Given the following balanced chemical equation: 2 NO(g) + O2(g) 2NO2(g) > 15.0 g of NO(g) and 15.0 g of O2(g) placed in this reaction a) Determine the limiting and excess reactant. Calculate the mass of product produced to support your answers. b) If 3.41 g of NO2(g) produced at the end of the reaction, what is the percent yield? Percent yield actual yield theoretical yield x 100 c) Calculate the mass of the excess reactant left over.
Given the following balanced chemical equation: 2 NO(g) + O2(g) 2NO2(g) > 15.0 g of NO(g) and 15.0 g of O2(g) placed in this reaction a) Determine the limiting and excess reactant. Calculate the mass of product produced to support your answers. b) If 3.41 g of NO2(g) produced at the end of the reaction, what is the percent yield? Percent yield actual yield theoretical yield x 100 c) Calculate the mass of the excess reactant left over.
Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter3: Stoichiometry
Section: Chapter Questions
Problem 7RQ: Consider the hypothetical reaction between A2 and AB pictured below. What is the balanced equation?...
Related questions
Question
Help plss
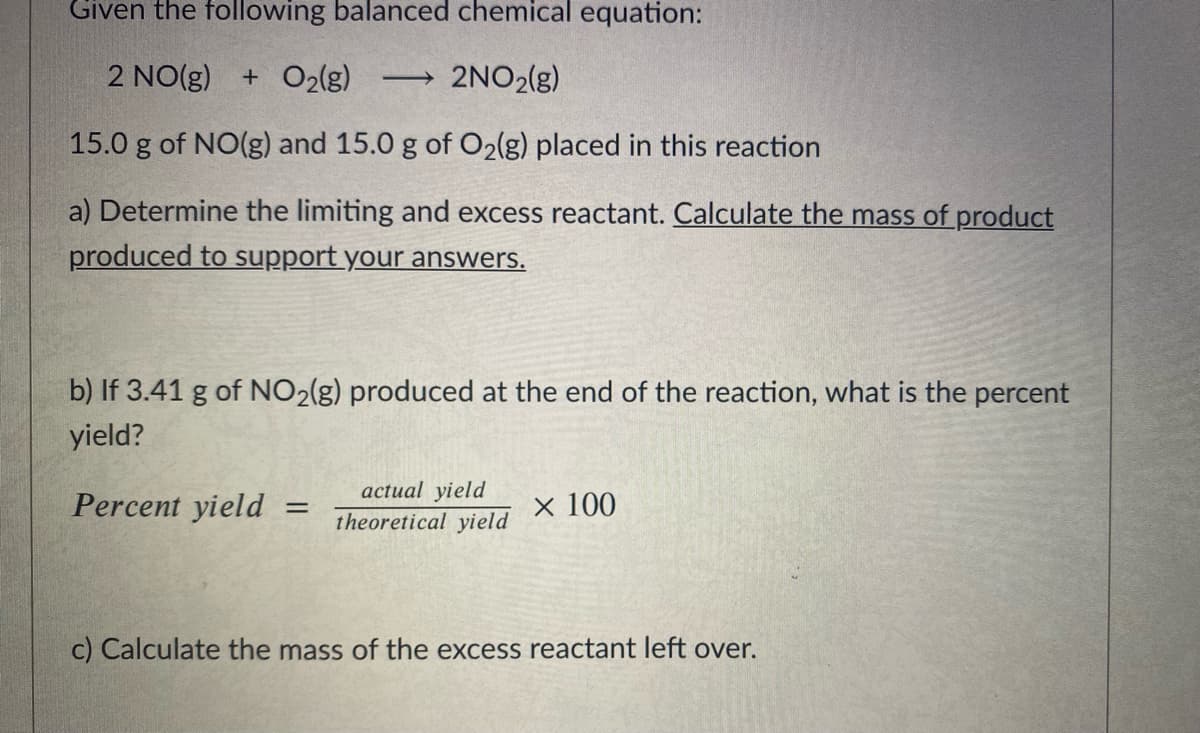
Transcribed Image Text:Given the following balanced chemical equation:
2 NO(g)
+ O2(g)
2NO2(g)
>
15.0 g of NO(g) and 15.0 g of O2(g) placed in this reaction
a) Determine the limiting and excess reactant. Calculate the mass ofproduct
produced to support your answers.
b) If 3.41 g of NO2(g) produced at the end of the reaction, what is the percent
yield?
actual yield
Percent yield
× 100
theoretical yield
c) Calculate the mass of the excess reactant left over.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 4 steps with 3 images

Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning


Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

World of Chemistry
Chemistry
ISBN:
9780618562763
Author:
Steven S. Zumdahl
Publisher:
Houghton Mifflin College Div

World of Chemistry, 3rd edition
Chemistry
ISBN:
9781133109655
Author:
Steven S. Zumdahl, Susan L. Zumdahl, Donald J. DeCoste
Publisher:
Brooks / Cole / Cengage Learning