Q: Define the structural relationship between the following molecules. CH3 он H он .Co,H H- OH VS Br OH...
A:
Q: In a glass bulb filled with gaseous neon two metal plates are used as electrodes separated by a dist...
A:
Q: 150 mL of polyethylene glycol has a density of 1.1333. What will be its weight?
A: Density is the amount of substance present per unit volume of solution. It can be expresses as- Den...
Q: Which of these compounds are oxoacids (also known as oxyacids)? HF hydrochloric acid chloric acid HC...
A:
Q: Calculate the molarity of 0.550 mol of Na, S in 1.30 L of solution. molarity: Calculate the molarity...
A:
Q: Draw suitable formulas for all of the position and configurational isomers possible) for the followi...
A: Isomers are compounds which have the same molecular formula but different physical and chemical prop...
Q: Analyze the following structure of bisabolene in terms of isoprene units joined by head-to-tail link...
A:
Q: In C6H12, how many constitutional isomers have atleast 1 3^o carbon? explain
A: Those carbon which is directly joint with three carbon called 3० carbon
Q: What is the concentration of hydroxide ion in a 0.160 M aqueous solution of hydroxylamine, ? What i...
A: Given: The concentration of the aqueous solution of hydroxylamine (NH2OH) is 0.160 M The Kb value is...
Q: 26 mL of oxygen gas at a temperature of 22 degrees Celsius is heated to 188 degrees Celsius. What is...
A:
Q: A student measures out exactly 0.101 g of salicylic acid and runs the experiment as dictated in the ...
A:
Q: what the three categories are. Are they talking about lone pairs, pi bonds, and sigma bonds?
A: Nucleophile is species that forms bonds with electrophiles by donating an electron pair.
Q: A sample of water of unknown mass undergoes a temperature change from 68.09oC to 72.61oC with a conc...
A: Given: Energy added = 205.8 J Initial temperature of water = 68.09 oC And final temperature of water...
Q: A 8.24 g sample of a compound contains 2.63 g of potassium, K, 2.38 g of chlorine, Cl, and oxygen, O...
A:
Q: Lonstants nodc Table Part A What is the balanced chemical equation for the reaction used to calculat...
A: Concept: Enthalpy of formation: The standard enthalpy of formation of a compound is equal to the cha...
Q: Write the balanced chemical equation for the reaction shown. = NO = NO, balanced equation:
A:
Q: Titanium metal requires light with a maximum wavelength of 286 nm to emit electrons. What is the min...
A: Given: The maximum wavelength of light required to emit electrons from Titanium metal is 286 nm.
Q: Spectrum 28 100% 75%- 50%- 25% 0% 3500 2500 1750 1250 750 4000 3000 2000 1500 1000 50 Wavenumber (cm...
A: Concept: Characteristics IR bond for Corbonyl: intensed bond near1715 cm-1 characteristic IR bond...
Q: What is the average atomic mass of titanium? Isotope Abundance Mass (u) 46Tİ 72.600% 45.95263 48Ti 1...
A: Isotopes are atoms of same atomic number but different mass number.
Q: A small volume within a 400 mL aqueous solution of methanol is shown in the image. Identify which im...
A: As we double the volume the molecules of methanol per unit area decrease as we have not added any...
Q: The Lewis structure of NH3 contains the following: 3 single bonds and 1 lone pair 2 double bonds 2 s...
A: To solve this problem we have draw the Lewis structure of NH3 and CH4 .
Q: Determine the number of water molecules necessary to balance the reduction half reaction of ___ MnO...
A:
Q: B. Nomenclature of Compounds: Study the examples given and fill up each table below. 1. Binary Acids...
A: Interpretation: We have to name binary acids.
Q: A 50.0 mL portion of a 1.60M solution is diluted to a total volume of 288 mL. A 144 mL portion of th...
A:
Q: The formation of a carbocation is the rate-determining step for an SN2 reaction. A. True B. ...
A: Given statement: The formation of a carbocation is the rate-determining step for an SN2 reaction. We...
Q: The activation energy for the gas phase decomposition of t-butyl chloride is 173 kJ. (CH3)3CCI–→(CH3...
A: The relationship between rate constant, temperature and activation energy is given by Arrhenius equa...
Q: use software to make 1H-NMR and 13C-NMR from the data in the paper and make a table with the follo...
A:
Q: 3. 4.50 mol of N2 gas (Cy. m = 20.6 J mol K) is enclosed in a piston-cylinder assembly (closed syste...
A:
Q: please draw the molecule and label it based on the data in the sheet and use the label in the data t...
A: Proton NMR: aromatic region : delta 6.5 to 8.0, Oxygen attached carbon having hydrogens at delta 3.5...
Q: 4. Calculate the work done under isothermal conditions for 5.5 mols of an ideal gas expanding from 1...
A:
Q: Vhich of these foods would be most like the 'plum-pudding-model of the atom? A chocolate chip muffin...
A: Answer - The correct option is (a) A chcolate chips muffin It resembles a plum pudding because the ...
Q: How many pi bonds are in C6H10NCI-- which has one ring--- (explain)
A: Given is, Molecular formula- C6H10NCI We can find out how many pi bond present in molecule by findin...
Q: Choose the bond below that is least polar. O P-F OC-Br OC-F O C-CI
A:
Q: A 19.89 gram sample of an organic compound containing C, H and O is analyzed by combustion analysis ...
A: We have to predict the empirical and molecular formula.
Q: Draw structure for butyl butanoate
A: In this question we have to draw structure of butyl butanoate. It is an organic compound that is an ...
Q: Question 22 of 27 A student needs to prepare 250 mL of a 0.850 M aqueous solution of sucrose, C,,H„0...
