Q: Use full-headed a. Br: :0 b. CH3-C-CH3 CI: curved arrows to show the movement of electrons in each…
A:
Q: 1. Draw the structures of hexane, cyclohexane, 1-hexene and cyclohexene.
A: Since you have asked multiple type questions, we will solve only first questions for you. If you…
Q: The volume of water needed to dissolve 0.0644 grams of barium sulfite is Assume no volume change…
A: Check the Ksp and then find out the molarity of the solution. Use moles and molarity to calculate…
Q: Compound H₂C O₂N NH₂ NH₂ Conjugate acid H3C O₂N + NH3 + NH3 pk, value 5.08 1.00
A: According to Bronsted-Lowry acid base theory the compounds which have ability to donate proton or…
Q: 16) Identify the major product when toluene reacts with Br2 in the presence of heat and light. a)…
A: The reaction is proceed by free radical mechanism , the mechanism is given below
Q: 8. Molecular iodine, I2(g), dissociates into iodine atoms at 625K with a first-order rate constant…
A:
Q: Consider the insoluble compound aluminum phosphate, AIPO4 . The aluminum ion also forms a complex…
A: The reaction of aluminum phosphate with hydroxide ions is written below. The charged AlOH4- is…
Q: NBS CCIĄ A
A: NBS is known as N-bromo succinamide, it proceeds reaction with radical mechanisms.
Q: Synthesis
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: b. OH CH₂CH₂OH Н+
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: < Back Tutorial-quantum and groupT... 13 Problem 13 a) The moment of inertia of 'Li₂ is 4.161 x…
A: a) To calculate the bond length of 7Li2, we can use the formula for the moment of inertia of a…
Q: 6. Which reaction has the (largest, smallest) equilibrium constant at 25°C? (A) Cl₂ (g) + F₂ (g) = 2…
A: We know that gibbs free energy and the equilibrium constant are related as ΔG° = – RT lnK (a) ΔG° =…
Q: 4. For the reaction, PC13 (4) + Cl₂ (g) = PCI; (s) (A) [PC3][Cl₂]/[PCla] what is the expression for…
A: Note: Here, I am answering question number 4 as you have asked. Equilibrium constant expression…
Q: 1. a) Which non-metallic compounds have density values between 1.4 and 2.9 g/mL? b) What…
A: Non metal having density in between 1.4 to 2.9g/ml are benzophenone, bromo form, chloroform,…
Q: Does a reaction occur when aqueous solutions of magnesium sulfate and copper(II) nitrate are…
A: Balanced molecular equation: Balance molecular equation can be define as the reaction in which…
Q: Suppose you have a 0.240 molar solution of Na_3PO_4. How many moles are in 10.0mL of that stock…
A: Given, concentration of Na3PO4 solution = 0.240 molar = 0.240 mol/L Volume of Na3PO4 solution = 10.0…
Q: In the laboratory you are given the task of separating Ca2+ and Ag+ ions in aqueous solution. For…
A: Precipitation reaction : It is defined as a chemical reaction occurring in an aqueous solution where…
Q: What is the pH of the solution that results when the following solutions are combined? 0.020…
A: Answer: Any solution that resists the change in its pH on adding small quantity of acid or base is…
Q: 20) What is the major product of the following reaction? 1. Excess NaBH4 iii 2. H3O+ 2) even НО c)…
A: -> NaBH4 is reducing agent but it can not reduce carboxylic acid and ester . -> NaBH4 can…
Q: Consider this reaction: 2N₂O, (e)-2N₂O₂ (g) + 0₂ (8) At a certain temperature it obeys this rate…
A:
Q: A student performed the first determination in Part 2. They reacted 61.33 g of 1 M HCl with 0.6615 g…
A:
Q: Consider the following pairs of structures. Identify the relationship between them by describing…
A: The stereo centers which are having non-superimposable relationship are called enantiomers and these…
Q: Draw the condensed structure of the smallest organic molecule that produces sodium…
A: A carboxylic acid has the general formula R-COOH, where R is an alkyl or aryl group. Salt and water…
Q: How many grams of Sr(OH)2 are needed to neutralize 4.5 mL of 0.34 HNO3? 0.56 g 1.6 g 0.098 g 0.093 g…
A: 3) Given, Concentration of HNO3 solution = 0.34 mol/L Volume of HNO3 solution = 4.5 mL = 0.0045 L…
Q: 2) Which of these molecules is most prone to decarboxylation? a) F3C مشر د م ش *OH A *OH
A: Decarboxylation means the loss of CO2. The compound having beta keto acid will undergo…
Q: Functional Group Name Chemical Formula -H3 -OH =O -COOH -NH2 -SH -PO4 Structural Formula Found in…
A: A question based on molecules. A table with blanks is prescribed, for which the blanks are to be…
Q: Find the Conductance values: Ion / compound + Na + H30* SO4-² -2 OH CH3COO C1 HC1 NaOH NaCl CH3COOH…
A: Conductance is the ability of a substance to conduct electricity, and it depends on the number of…
Q: Which statements are true about the p orbital shown? 1. It can hold up to two electrons. 2. It can…
A: Two mcq are given below correct and incorrect options are explained.
