Q: The combustion of ammonia is : 4NH3(g)+5O2(g)--> 4NO(g)+6H2O(g) DeltaH= -905kJ what is the...
A: Given Heat of formation data: NO(g) = 90.4 kJ/ mol H2O(g) = -241 kJ/mol 4NH3(g)+5O2(g)--> 4NO(g...
Q: B. What is the pH of the solution containing 0.20 M NH3 and 0.15 M NHẠCI? Section Break(Next Page) -
A:
Q: if the pOH of a solution is 5.4, the pH is? the answer is 8.6. Can somone please walk me through i...
A: Answer: pH of the solution is 8.6
Q: Indicate the number of electrons lost or gained when each of the following atoms forms an ion. a. 37...
A: Explanation to the correct answers are given below:
Q:
A: Given: Mass = 5 kg Force = 20 N
Q: At 22 °C, an excess amount of a generic metal hydroxide, M(OH),, is mixed with pure water. The resul...
A: We know that, pH can be expressed as negative logarithm of H+. i.e., pH = - log...
Q: The dimensions of aluminum foil in a box for sale in supermarkets are 662/3 yards by 12 inches. The ...
A: Given The dimensions of aluminum foil in a box for sale in supermarkets are 66.66 yards by 12 inche...
Q: What is the density of an object that raises the level of water in a graduated cylinder from 15.00 m...
A:
Q: Over the years, the thermite reaction has been used for welding railroad rails, in incendiary bombs,...
A: Given: Thermite reaction: Fe2O3 ( s ) + 2 Al ( s) → 2 Fe ( l ) + Al2O3 ( s)
Q: Using the concept of trans activating ligands, predict the following reaction products (A, B, C, D, ...
A:
Q: Write balanced equations that describe the following reactions,a.the dissociation of perchloric acid...
A: a. The dissociation of perchloric acid in water = The balanced equation for the given reaction is, H...
Q: At a certain temperature, the equilibrium constant for the chemical reaction shown is 5.26 x 10-3. A...
A: Equilibrium constant =5.26×10^-3 [AB]=2.825M [BC]=1.425M [AC]=0.160M [B]=?
Q: The effusion rate of an unknown gas is measured and found to be 31.50 mL/min. Under identical experi...
A: Given : Rate of effusion of unknown gas = 31.50 mL/min. And rate of effusion of O2 gas = 30.50 mL/mi...
Q: In Einstein's photoelectric experiment, the kinetic energy (K.E.) of an electron displaced from a me...
A:
Q: Adentify the expected major organic product generated from the reaction sequence shown. 1. OsO, 2. N...
A:
Q: Select the expected major product(s) of the following reaction. HBr Br Br Br A and II I and III
A:
Q: Which of the following bonds is most polar: S ¬ Cl, S ¬ Br, Se ¬ Cl, or Se ¬ Br?
A:
Q: Classify each of the following alcohols as a primary, secondary, or tertiary alcohol.
A: Hi, since you have posted multiple subparts questions. We will answer the first three subparts for y...
Q: Enough of a monoprotic acid is dissolved in water to produce a 1.16 M solution. The pH of the result...
A: Given, the concentration of the monoprotic acid = 1.16 M pH of the solution of monoprotic acid = 2.6...
Q: The equilibrium constant for the reaction 2CrO42- (aq) + 2H+ (aq) ⇌ Cr2O72- (aq) + H2O is 3×1014. Wh...
A: Given: 2CrO42- (aq) + 2H+ (aq) ⇌ Cr2O72- (aq) + H2O K = 3×1014
Q: Pre-lab question #6-1: Calculate the percent (%) C2042- in H2C204. 53.0 57.1 65.7 97.8
A: Mass percent: Mass percent of ion = atomic mass of the ionMolar mass of the compound×100
Q: Which of the following compounds are chiral? Draw them, and label the chirality centers. (a) 2, 4-Di...
A: The chiral compounds have to be identified and their structures have to be drawn and the chiral cent...
Q: Determine the maximum number of electrons that can be found in each of the following subshells: 3s, ...
A: The maximum number of electrons depends on the angular quantum number (l) and not on the principal q...
Q: In the beginning of the twentieth century, some scientists thought that a nucleus may contain both ...
A:
Q: The decomposition of nitrosyl chloride was studied: a. What is the rate law? b. Calculate the value ...
A: Given: Decomposition of nitrosyl chloride: 2NOCl(g) <----> 2NO(g) + Cl2(g)
Q: 3)can someone explain the question
A: Given: Initial temperature (T1) = 400 K Final temperature (T2) = 450 K Initial rate constant (K1)...
Q: a. Which is greater, the initial pressure of helium or the initial pressure of neon? How much greate...
A: For a:
Q: Draw the structural formula of the monomer(s) from which each of the following polymers was made.
A: The question is based on the concept of polymerization. we have to write the monomers of the followi...
Q: An electron in a certain atom is in the n = 2 quantum level. List the possible values of ℓ and mℓ th...
A: An electron in a certain atom is in the n= 2 quantum level and in n=2 quantum level 2s and 2p subsh...
Q: energy content when eaten food cal kcal kJ 4 a raw carrot 3.00 x 10 30 125.52 a tablespoon of corn o...
A: The question is based on the concept of unit conversions. We have to convert the values in appropria...
Q: A mixture of 0.452 M H,0,0.448 M CI,0, and 0.737 M HCIO are enclosed in a vessel at 25 °C. H,O(g) + ...
A: Given equation :
Q: Is it possible for a fluorescent material to emit radiation in the ultraviolet region after absorbin...
