Q: 1. Draw all the possible resonance structures for the Meisenheimer complex below. F OCH 3 O₂N.
A: We have been given meisenheimer complex. We have been asked to draw all possible resonance…
Q: Rank the following compounds in order of increasing strength of intermolecular forces: KF, CH₂OH,…
A: The attraction force present between the molecules is known as intermolecular force.
Q: mechanism 1 overall reaction is 2NO + Cl2 --> 2NOCl NO + Cl2 = NOCl2 fast equilibrium NOCl2 +…
A: Answer: Slowest step of the reaction is considered as rate determining step and rate of reaction is…
Q: Below is a phase diagram for compound Y. The normal melting point of Y is most likely: K 0 10 20 P…
A: The melting point is the temperature at which a solid melts into a liquid at atmospheric pressure,…
Q: A certain reaction was found to have a rate constants 7.04x10-3 min-1 at 152°C and 5.54×10-2 min-1…
A:
Q: At a particular temperature, N₂O5 decomposes according to a first-order rate law with a half-life of…
A:
Q: Geologists can estimate the age of rocks by their uranium-238 content. The decay of U-238 is…
A: Given : half life = 4.5 billion years Tip : apply first order reaction
Q: 100 mL of water contain 100 mg of strontium Sr²+. What mass of solid potassium chromate K₂ CrO4…
A:
Q: 2. Calculate the amount of energy (in kilojoules) needed to heat 346 g of liquid water from 0°C to…
A: Mass of water = 346 g Increase in temperature of water = from 0°C to 182°C The specific heat of…
Q: Draw the line form into condensed formula. H₂C CH3 CH₂ CH3 CH3 CH3 CH3
A:
Q: Lab Report Sheet: Part A: Determination of Mass Percent of Iron (Fe) Mass of evaporating dish and…
A: We need to set up the equations for Part A, B and C.
Q: H H
A: Electrons move from electron rich center towards electron deficient center. Here arrow notation is…
Q: Can u help me with the question 2 for wrtie the fomula for each of the following ionic compounds:…
A: Given : name of compounds
Q: Predict the major product(s) for each of the following reactions. Show both enantiomers if a racemic…
A: -> In presence of peracid there is formation of epoxide . -> Alkene give markovnikov addition…
Q: Part A Calculate the osmotic pressure of a solution containing 20.00 mg of hemoglobin in 13.4 mL of…
A: Given -> Weight of hemoglobin= 20.00 mg Volume= 13.4 ml Temperature= 15°C = 15 + 273 = 288 K…
Q: 11. Below is the reaction rate of a particular enzyme in the body. Explain what how the enzyme…
A: For an enzyme based reaction, the quantity of product generated per minute is described by the rate…
Q: What is the mass of 4.89 x 1021 platinum atoms?
A: Mass of 6.0221 x 10^23 platinum atoms is 195.08g Mass of 4.89 x 10^21 platinum atoms is(m) ____g
Q: 2 3 5 6 Predicate the products of each reaction: Aldehydes + Ketones CH;CH + HC=N o O CH₂-C-OCH, =O…
A: Here, we have to draw the products for the given organic reactions.
Q: O + 2 A
A: When a conjugate diene reacts with dienophile in presence of heat they undergo cyclo addition…
Q: If a reaction is exothermic, and K=100 and Q=1,000, would reactants or products be favored and in…
A: Given: K = 100 Q = 1000 To determine: What direction would equilibrium shift?
Q: How many micrograms of KMnO4 (Formula Weight: 158.04 g/mole) are contained in 50mL of 125mM KMnO4…
A: Given -> Formula weight of KMnO4= 158.04 g/mole Volume= 50 ml = 0.050 L (1L = 1000ml) Molarity=…
Q: (1) The element with an electron configuration of 1s22s22p63s²3p² is in group (2) The element with…
A: Prediction of Group from electronic configuration : For s block: Group Number = Number of valence…
Q: 13. Draw all potential constitutional isomers and stereoisomers for the compound below; AND point…
A: Constitutional Isomers and Stereoisomers Constitutional (structural) isomers arecompounds with the…
Q: Find the nuclear binding energy of uranium-234 (atomic mass = 234.040947 amu) in units of joules per…
A: Given: The atomic mass of uranium-234 is 234.040947 amu. The mass of the proton is 1.672623×10-24 g.…
Q: Nat - Hi + t →Nat X Pact A: Identify the lewis acids, bases; determine if they are bronted ac pact…
A: Since you have asked multiple questions, we will answer only first question for you. In order to get…
Q: 1) 235 U+ n20 Sr+ ? + 3 n 38 13 2) 12 C + " He →→ 140 + ? ➜>>> 6 2
A: Given are nuclear reactions. Nuclear reactions are reactions in which two or more radioactive nuclei…
Q: Fill in the orbital energy diagram for nickel. 4s E 3s- 2s 1s 3p- 2p- 3d AV AV The lowest E levels…
A: This question is related to atomic structure. In the given question, it is asked to draw orbital…
Q: A gas occupies 2.22 L at 367 K. What is the volume at 696 K? 4.21 L 1.17 L 0.289 L 4.20 L 3.21 L
A: Given that, Volume of the gas V1=2.22L at temperature T1=367K We have to calculate volume of the…
Q: This graph shows how the vapor pressure of three liquids varies with temperature: vapor pressure,…
A: We have find out the answer.