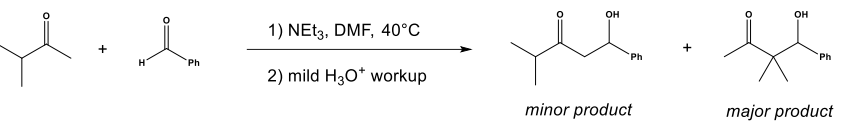
Q: Give detailed Solution with explanation needed
A: Ketals and acetals on hydrolysis gives ketone as the main product and alcohols as byproducts.
Q: Draw the product(s) of the following reactions. -CEC-CH₂ Na/NH₂(1) H₂C . Consider E/Z…
A: Answer :
Q: What is the point group for molecule C? I already found the point group for molecule A (C2h) and B…
A: Well, we will first find out the major rotational axis of symmetry and then we will observe the…
Q: Match the values below with the arrows that depict the value. Potential Energy B Reaction coordinate…
A: Reaction enthalpy - Difference of energy between product and reactant species Activation energy -…
Q: What values of J may occur in the terms ³D, 4D, 2G? How many states belong to each level?
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: When the volume of a gas is changed from 3.75 L to 6.52 L, the temperature will change from 65.0 °C…
A:
Q: For the reaction below, Kc = 4.60 × 10⁻⁶. Note Kc is sometimes called K. What is the equilibrium…
A:
Q: What is the pH after a 30.0 mL of 0.10 M HCOOH solution is titrated with 20.0 mL of 0.20 M NaOH?…
A: Answer: When acid and base solutions are mixed then neutralization reaction takes place and pH of…
Q: solution contains 1.13×10-2 M chromium(III) nitrate and 1.16×10-2 M nickel(II) acetate. Solid…
A: The question is based on the concept of solubility product principle. It that states that when a…
Q: Explain based on the molecular structure of Teflon why it would have the non-stick and chemically…
A: Teflon is a brand name for polytetrafluoroethylene (PTFE), which is a synthetic polymer made up of…
Q: The pH of a 0.0830 M solution is 5.1. What is in that solution? = M. What is = M.…
A:
Q: Macmillan Learning Imagine two solutions with the same concentration and the same boiling point, but…
A: Given Ethanol normal Boiling point: 78.4 Kb : 1.22 CCL4 normal boiling point: 76.8 Kb : 5.03
Q: Sketch the ultraviolet absorption spectrum of 1,3-Butadiene, matching peaks to electronic…
A:
Q: The rates of heterogeneous reactions typically depend on the solid and a liquid. surface area…
A: We know, there are two types of reactions - Homogeneous and Heterogeneous Reactions.
