Q: Which halide is least reactive towards SN2 reactions ? O a. tert-butyl bromide O b. sec-butyl bromid...
A: Sn2 reaction are bimolecular nucleophilic substitution reaction. This is single step reaction , and...
Q: Ethylammonium chloride, CH;CH;NH3CI, is an acidic salt. (i) Write equations to show the dissolving o...
A: fullscreen
Q: Balance the equation ? N₂H₄ + ? N₂O₄ → ? N₂ + ? H₂O How many moles of N₂ will be produced for every ...
A: First, we balanced the chemical equation and then with the help of the balanced chemical equation st...
Q: What are the properties of matter influenced by intermolecular forces?
A: We'll answer the first question since the exact one wasn't specified. Please submit question specify...
Q: Can you help me with this?
A: Acid dissociates into its conjugate base.
Q: what are the pros and cons of lignin process for producing vanillin?
A: Answer - Lignin process - Lignin is an important organic polymer which is abundant in cell walls of ...
Q: 1 mole of N2 gas in a volume of 300K and 2L undergoes an isothermal reversible expansion up to 20L f...
A: a. The work done by ideal gas in isothermal reversible expansion, Wrev =- nRTln(V2/V1) = -(1*8.314*3...
Q: The vapour pressure of pure CCI4 at 65 °C is 504 mmHg .How many grams of glucose (C6H12O6) must be a...
A: The vapour pressure of a solution can be determined as below : Psolution = Xsolvent. Psolvent Where...
Q: 2.What is the molarity if a 900. mL solution of HCl was made using 1.45 g of HCl? Numerical answer: ...
A:
Q: Draw the structure that would result from the reactions
A:
Q: Please I need help with question
A: Note: According to our guidelines we are supposed to answer only first three subpart
Q: How many moles of NaOH are present in 21.6 mL of 1.00 M NaOH?
A:
Q: Conversion of Compound into complete structure. A.CH3(CH2)8CH3 B. CH3(CH2)4OH C. CH3CCl3 D. CH3(CH2)...
A: The given compounds has to be converted into its complete structure.
Q: Consider the reaction A B with AH° = 25.39 kJ/molxn: What should be the AH° in kJ/molxn for the reac...
A: Given reaction is A -----> B : enthalpy of reaction is = 25.39 Kj/mole means fo...
Q: Harriet Rowki prepared 100.0 mL of 2.50 M H3BO3-NaH2BO3 buffer solution (pH = 8.50). The pKa value o...
A: A buffer solution is an aqueous solution consisting of a mixture of a weak acid and its conjugate ba...
Q: An aqueous solution containing 8.19 g of lead(II) phosphate is added to an aqueous solution containi...
A: The answer to the following question is-
Q: What is the limiting reactant when 10.0 g of carbon monoxide (CO, molar mass = 28.01 g∙mol⁻¹) and 15...
A: CO = 10.0 g Fe2O3 = 15.0 g
Q: How many grams of gallium chloride would be produced from 177 g hydrochloric acid? 2Ga + 6HCl ⟶ 2GaC...
A: The given reaction is, 2Ga + 6HCl ⟶ 2GaCl3 + 3H2 To find, the amount of Ga formed from 177 g hydroch...
Q: A 500 g sample of aluminum sulfate is reacted with 450 grams of calcium hydroxide. A total of 596 ...
A: The limiting reagent is the reactant which is consumed completely during the reaction and thus stops...
Q: Upon reaction of 1.27 g of copper sulfate with excess zinc metal, 0.377 g copper metal was obtained ...
A: No. of moles of the product will form is depends upon mole ratio used in the Molecular reaction. b...
Q: [Ni(en)3]SO4 ,Calculate the KAYE(Crystal field cleavage energy)
A: For an octahedral complex, the five fold degenerate d-orbital is splitted into two different sets of...
Q: The black soot is given off when certain hydrocarbons are burned because __ the fuel is undergoin...
A: Dear student I have given answer to your question in the image format.
Q: Suppose that the molar enthalpy of a reaction is 56.94 kJ/mol and 16.89 kJ of heat are absorbed by t...
