Goal is to determine the specific heat of cylinders. 1. Data is provided in the table. You need to calculate and enter the answers in the empty columns. 2. Put the correct units in the space provided for the units in each column of the table. 3. Answer questions (1-3) and submit for the grading. 3. Use the following formulas to calculate: a) Mass of water = mass of cup and water - mass of empty cup. b) AT of water = Final temp of water - initial temp of water. c) Q (heat) into water = Specific heat of water x mass of water x AT of water. %3D %3D %3D Specific .heat of water =1 calorie / g °c ( calorie per gram per degree Celsius) d) AT of cylinder = Final temp of cylinder- Initial temp of cylinder. e) Q out of cylinder = - Q into water. (For a closed system Heat in = - Heat out) f) Specific heat of cylinder = Qout of cylinder / (mass of cylinder x AT of cylinder) %3D = Qout divided by the product of mass of cylinder and AT of cylinder. %3D Final temp. of H20 Q into Н-О Units: AT of Initial temp. of H20 Units:0C Material Mass of Mass of Mass of Н.О Units: Н-0 Units: empty cup Н.О Units: g Units:0C cup Units:g 9. 23.8 27.4 10 23.8 26.5 9. 11 23.8 27.9 10 12 23.8 26.8 g is gram here. Final temp. of cylinder Units: Q out of cylinder Units: Specific heat of Material Mass of Initial AT of cylinder Units: g temp. of cylinder Units: cylinder Units: cylinder Units: 71.6 101 27.4 69.3 101 26.5 68.2 101 27.9 67.8 101 26.8
Goal is to determine the specific heat of cylinders. 1. Data is provided in the table. You need to calculate and enter the answers in the empty columns. 2. Put the correct units in the space provided for the units in each column of the table. 3. Answer questions (1-3) and submit for the grading. 3. Use the following formulas to calculate: a) Mass of water = mass of cup and water - mass of empty cup. b) AT of water = Final temp of water - initial temp of water. c) Q (heat) into water = Specific heat of water x mass of water x AT of water. %3D %3D %3D Specific .heat of water =1 calorie / g °c ( calorie per gram per degree Celsius) d) AT of cylinder = Final temp of cylinder- Initial temp of cylinder. e) Q out of cylinder = - Q into water. (For a closed system Heat in = - Heat out) f) Specific heat of cylinder = Qout of cylinder / (mass of cylinder x AT of cylinder) %3D = Qout divided by the product of mass of cylinder and AT of cylinder. %3D Final temp. of H20 Q into Н-О Units: AT of Initial temp. of H20 Units:0C Material Mass of Mass of Mass of Н.