Q: When 22.0 mL of a 9.14x10-4 M magnesium nitrate solution is combined with 15.0 mL of a 7.17x10-4 M…
A: Solubility product quotient, Qsp is the product of the concentrations of the products, each raised…
Q: When 12.0 mL of a 6.96x10-4 M sodium carbonate solution is combined with 25.0 mL of a 2.46x10-4 M…
A: Given: Concentration of Na2CO3 = 6.96 × 10-4 M Concentration of AgF = 2.46 × 10-4 M Volume of AgF…
Q: What is the voltage of this cell at 298 K? Cu(s) Cu²+ (0.406 M) ||Ag+ (2.72 x 10-³ M) | Ag(s) E…
A:
Q: Write the general formula for calculating the average rate based on the disappearance of thiosulfate…
A: For any reaction: aA+bB→cC+dD The average rate is denoted as: rate=-1a∆A∆t=-1b∆B∆t=1c∆C∆t=1d∆D∆t…
Q: Consider the reaction, A (g) + B (g) = 2 C (g) R I C E Kc = 2.0 If the initial concentrations are…
A: Equilibrium concentration can be calculated by applying ICE table calculations.
Q: The configuration for the alkene is OE OZ O neither E or Zin this case H3C- CH₂OCOCH3 H₂C OH COOH…
A:
Q: Calculate the mass of water produced when 8.50 g of butane reacts with excess oxygen. 2C4H10(g) +…
A:
Q: Each of the insoluble salts below are put into 0.10 M hydrochloric acid solution. Do you expect…
A: Solution: Introduction- The solubility of a sparingly soluble salt is decreased when it comes into…
Step by step
Solved in 2 steps with 2 images

- Give the functions of the following ingredients, then name a branded/commercial skin or hair care product where the said material is used. Tetrasodium EDTA Cholesterol Silicone Oil 200/50 cst BETAINE Carbopol Ultrez D & C Red No. 17 ( CI No. 26100) Bisabolol (Alpha Bisabolol) Ceresin Wax Disodium Lauryl Sulfosuccinate Hydroxypropyl MethylcelluloseCarbonic anhydrase of erythrocytes (?r 30,00030,000) has one of the highest turnover numbers known. It catalyzes the reversible hydration of CO2.CO2. H2O+CO2↽−−⇀H2CO3H2O+CO2↽−−⇀H2CO3 This is an important process in the transport of CO2CO2 from the tissues to the lungs. If 33 ?g of pure carbonic anhydrase catalyzes the hydration of 0.10 g0.10 g of CO2CO2 in 1 min1 min at 37 °C37 °C at ?max, what is the turnover number (?cat) of carbonic anhydrase? Please provide only typed answer solution no handwritten solution needed allowedPlease don't provide hnadwrritten solutrion .....
- TRUE OR FALSE SAPONIFICATION: I. Can be carried out with HCl II. NaOH undergoes hydrolysis III. Its principle is used to determine the number of milligrams of KOH required to neutralize the fatty acids from the complete hydrolysis of 1g of fat.Determine the branch points and reducing ends of amylopectin. A 0.5g sample of amylopectin was analyzed to determine the fraction of the total glucose residues that are branch points in the structure. The sample was methylated and then digested, yielding 50 μmol of 2,3-methylglucose and 0.4 μmol of 1,2,3,6-tetramethyl glucose. (MWglucose = 162 g/mol, minus H2O) What fraction of the total residues are branch points? How many reducing ends does the amylopectin have?Do 5(c) asap please
- will #4 not be adressed?A bloodhound, with an acute sense of smell, sits at one end of a long, unventilated corridor.At the other end of the corridor a small amount of an aerosol is released containing the volatile, fragrant compounds: acetaldehyde (C2H4O) jasmine lactone (C10H16O2) acetoin (C4H8O2) hexyl acetate (C8H16O2) 1-octen-3-one (C8H14O) Rank the compounds in the chronological order they are detected by the hound. 1-octen-3-one acetoin jasmine lactone hexyl acetate acetaldehydeMention two examples of emulsifying agents for o/w emulsions and w/oemulsions.
- 1. What is the purpose of rinsing and disinfection in hemodialysis treatment?2- When and how often needs to do disinfection and rinsing? 3.Which process is first rinsing or disinfection?4- Did you experience any troubleshooting procedure during the lab? If yes, pleaseexplain how identified and solved the problem?Give the examples of emulsion prepare by these method:- 1)dry gum method 2)wet gum method 3)bottle gum methodPlease don't provide hand writtin sollution...