A: The amount of heat required to raise the temperature of the sample is given by: q = mc∆T 'q' is the ...
Q: As a general rule, most hydrocarbons are _____ than water. more dense less dense there is no g...
A:
Q: The solubility of CO2 in water at 25°C and 1 atm is 0.034 mol/L. What.. is its solubility at a parti...
A:
Q: 11. Pb(NO), # of molecules: # of elements: Name of element: # of atoms Total # of atoms: The #3 is a...
A:
Q: 0.55 g of a compound X is dissolved in 300 cm3 of an aqueous solution. Given that compound X is comp...
A: Given data: The mass of the compound X is m = 0.55 g. Temperaure T = 25°C = (25+273) K = 298 K. The ...
Q: A compound A, C7H12, was found to be optically active. On catalytic reduction over platinum catalyst...
A: Alkynes are organic compounds containing carbon and hydrogen atoms. In alkynes, there is a presence...
Q: What is the mass percent of an ammonium carbonate solution prepared by dissolving 33.2 grams of soli...
A: The answer to the following question is-
Q: 3.How many grams of LiOH would be used to make a 1.50 M solution that hsa a volume of 1.00 L? Numeri...
A:
Q: Need help
A: The answer to the following question is-
Q: Ethanamine, CH;CH;NH2, is a weak base. pK«(CH;CH;NH3') = 10.6 (a) (i) Write an equation to show the ...
A: Ethanamine: CH3-CH2-NH2 = weak base. pKa = CH3-CH2-NH3+ = 10.6 a) 1) Write an equation to show the r...
Q: Complete and balance the following chemical equation for theComplete combustion of Hexane: (List pro...
A: The complete combustion equation of hexane has to be given.
Q: Write the empirical formula for at least four ionic compounds that could be formed from the followin...
A:
Q: What are the uses and properties of Astatine and Tennessine?
A: Uses and properties of Astatine and Tennessine has to be given.
Q: Suppose that the molar enthalpy of a reaction is -69.06 kJ/mol and 1.134 moles are reacted. How many...
A: The molar enthalpy of a reaction is the change in enthalpy of a reaction for one mol of the reactant...
Q: What volume will 5.10 g of sulfuryl fluoride, SO₂F₂, occupy at STP? Atomic Molar Masses F 19.0 g/mol...
A:
Q: Biotin is being studied in a lab. a. What is the IMF(s) of biotin (C10H16N2O3S)? b. What are the sig...
A: The given molecule is biotin. a. What is the IMF(s) of biotin (C10H16N2O3S)? b. What are the sigma/p...
Q: Dichlorine monoxide, Cl,0, is sometimes used as a powerful chlorinating agent in research. It can be...
A: The given reaction is as follows: The theoretical yield of the reaction = 8.08 g The actual yield o...
Q: Consider this reaction: N2 (s) + 2 O2 (g)→ 2NO2 (g) AS° = -121.77 J/K.mol; AH° = 66.36 kJ/mol which ...
A: The given data's are, ΔSo = -121.77 J/k.mol, ΔHo = 66.36 kJ/mol To find the true statement?
Q: Calculate the ∆G⁰reak value for the following reaction running at 298K.CO (g) + ½ O2 (g) ↔ CO2 (g)As...
A: Gibb's free energy of the system is a thermodynamic property which is a state function. The change i...
Q: Indicate whether each of the following compounds is ionic (I) or covalent (C). PCl3 ...
A: Indicate whether each of the following compounds is ionic (I) or covalent (C).
Q: Do a little research and explain why toluene is used in school labs rather than benzene. Use your o...
A: ANSWER IS
Q: Suppose that extra volume (more than the actual volume) of 0.2 M NaOH solution was used to reach the...
A: fullscreen
Q: Write the balanced chemical equation for the combustion of methane, CH,(g), to give carbon dioxide a...
A:
Q: Need help with this question Question 8
A: Chemical reaction is just a rearrangement of atoms. It can neither create nor destroyed during the c...
