Given the reaction that I added. How many grams of [Ni(en)3]SO4 should you use in order to make 1.25g of PbSO4? How many milliliters of 0.10M Pb(NO3)2 should be used to make this reagent in 10% mole excess?
Given the reaction that I added. How many grams of [Ni(en)3]SO4 should you use in order to make 1.25g of PbSO4? How many milliliters of 0.10M Pb(NO3)2 should be used to make this reagent in 10% mole excess?
Introductory Chemistry: A Foundation
9th Edition
ISBN:9781337399425
Author:Steven S. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Donald J. DeCoste
Chapter21: Biochemistry
Section: Chapter Questions
Problem 78AP
Related questions
Question
Given the reaction that I added.
How many grams of [Ni(en)3]SO4 should you use in order to make 1.25g of PbSO4? How many milliliters of 0.10M Pb(NO3)2 should be used to make this reagent in 10% mole excess?
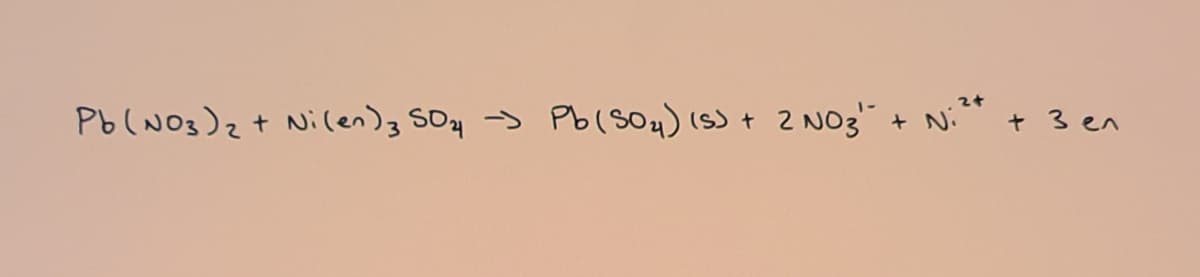
Transcribed Image Text:Pb(NO3)z+ Nilen)3 SOy >
Pb(SO4) (S) + 2 NO3"
+ N: + 3en
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 5 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