A: The volumetric flask should be used that is C.
Q: Does a precipitate form when A and B empirical formula of solution A solution B precipitate are mixe...
A: The data given is,
Q: Formula Valence Diagram Drawn Total Electronic Molecular Hadridiz-Polar (yor N) Electrons Bonding Do...
A:
Q: 4.62mL of .1510mL NaOH was needed to neutraliz 50.mL of H2SO4 solution. What is the concentration o...
A: In the given question we have to calculate the concentration of original sulphuric acid solution. th...
Q: (1R,2R)-1-Bromo-2-methylcyclopentane is reacted with sodium methoxide. Given the product(s) and show...
A: An elimination reaction in organic chemistry is a reaction where a base abstract An acidic proton wh...
Q: Quntion of The typical range of observed peaks in a "C NMR spectrum is A08 pom BIO-14 pom C) 0-160 p...
A: Range of 13C NMR depends on the different scale .
Q: NaI (aq) + Cl2 (g) → I2 (s) + NaCl (aq) Given this equation calculate how much NaI is required to o...
A: Consider the given information is as follows; Mass of I2 produced = 3.44 kg = 3440 g Mass of NaI req...
Q: Question 15 Which one of the following species has as many electrons as it has neutrons? o 'H (Z=1) ...
A: number of electrons in a given element = Z number of neutrons = atomic mass - atomic number(Z)
Q: Using the noble-gas shorthand notation, write the ground state electron configurations for the fol- ...
A:
Q: Place the following in order of decreasing dipole moment. |- CH4 II - CH3OH III - CH3SH O III > I > ...
A: Dipole moment can given by the difference of electronegativity of atoms Higher the difference of el...
Q: What is the hybridization at the bond being pointed to?
A:
Q: a liquid in the lab has a density of 1.2 g/cm^3 what is the mass in grams of 254ml of the liquid
A:
Q: How many moles are in a 725.0 mL sample of gas at 299 K and 0.800 atm? R=0.08206 L atm / mol K
A: Given Volume of gas ( V ) = 725 mL = 725 × 10-3 Liter (...
Q: What is the molarity of a solution of nitric acid if 0.216 g of barium hydroxide is required to neut...
A: Balanced reaction is: 2HNO3(aq) + Ba(OH)2(aq) ....> Ba(NO3)2(aq) + 2H2O(l) Mass of Ba(OH)2 = 0....
Q: Anucleus that is shielded will appear in the NMR spectrum A) downfield B) upfeld
A: Shielded nucleus means electromagnetic field applied is not experienced as much by nucleus as it is ...
Whay are the products. Explain
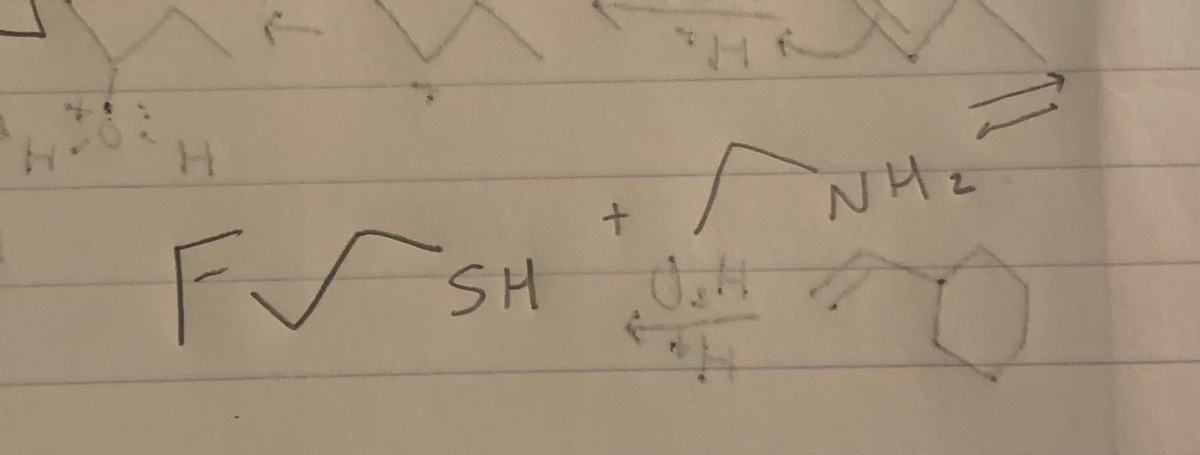
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- A student had a sample of CaCl2 • nH2O. He heated the sample until the mass did not change. From this data, calculate (a). the percentage of CaCl2 in the original sample and (b). the formula of the hydrate Mass of crucible + cover = 28.39 gMass of crucible + cover + sample = 30.41 gMass of crucible + cover + residue = 29.75Alchemists produced elemental mercury by roasting cinnabar ore in air: HgS(s)+O2(g)→Hg(l)+SO2(g) The volatility and toxicity of mercury make this a hazardous procedure, which likely shortened the life span of many alchemists. Given 100 g of cinnabar, how much elemental mercury can be produced from this reaction?Find the limiting reactant ____ Li3N + ____ NH4NO3 -----> ___ LiNO3 + ___ (NH4)3N
- Take Hg(l) + Br2(aq) <---> 2Br^-(aq) + Hg2^+(aq) If 4.20 grams of Hg are consumed, how many grams of Br- ions are there?In one sentence explain what is wrong with them, and rewrite them so that the coefficients are correct. 2 Mg(OH)2(s)+4HCl(aq)->2MgCl2(aq)+4H2O(l)In following reaction, NH2 is relaced by Br H Cu NO2

