Q: A 15.0 g sample of qu water at 30.0 °C and Assuming no heat is a answer has the correc
A: i have the final answer which is 19.3 degree Celsius but i dunno how to get it., i used the formula…
Q: What is (a) temperature, (b) heat, (c) work, (d) energy?
A: Since we answer only upto 3 sub-parts, we'll answer the first three. Please resubmit the question…
Q: Equal amounts of heat are added to equal masses of substances A and B at the same temperature, but…
A: Let us assume both the substances A and B have mass of m kg each, initial temperature be T. Let the…
Q: ", heat HO
A: Given : We have to write the reaction .
Q: cal was 69.8°C. c heat of the unknown
A: Given, Mass of metal =15.963 g Mass of water = 60 g Intial temperature of water = 24.9 °c Intial…
Q: In the laboratory a student finds that it takes 173 Joules to increase the temperature of 11.1 grams…
A: Given heat is 173 j
Q: How is kinetic and potential energy differ? How are they related? Please citean everyday situation…
A: When an object is at rest the body is said to possess potential energy. in another case when the…
Q: a. K2Cr207 / Aq. H2SO4 /heat b. HNO3 / H2SO4 c. Br2 / FeBr3 d. NaOH, then H2O2 e. Sn / HCI f. NANO2…
A: In electrophilic aromatic substitution reaction, methyl group acts as ortho para directing group due…
Q: What are the units of energy?
A: The potential of the body to do some work is defined as energy. There are various forms of energy…
Q: CH3 HBr a) Br NaOC,H5 /heat b) 1.NABH, c) 2. НаО CO,C2H5 1. Hg(OAc)2/THF/H20 2. NABH,OH co.C,H, heat…
A:
Q: C. What is the overall temperature change? i °C
A:
Q: In the laboratory a student finds that it takes 49.2 Joules to increase the temperature of 14.8…
A:
Q: CuCl Cu,O, Cu²*, H,O CuBr NO2 NH2 N2 CUCN KI NH2 HBF4, heat H3CO. H.PO2,H„O
A: N2 is goood leaving group so it will eliminate and nucleophile add on Benzene ring.
Q: When 305.2 calories of heat are added to 32.02 g of copper (specific heat capacity of 0.0920 cal/g…
A: The heat 'q' which is required to raise the temperature of a sample of mass 'm' having specific heat…
Q: 1BUOK 1BUOH, heat 1BUOH, heat e .CI EESH minimum temp NaCN Br acetone minimum temp
A:
Q: In the laboratory a student finds that it takes 764 Joules to increase the temperature of 10.3 grams…
A: Given;
Q: A 100.0 g sample of solid ammonia is heated from –77.65 °C to –40 °C. How much heat (in kJ to two…
A: Mass of solid ammonia = 100 gram Initial temperature = - 77.65℃ Final temperature = - 40℃ The…
Q: KMNO4, -OH HO. C H3O+, heat H,SO4
A: We have given the organic reaction and we have to find the major product and the mechanism of the…
Q: 500.0 g of acetone is heated from –100.0 °C to 60 °C. How much heat (in kJ to two decimal places) is…
A: Given, Mass of acetone, m = 500 g = 0.5 kg Change in temperature, ∆T = 60-(-100) = 160 °C Cp for…
Q: The flow of energy due to a temperature difference is known as __________. A.Heat B.Temperature…
A: We know that Heat is form of thermal energy which is transfered between two systems with…
Q: How much heat is required to raise the temperature of 10.0 g of H2O (s) from -10 degrees C to H2O…
A:
Q: A 35.17 g35.17 g sample of a substance is initially at 24.4 ∘C.24.4 ∘C. After absorbing 2097 J2097…
A: Given mass (m) = 35.17g heat (q) = 2097 J T1 = 24.4°C T2 = 137.9°C
Q: In the laboratory a student finds that it takes 214 Joules to increase the temperature of 13.3 grams…
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: Fill in the blanks: 1. The two things that make up the are the system and In an exothermic process,…
A: We have to fill in the blanks below
Q: 1) H2SO4, heat 2) Cl2, AICI2 а) 3) dilute H2SO4, heat b) 1) `CI » AICI3 2) H,О 3) Н-(g), Pd/C H2N.…
A:
Q: heat N(CH3)3 OH H2N V NaN3, heat and then H20
A: Since we only answer up to 3 sub-parts, we'll answer the first three. Please resubmit the question…
Q: H', heat CH,CH.CH CH,CHC–OCHCHCH;CH, + H,O
A: We have to predict product along with IUPAC nomenclature.
Q: A 225.0 gram sample of copper absorbs 735 J of heat from its surroundings. What is the temperature…
A: We have given that A 225.0 gram sample of copper absorbs 735 J of heat from its surroundings. What…
Q: Calculate the amount of heat required t
A: Q= mc∆T Q= heat required c is specific heat ∆T = change in temperature
Q: A system releases 50 kJ of heat while having 25 kJ of work done on it.The change in energy ΔE is?
