configuration ratio Na 1s? 2s? 2p° 3s 1s? 2s? 2p* Each Na atom will lose one 2:1 Na,0 electron and each O atom EXAMPLE needs two electrons Mg 1s 2s? 2p° 3s? F 1s 2s 2p 1s 2s 2p° 3s? 3p 4s1 K CI 1s 2s? 2p° 3s? 3p Li 1s 2s' 1s 2s? 2p° 3s? 3p" 1s 2s 2p° 3s? 3p 4s? Ca N. 1s 2s? 2p3 1s 2s 2p° 3s? 3p Al 1s 2s? 2p* 1s 2s 2p° 3s? 3p° 4s 3d10 4ps Be 1s 2s? Br
Given the element pairs and electron configuration explain how the electrons are transferred and also identify the correct ratio in which they will form the ionic bond Element Electron Element Electron Explanation of electrons transfer Cation:Anion Ionic Formula configuration configuration ratio Na 1s? 2s? 2p° 3s 1s? 2s? 2p* Each Na atom will lose one 2:1 Na,0 electron and each O atom EXAMPLE needs two electrons Mg 1s 2s? 2p° 3s? F 1s 2s 2p 1s 2s 2p° 3s? 3p 4s1 K CI 1s 2s? 2p° 3s? 3p Li 1s 2s' 1s 2s? 2p° 3s? 3p" 1s 2s 2p° 3s? 3p 4s? Ca N. 1s 2s? 2p3 1s 2s 2p° 3s? 3p Al 1s 2s? 2p* 1s 2s 2p° 3s? 3p° 4s 3d10 4ps Be 1s 2s? Br
Chapter3: Mechanisms
Section: Chapter Questions
Problem 105EQ
Related questions
Question
I need help answering all this please
(Not honor class)
(Not grading)
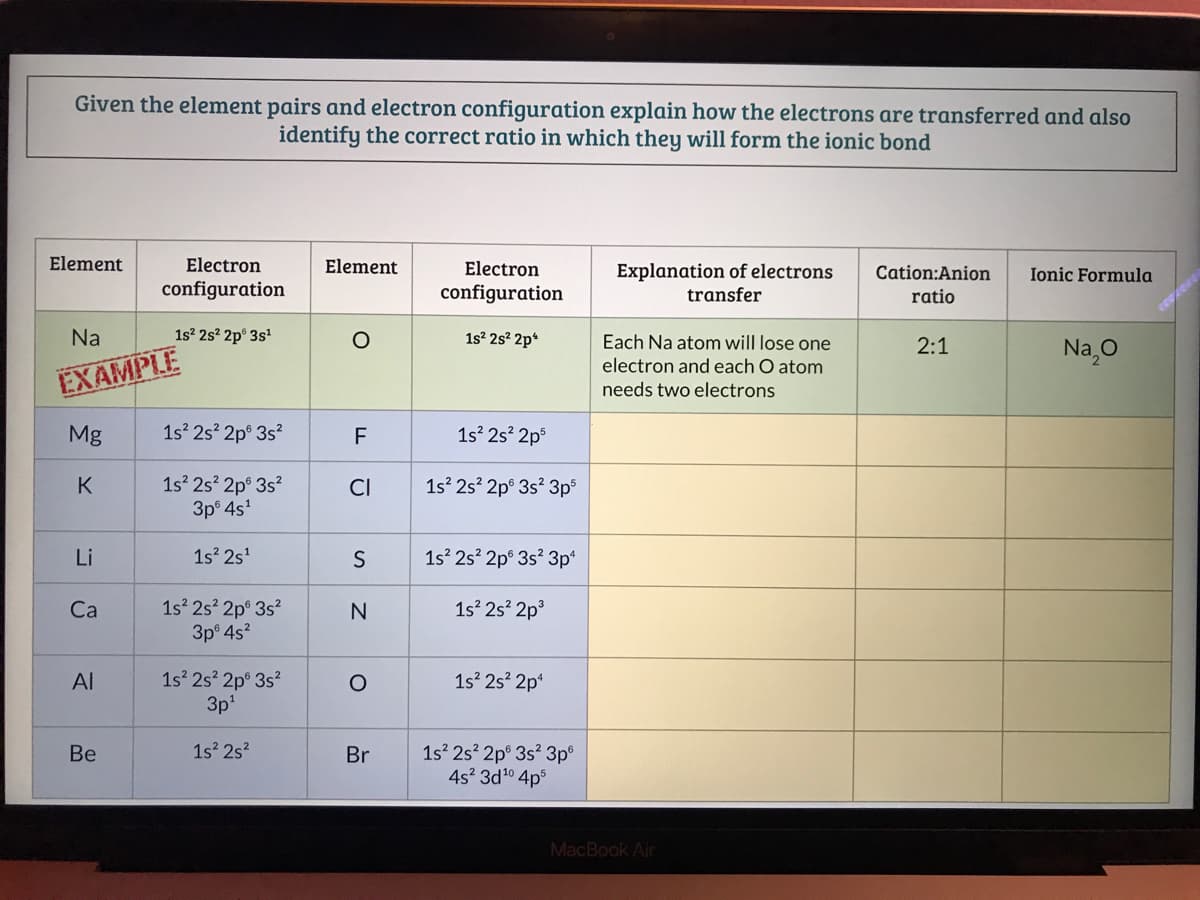
Transcribed Image Text:Given the element pairs and electron configuration explain how the electrons are transferred and also
identify the correct ratio in which they will form the ionic bond
Element
Electron
Element
Electron
Explanation of electrons
transfer
Cation:Anion
Ionic Formula
configuration
configuration
ratio
Na
1s? 2s? 2p° 3s
1s2 2s? 2p*
Each Na atom will lose one
2:1
Na,0
electron and each O atom
EXAMPLE
needs two electrons
Mg
1s 2s? 2p° 3s?
F
1s 2s 2p°
1s 2s 2p° 3s?
3p 4s'
K
CI
1s 2s 2p° 3s? 3p
Li
1s 2s'
1s 2s? 2p° 3s? 3p*
1s 2s? 2p° 3s?
3p 4s?
1s 2s? 2p
Ca
1s 2s 2p° 3s?
3p'
Al
1s 2s? 2p*
1s 2s?
1s 2s? 2p° 3s? 3p°
4s? 3d10 4ps
Be
Br
MacBook Air
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry for Engineering Students
Chemistry
ISBN:
9781337398909
Author:
Lawrence S. Brown, Tom Holme
Publisher:
Cengage Learning