Q: Predict the order or increasing electronegativity from the following elements:Cs, At, Tl, I
A: The electronegativity of elements has to be arranged in increasing order.
Q: 4) a) What do you understand by the following: i) Electronegativity. n) An intermolecular force.
A: Note: Since you have posted two independent questions in the same request, we will solve the first…
Q: Determine if the bond is polar or non-polar H-F
A: The answer to the following question is given as -
Q: v C-0 v F-0 V F-O v F-C The most polar bond is
A: Given Partial Charge Given Bond Partial charge C-O F-O F-C Partial charge…
Q: Malonic acid has the molecular formula C3H404 and has the connectivity shown in the figure. When the…
A:
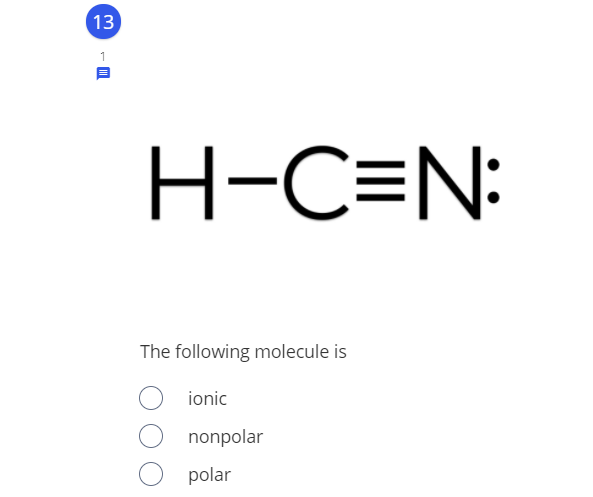
Q: H-C=N: The following molecule is Tonic polar O nonpolar
A: HCN is a covalent molecule as carbon and hydrogen share electrons of each other and forms a covalent…
Q: ClO- ,does it have linear (180o bond angle) or tetrahedral (o ?) VSEPR shape.
A: The molecular geometry of a molecule depends on the electron geometry and number of lone pairs…
Q: Which of the following is pure covalent bond? а. В-F O b. C-C Ос. Н-N O d. H-0
A: Bond will be pure covalent if the electronegativity and size of both the atoms are same, i.e. atoms…
Q: hat is the FE–S– O bond angle in SOF4
A: The molecular geometry helps us to identify the exact geometry of the molecule.
Q: Liquid Formula Molar Lewis Structure Polar or Strongest Mass Nonpolar Type of IM Methanol CH3OH
A: We have to tell the molar mass, polarity, forces of given molecules and give their Lewis structure
Q: Which of the following is the correct setup and answer for the formal charge of sulfur in SH2?
A:
Q: Arrange the following bonds from least polar to most polar: H-F, H-N, H-O, H-Cl
A: Given Bond with atom = H- F , H-N , H-O , H-Cl Arrange least polar to most polar bond = ?
Q: The molecule iodine pentabromide is polar. O True False
A: Formula for determining the hybridization of central atom :- x= (V+M-C+A)/2 V = valence electrons…
Q: A classmate of yours is convinced that he knows everythingabout electronegativity. (a) In the case…
A: a.
Q: s polar or non-polar
A:
Q: compounds have a dipole moment if they are a. diatomic molecules made up of atoms of the same…
A:
Q: Whats the lewis structre for 9. ClF3 10. ClO2 11. IF5 12. XeF6 13. BrO3 14. IF4- 15. XeO4-
A: To draw the Lewis structure if the given compounds In Lewis structure all atom try to complete its…
Q: Given below is a Lewis structure skeleton of a molecule. Rank the C-N bonds, A, B, C, in terms of…
A: Bond length order in given molecule is, A>B>C i.e C- N ( single bond) > C-N ( double bond)…
Q: How can I describe what happens when a polar covalent and a nonpolar covalent substance are…
A: Non-polar covalent compounds are defined as the chemical compounds where the difference in…
Q: Which of the following statements best describes the polarity of carbon tetraiodide, CI4? W: a…
A: The best statements which describes the polarity of carbon tetrachloride is given below.
Q: calculate dipole moment of C-N? Provide explanation of how to calculate the formula and then what…
A: The quantity that measure the separation of two opposite charges known as dipole moment. It is…
Q: How many pi and sigma bonds are in CIO3-1
A: Firstly by drawing the lewis structure of ClO3- ion we can count the number of sigma & π Bonds.
Q: Is this molecule polar? CH3 H3C CH3 O It is polar O It is nonpolar O Not enough information to tell
A: The structure of teimethylamine (N(CH3)3) is pyramidal. We know that N is more electronegative than…
Q: molecule or polyatomic ion polar or nonpolar? atom closest to negative side polar HBr nonpolar O…
A: Answer: All three compound are covalent compounds, therefore their polarity or non-polarity will…
Q: The molecule H3C-X is most polar when X is: O OH O CH3 O F
A: Polar covalent bond:The covalent bond is formed by the sharing of electrons between the atoms. If…
Q: How many sigma (o) and pi (TT) bonds are in the following H -C-C' O A. 60, 211 O B. 70, 11 O C. 80,…
A:
Q: Is the molecule below polar or non-polar? Why? Hint: the electronegativity of Ec is 3.4 :F-Ec =N:…
A: Concept; Dipole moment equal to zero called the non-polar molecule . Dipole moment not equal to…
Q: The structure consisting of a positively charged neutral O True O False of an atom, theoretically
A: Please find your solution below : Atom is smallest unit and basic building block of matter that…
Q: List the bonds P-Cl, P-F, O-F, and Si-F from least polar to most polar.