A: According to the First law of Thermodynamics: where ∆E is the change in the Internal Energy of the…
Q: Is heat a matter or not? Why
A: Matter is defined as the substances that has mass and occupies space. Matter is composed of atoms or…
Q: Heat is typically measured in A. C B. F C. Joules D. Grams
A: Heat is a form of energy which is transferred from one body at higher temperature to another body at…
Q: e Joule e Energy is absorbed e Energy can change forces from one type to another e freezing e…
A: Thermodynamics is a branch of science which deals with the concept of heat and temperature and also…
Q: Br2 H,C=P(Ph); E F G CH;COOH H,C=CH2 heat H
A: Compound H is 1-ethyl-2-methylcyclohex-1-ene
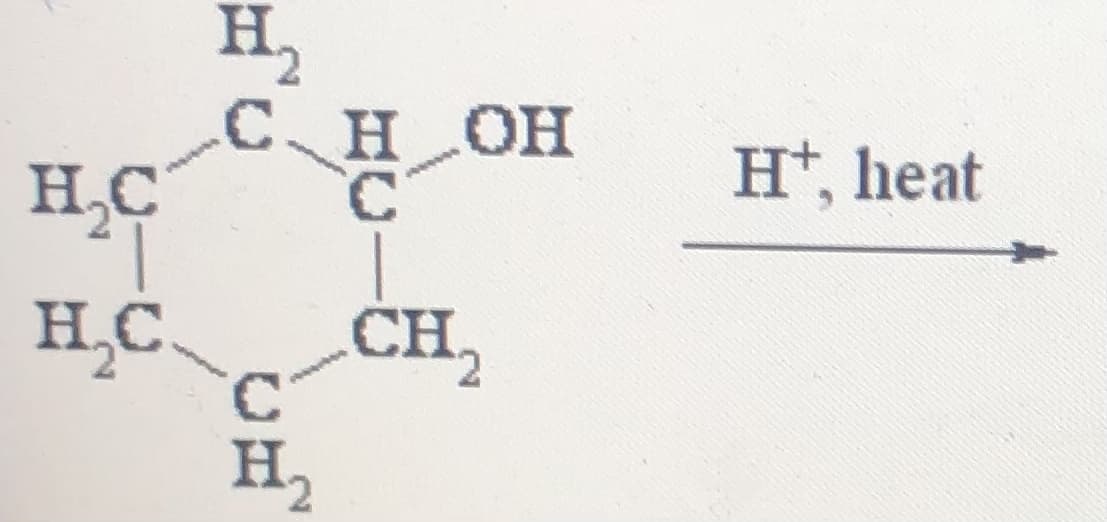
Q: H*, heat OH C. H, H.C
A:
Q: How heat was measured traditionally ?
A: Heat is always defined as the measure of transferred energy. In the exothermic process, heat is…
Q: 1. KMNO4, HO“, heat 2. H;O*
A:
Q: A 25.0 mL sample of ethanol (density=0.789 g/mL) initially at 7.0 Celsius is mixed with 35.0 mL of…
A: Volume of ethanol = 25.0 mL Density of ethanol = 0.789 g/mL So, mass of ethanol = volume * density =…
Q: _cOOEt H,O" heat (A) (B) COOH (D) (C)
A: Under this given condition, I. e. Heating condition in acidic medium, carboxylic acid undergoes…
Q: H,SO4, heat CH;COOH он HCI CH3 H,C KMNO, HCI PCI4 HBr HCI PBR3 2 NaOH
A: The given compound is Butan-2-ol as reactant.
Q: SOCI, HO heat
A: Given reactant = butanoic acid and SOCl2 To determine :- major organic product of this reaction
Q: The specific heat of toluene (C7H8), is 1.13 J/g.K. How many joules of heat are needed to raise the…
A: The given data is as follows: The specific heat of toluene = 1.13 J/g.K The mass of toluene = 62.0…
Q: 11) C,H,O" OC,Hs OH ? H" heat
A:
Q: ch heat is
A:
Q: How much heat energy is required to raise the temperature of 0.361 kg of copper from 23.0 °C to 60.0…
A: Given, Mass of copper = 0.361 kg Initial temperature = 23.0 ∘C Final temperature = 60.0 ∘C Specific…
Q: What is heat? How does heat differ from thermal energy? Under what condition is heat transferred…
A: Heat: Heat is a form of energy that is transferred from one body to another because of difference in…
Q: Use an example to explain why heat is a path function and not state function.
A:
Q: What is heat capacity?
A: Heat capacity is a physical property of matter, defined as the amount of heat needed to be supplied…
Step by step
Solved in 2 steps with 2 images

- ballance these b) (NH4) 2O (aq) + Pb(C2H3 O2)2 (aq) --> PbO (s) + NH4 C2H3 O2 (aq)c) H2SO4 + B(OH)3 ----> B2(SO4)3 + H2OUse the rule of 13 and mass spect data attached to propose the molecular formula. 151 represents both the M+ and base pwakZn + 2 HCl ------> ZnCl2 + H2 2) When HCl(aq) is poured over the zinc metal, the liquid begins to fizz with small bubbles. What are the bubbles made of? Combustion Tests of Hydrogen –
- 3. Please complete all parts to the following table:Cocaine(C17H21O4N) is a natural substance found in the leaves of the coca plant, which has been used for centuries as a local anesthetic and stimulant. Illegal cocaine arrives in the USA either as a pure compound or as the hydrochloride salt(C7H21O4NHCl). At 25 degrees celsius, the salt is very soluble in water(2.59kg/L), but pure cocaine is much less so(1.70g/L) a. What is the maximum amount of salt in grams that can dissolve in 50.0mL of water? b. When a 25mL aqueous solution that contains 62.50 g of the salt is treated with NaOH, the salt is converted to pure cocaine. How much water in liters is needed additionally to dissolve the formed pure cocaine,(AH is all in kJ mol-1) AH 2 Na = 0 AH 2 H20 = -285.8 AH 2 NaOH = -470.1 AH H2 = 0