Q: Consider the reaction A B with AH° = -42.46 kJ/molrxn: %3D What should be the AH° in kJ/mol,m for th...
A: The given reaction is as follows: ........(1) To calculate: ...... (2)
Q: A piece of metal is heated to a temperature of 50.0°C and then placed in a calorimeter containing 50...
A: To solve this problem we will use the formula Q = m • c • ∆T Where , Q = heat released or abso...
Q: Need help
A: Different fuels are used in automobiles and they release different energy on burning. What are the c...
Q: Calculate the pKa of a acid at 25°C if its conjugate base has a pKb = 1.19
A: The relation between pKa and pKb is given as: pKa + pKb = pKw ..........(1) Also, pKa = -logKa pKb =...
Can you please help with this?
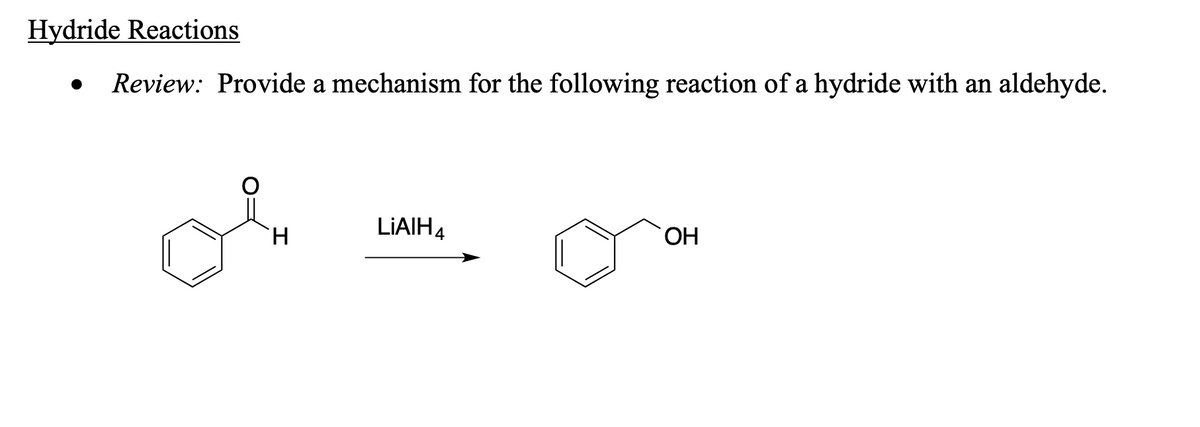
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Write a mechanism for the reaction below. Hint: think retro!Give a step-by-step mechanism for the following substitution reactions:Boron tribromide (BBr3) cleaves ethers to give alkyl halides and alcohols.R¬O¬R′ + BBr3 ¡ R¬O¬BBr2 + R′BrR¬O¬BBr2 + 3 H2O ¡ ROH + B(OH)3 + 2 HBrThe reaction is thought to involve attack by a bromide ion on the Lewis acid–base adductof the ether with BBr3 (a strong Lewis acid). Propose a mechanism for the reaction ofbutyl methyl ether with BBr3 to give (after hydrolysis) butan-1-ol and bromomethane.
- (a) Propose a plausible mechanism for the reaction given below: (b) Show a plausible step by step synthesis scheme for each of the following transformations:Provide a structure for the major products of the reactions and include the steps for each reaction.Propose a mechanism for the following reactions. Use an appropriate organic chemistry software (e.g. chemsketch, chemdraw, etc.) to draw the structures and mechanism. Explain it.
- Propose a mechanism for the reaction in Question2.1.3.Enamines formed from the cyclic secondary amine pyrrolidine are important intermediates in the synthesis of 1,5-diketones. (1st pic) On the structures provided below, draw arrows showing electron flow for the reaction mechanism for the acetic acid-catalyzed formation of an enamine from cyclohexanone and pyrrolidine. (2nd pic)Propose a detailed mechanism for the reaction below (in the attached picture), showing the structure of thestable intermediate and using curved arrows to indicate electron flow in each step