A: Given : Is it possible for a fluorescent material to emit radiation in the ultraviolet region after ...
Q: a) Explain the fundamentals of gas chromatographyb) Explain the fundamentals of mass spectrometry
A: 1) Components in the mixture are distributed between two phases, one of which is a stationary phase,...
Q: What is the number of moles in a sample that originally occupied 300 mL with 1.50 moles and then occ...
A:
Q: Calculate the heat released when 2.00 L of Cl2 (g) with a density of 1.88 g/L reacts with an excess ...
A: Given: Volume of chlorine = 2.00L Density = 1.88g/L Temperature = 250C Pressure = 1 atm To find: The...
Q: The first line of the Balmer series occurs at a wavelength of 656.3 nm. What is the energy differenc...
A: Answer:- This question is answered by using the simple concept of calculation of energy change durin...
Q: mL In the titration of 46.0 mL of 0.400 M HCOOH with 0.150 M 1 3 LIOH, how many mL of LIOH are requi...
A: To achieve half way equivalence point, half of the number of mol is required. At equivalence poin...
Q: 4) can someone explain the question
A: A question based on dissociation kinetics of HBr, which is to be accomplished.
Q: 12. What is the main factor in not observing the glass transition behavior in highly cross-linked an...
A: Glass transition behavior in highly cross-linked and highly crystalline polymers.
Q: In the given reactions is the starting material being oxidized, reduced or is there no change in the...
A:
Q: Calculate the pH and [S2−] in a 0.10-M H2S solution. Assume ka1= 1.0 × 10−7; ka2= 1.0 × 10−19.
A: Answer:- This question is answered by using the simple concept of calculation of pH of a weak acid u...
Q: An iron ore sample contains Fe2O3 plus other impurities. A 752-g sample of impure iron ore is heated...
A: Answer:- This question is answered by using the simple concept of stoichiometry which involves the r...
Q: Arrange the following substances in order of increasing mass percent of carbon. a. caffeine. C8H10N4...
A:
Q: Describe how you would prepare 2.00 L of each of the following solutions.a. 0.250 M NaOH from solid ...
A: Note: “Since you have posted a question with multiple sub-parts, we will solve the first three sub-p...
Q: Write the chemical reaction for chlorous acid in water, whose equilibrium constant is Ka. Include th...
A: equilibrium constant is defined as products concentration divided by reactent concentration to the p...
Q: Given: Volume of sample = 50.00 mL NaOH (mL) used = 33.20 mL Molarity of NaOH = 0.09693 M Molar mass...
A: Answer:- firstly from the chemical reaction of acetic acid and NaOH, molarity of acetic acid solutio...
Q: An ideal gas is contained in a cylinder with a volume of 5.0 x 102 mL at a temperature of 30.°C and ...
A: Answer:- This question is answered by using the simple concept of ideal gas equation which involves ...
Q: Use the Aufbau principle to obtain the ground-state electron configuration of technetium.
A: Aufbau principle states that while filling the electrons, the lower energy orbital are filled before...
Q: Shown below is a carbocation intermediate in an electrophilic addition reaction of HCl with an alken...
A: It is the dire sigma bonding pair interaction of C-H bond with vacant p orbital of carbocation. Here...
Q: 2) Can some one explain the question
A: We have given that A + 2B -----> 3C + D Rate constant : The rate constant is the proportion...
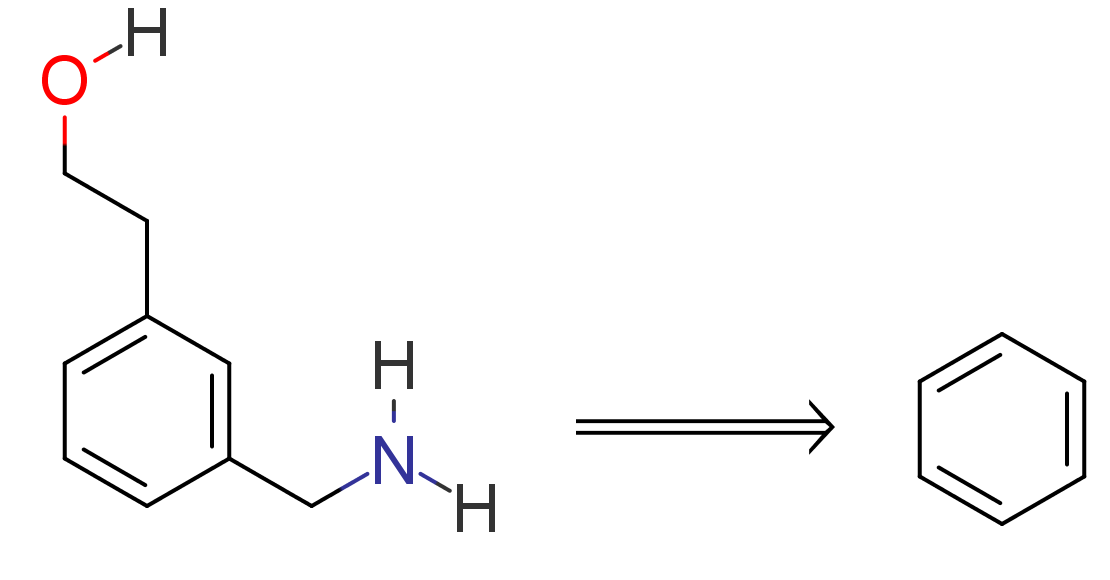
Propose a synthesis route to prepare 2‐[3‐(aminomethyl)phenyl]ethan‐1‐ol starting from benzene, without Stille reaction:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images