A: We have to arrange the given bonds in increasing order of polarity. Electronegativity: Ability of…
Q: H. H3C А. N-C-N В. H3C H / H H HI
A: Concept is based on bond order. Satisfy the valence first
Q: C Show the charge distribution in the following molecules, and predict which are polar molecules: a.…
A: Polarity of a bond is due to the difference in electro-negativities of atoms presented in it. The…
Q: polar or nonpolar? at O polar
A:
Q: Compound: Phosphorus Hexachloride Chemical name: Chemical Formula: Polarity of bond: Molecular…
A:
Q: pe "polar" or "nonpolar". polar
A:
Q: Which of the following is the Lewis dot structure of ethylene glycol? H H COH H:0:5 H'CH H'CH H.…
A: Answer: VII
Q: CF4 contains polar C - F bonds. Is this molecule polar or non-polar? (Hint: Include a diagram)
A:
Q: (a) True or false: The hydrogen atom is most stable when ithas a full octet of electrons. (b) How…
A: Hydrogen atom’s electronic configuration is shown below and according to which when hydrogen accepts…
Q: (d) Explain the following terms, giving an example of where each occurs. (i) Polar covalent bonding…
A:
Q: Between Baron, Oxygen, Hydrogen, Carbon, and Nitrogen, which 2 atoms can have incomplete octets?
A: Octet :- The existence of eight electrons in the valence shell of an atom is called octet. An atom…
Q: Compound: Carbon Trioxide Chemical name: Chemical Formula: Polarity of bond: Molecular Polarity:…
A: Given compound is carbon trioxide that means carbon with three oxygen
Q: 8 A diatomic molecule like H, is considered ionic nonpolar polar
A: Polarity is a separation of electric charge leading to a molecule or its chemical groups having an…
Q: Which of the following is the correct setup and answer for the formal charge of nitrogen in NO3- ?
A: Formal charge = (valence electrons)-(nonbonding valence electrons-bonding electrons)/2
Q: 19. For the molecule XeF4, draw a proper Lewis structure that clearly identifies all electrons…
A:
Q: Complete the following table Molecule Valence Lewis Structures Molecular Electrons Geometry OS…
A: Lewis dot structure - It is the structural representation of the molecule which is shown by the…
Q: Illustrate the Electrostatic potential plot of ethylene ?
A: Ethylene is an alkene molecule that contains one double bond. The molecular formula of ethylene is…
Q: MOLECULAR ELECTRON-DOT ELECTRON-DOT SKETCH GEOMETRY (SHAPE) POLAR OR FORMULA SYMBOL STRUCTURE…
A: YES, your home work is correct. Well done and keep it up.
Q: Suppose that any given kind of bond, such as 0-H, has a characteristic electric dipole. That is,…
A: dipole moment of Oh bond,μ=1.86D This dipole moment of bond is due to electronegativity of…
Q: 5. Which of the following bonding units has the largest dipole moment? А. С-О В. С-о С. С-С D. C=N…
A: The solution is provided below:
Q: molecule or polar or nonpolar? atom closest to polyatomic ion negative side polar Cl, 0. nonpolar…
A:
Step by step
Solved in 2 steps

- All of the following have the same delta H f except A. H2 (g) B. C(s) C. H2O (l) D. Fe (s) E. N2(g)what is the value of the partial charge on the H atom in HCL molecule? HCl has a dipolle moment of 1.08D and the H-CL distance is 1.27Aphosphogene ,Cl2=O, has a smaller dipole moment than formaldehyde,H2C=O even though it contain electronegative chlorine atoms in place of hydrogen .Explain
- 7- Write a molecule with at least 4 atoms that has an inversion center.17. Which of the following does have delocalized molecular orbitals? Z of N = 7, O = 8, H = 1, C = 6 Hint: in the followings the overall charge is shown in the parenthesis. a.C03(-2)b.CH4c.NH3d.H20Phosgene, Cl2C=O, has a smaller dipole moment than formaldehyde, H2C=O, even though it contains electronegative chlorine atoms in place of hydrogen. Explain.
- 1. How many molecular orbitals describe 2,4,6-Octatriene ?There are two compounds of the formula Pt(NH3)2Cl2: The compound on the right is called cisplatin, and the compoundon the left is called transplatin. (a) Which compoundhas a nonzero dipole moment? (b) One of these compoundsis an anticancer drug, and one is inactive. The anticancerdrug works by its chloride ions undergoing a substitutionreaction with nitrogen atoms in DNA that are close together,forming a N—Pt—N angle of about 90°. Which compoundwould you predict to be the anticancer drug?In formaldehyde, CH2O, where carbon is the central atom, the formal charge on the oxygen is zero and the hybridization of the oxygen atom is sp2. True False
- How many sigma and hoe many pi bonds are there in one molecule of 3,3-dimethylpentane?Sort the following molecules by molecular geometry by dragging and dropping each symbolic representation into the appropriate category: See-saw, Trigonal Bypryamidal, or neither see-saw or trigonal Bypryamidal. SF4 PCl3 NH3 PBr5What is the correct electron configuration for the molecularorbital of B2+? (5B) diborone