О Units: Н-0 Units: empty cup Н.О Units: g Units:0C cup Units:g 9. 23.8 27.4 10 23.8 26.5 9. 11 23.8 27.9 10 12 23.8 26.8 g is gram here. Final temp. of cylinder Units: Q out of cylinder Units: Specific heat of Material Mass of Initial AT of cylinder Units: g temp. of cylinder Units: cylinder Units: cylinder Units: 71.6 101 27.4 69.3 101 26.5 68.2 101 27.9 67.8 101 26.8
Chapter4: Physics Of Matter
Section: Chapter Questions
Problem 1MIO: 1. In Section 4.1 in the description of matter, the following terms were introduced: elements,...
Related questions
Question
I'm having a hard time figuring out which equation I need to be using to plug in the data into the table. Help!
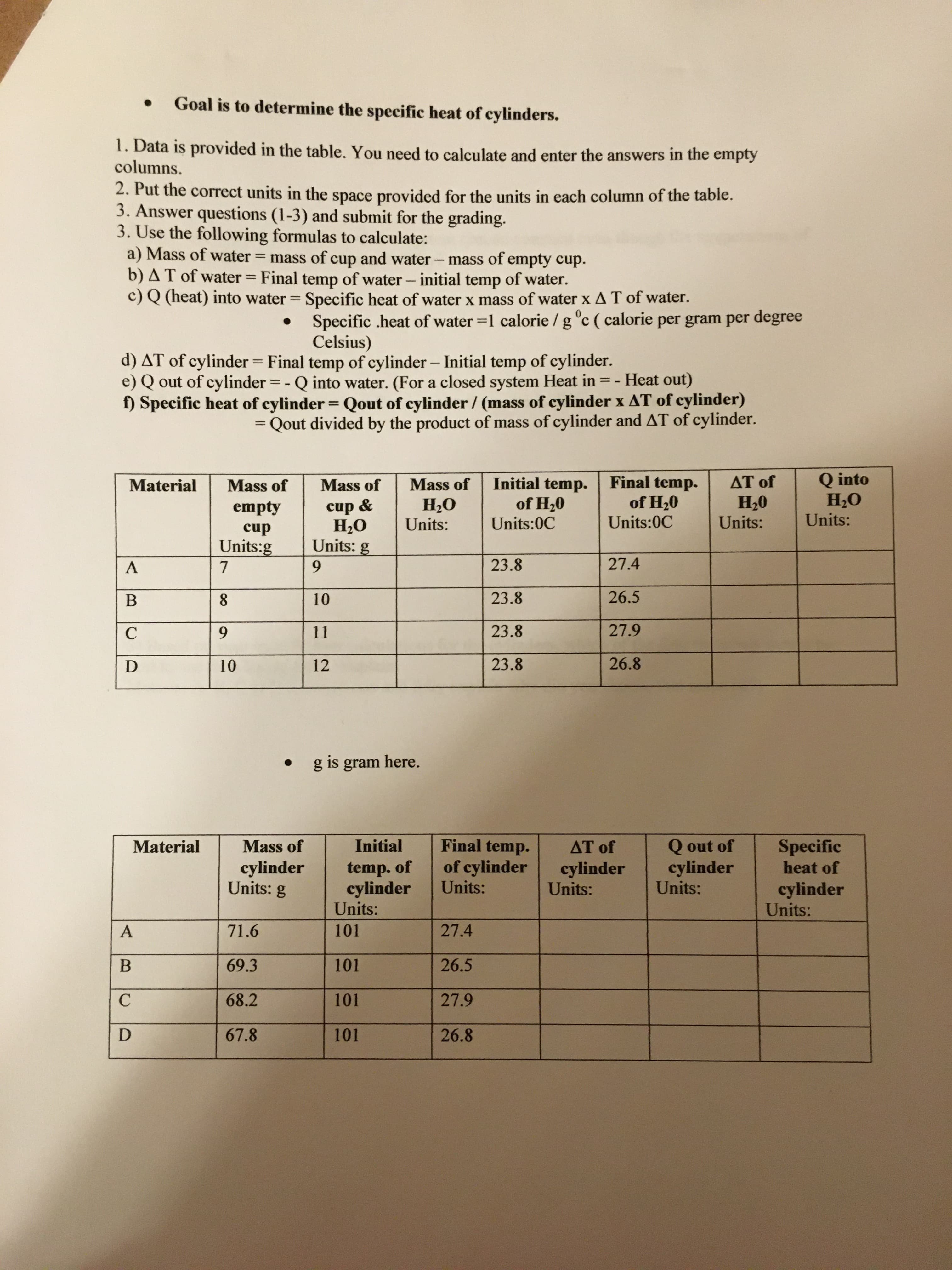
Transcribed Image Text:Goal is to determine the specific heat of cylinders.
1. Data is provided in the table. You need to calculate and enter the answers in the empty
columns.
2. Put the correct units in the space provided for the units in each column of the table.
3. Answer questions (1-3) and submit for the grading.
3. Use the following formulas to calculate:
a) Mass of water = mass of cup and water - mass of empty cup.
b) AT of water = Final temp of water - initial temp of water.
c) Q (heat) into water = Specific heat of water x mass of water x AT of water.
%3D
%3D
%3D
Specific .heat of water =1 calorie / g °c ( calorie per gram per degree
Celsius)
d) AT of cylinder = Final temp of cylinder- Initial temp of cylinder.
e) Q out of cylinder = - Q into water. (For a closed system Heat in = - Heat out)
f) Specific heat of cylinder = Qout of cylinder / (mass of cylinder x AT of cylinder)
%3D
= Qout divided by the product of mass of cylinder and AT of cylinder.
%3D
Final temp.
of H20
Q into
Н-О
Units:
AT of
Initial temp.
of H20
Units:0C
Material
Mass of
Mass of
Mass of
Н.О
Units:
Н-0
Units:
empty
cup
Н.О
Units: g
Units:0C
cup
Units:g
9.
23.8
27.4
10
23.8
26.5
9.
11
23.8
27.9
10
12
23.8
26.8
g is gram here.
Final temp.
of cylinder
Units:
Q out of
cylinder
Units:
Specific
heat of
Material
Mass of
Initial
AT of
cylinder
Units: g
temp. of
cylinder
Units:
cylinder
Units:
cylinder
Units:
71.6
101
27.4
69.3
101
26.5
68.2
101
27.9
67.8
101
26.8
Expert Solution
Heat transferred into water

Specific heat of the cylinder

Step by step
Solved in 4 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College


College Physics
Physics
ISBN:
9781938168000
Author:
Paul Peter Urone, Roger Hinrichs
Publisher:
OpenStax College