Q: 1A H 2A Li Be Na Mg 3B 4B 5B 6B 7B8B K Ca Sc Ti V Cr Mn Fe Co Ni Cu Zn 1B 2B 8A 3A 4A 5A 6A 7A He B…
A: The electronic configuration can be written by using three rules : Aufbau principle The Pauli's…
Q: Which drawing best represents the resonance hybrid for the given molecule? OH to Об Oo'x Oo OH 5- OH…
A: Resonance structure: If the Lewis structure of a molecule or ion cannot explain by a single…
Q: A nuclear stress test utilizes a gamma-emitting radioisotope such as thallium-201 to follow the flow…
A:
Q: Which of the following is an example of potential energy? O A compressed spring OA battery O Water…
A: Please find your solution below : Potential energy is the form of energy held or stored by an object…
Q: ОН POCIĄ pyridine
A: In presence of POCl3 and pyridine there is removel of Water and double bond formed.
Q: A 'H NMR spectrum is shown for a molecule with the molecular formula of C9H10O2. Draw the structure…
A: Molecular formula = C9H10O2
Q: Consider the fictional element A (which has a molar mass of 7.00 g/mol) and the fictional element D…
A: Given: The molar mass of element A and B is 7.00 g/mol and 13.00 g/mol, respectively. The mass of…
Q: Can you help me with the explanation of the molecular polarity, partial charge, formal charge and…
A: we have to define , molecular polarity, partial charge, formal charge with example
Q: 1. Can you help me what does mean for the Valence Shell Electron Pair Repulsion (VSEPR) Theory and…
A: “Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: 9) Which of the following compounds does not have the molecular formula C6H₁4O? (a) 1-hexanol (b)…
A: The molecular formula is the expression of the number of atoms of each element in one molecule of a…
Q: The reduction of iron(III) oxide (Fe,O,) to pure iron during the first step of steelmaking, 2 Fe₂O₂…
A:
Q: If a set of consitutional isomers had the same melting point it would be a coincidence, as…
A: Constitutional isomers are molecules with the same molecular formula but different arrangement of…
Q: Having watched the video, you should have an understanding of which bonds can and cannot rotate…
A: Given : structure of molecule Tip : single bond can rotate freely but double bond and triple bond…
Q: Explain why the first ionization energy of sodium (Na) is significantly less than that of neon (Ne).
A: Ionization energy is the amount of energy required to remove an electron from an atom or a positive…
Q: Calculate the height of a column of water equivalent to 1.00 bar. The density of water is 1.00…
A: We have find out the height of a column of water equivalent to 1.00 bar.
Q: Four liquids are described in the table below. Use the second column of the table to explain the…
A: Given 2.1 g potassium nitrate in 150. mL water 2.1 g of potassium chloride in 150 mL water 2.1 g HI…
Q: Fill in the orbital energy diagram for calcium.
A: Answer: Atomic number of calcium is 20, a neutral atom of Ca will be having 20 electrons in its…
Q: Sometimes in lab we collect the gas formed by a chemical reaction over water (see sketch at right).…
A: The given question can be solved using the Ideal Gas Law: PV = nRT Where: P is the pressure V is…
Q: 3-3. The density of 2-methyl-2-butanol is 0.806 g/mL. How many mgs represent an aliquot of 430 µL?…
A: 3-3. Given,Density of 2⎯methyl⎯2⎯butanol = 0.806 g/mL Formula weight of 2⎯methyl⎯2⎯butanol = 88.15…
Q: What reagent(s) is/are needed to get the desired alcohol selectively as the major product? ? OH
A: Here it is an reduction Reaction. The carbonyl compound is reduced to the secondary alcohol. There…
Q: Consider the following data on some weak acids and weak bases: name acid nitrous acid hydrocyanic…
A: Salts are formed by the neutralisation reaction between acids and a bases.Salts underogo ionization…
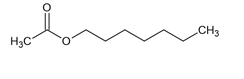
Help naming this one. Than k you!
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images


