Q: a sample of gas has a volume of 7.00 L and a pressure of 2.750 kPa when its temperature is 45.0 C if…
A: P1 = 2.750 kPa V1 = 7.00 L T1 = 45.0 C + 273.15 = 318.15 K P2 = 1500 kPa V2 = 14.00 L
Q: я NaBH4 OCH₂CH3 CH3CH₂OH
A: NaBH4 is reduces only ketone not ester.
Q: Provide the correct IUPAC name for the compound shown here. Reset & para-meta- ortho- 2- 1,3- 2,6-…
A: According to the IUPAC nomenclature, First longest chain of carbon atom is identified. Numbering is…
Q: Atram AS rxn AG 5. Use the and/calculated in question 2 as well as the equation below to calculateat…
A: The sign of ∆G0rxn defines the spontaneity of reaction. If the sign of ∆G0rxn <0 , then it is…
Q: What mass in grams of potassium fluoride is needed to make 150.0 mL of a 0.450 M hydrofluoric…
A: Buffer solutions: When a tiny amount of acid or base is added to a solution, buffer solutions resist…
Q: What would the starting reactant be? How would you find it if you know what the product is?
A: A reaction scheme is shown below We have to identify the starting material.
Q: How many miciolters of1.000MNaOHsolution must be added to25.00 mLof a0.1000Msolition of lactic…
A:
Q: A chemistry graduate student is studying the rate of this reaction: 2H, PO4 (aq) → P₂O, (aq) + 3H₂O…
A: Answer: This question will be solved by using trial and error method where we will check in which…
Q: A closed-ended manometer is filled with gas. The ∆h of the column of Hg in the manometer is 3.56 cm.…
A: To determine the pressure of the gas in the manometer, we need to use the hydrostatic pressure…
Q: What are the products of the following reactions: a) ● b) c) 1. AICI3 2. H₂O 1. AICI3 2. H₂O 1.…
A: In Friedel Crafts reactions, the lewis acid ( example ; AlCl3) is used to generate electrophile.
Q: What is the pH of a 0.0380 M solution of HONH CI (Kb of HONH₂ is 1.1 x 10°)?
A: The pH of any acidic mixture is always low.
Q: 7. Which halide will react most rapidly via E2? a. Me Me Br b. Me Me Br
A: Since you have posted multiple question, we can solve only first question for you. If you want other…
Q: if you had a 0.85 M weak acid solution and wanted to make a buffer, what should you add to the…
A: To make a buffer, you need to add a weak acid and its conjugate base in roughly equal amounts. The…
Q: What is the common name for the following acid?
A:
Q: At a certain temperature lead(II) fluoride has a K = 3.96x107. What is the solubility of lead(II)…
A: “Since you have posted multiple questions, we will provide the solution only to the first question…
Q: Draw the reactants required to make each Aldol or Claisen product. عه OH
A: Aldol reaction: The reaction in which an enolate ion reacts with aldehyde or ketone to form a…
Q: A chemistry graduate student is studying the rate of this reaction: 2SO₂ (g) -2SO₂ (g) + O₂(g) She…
A: In the given question, the data for a reaction is given. We have to find the rate law and rate…
Q: Specify the number of tertiary carbons in the molecule below. l 04 01 02 3
A: A tertiary carbon atom is a carbon atom which bound to three other carbon atoms.tertiary carbon…
Q: 3. Which ketone has the largest dipole moment? Explain.
A: If the species formed is aromatic, the it would remain as the separated charged molecule with…
Q: The decomposition of a generic diatomic element in its standard state is represented by the equation…
A: Solution- At 2000K ∆G0f =4.43 kJ/mol = 4430 kJ/molWe know, ∆G0f =– RT In Kor, In K=∆G0f-RThere, In…
Q: 1. which of the following is true regarding an sn1 reaction?…
A: An SN1 reaction involves a unimolecular substitution of a leaving group with a nucleophile. The…
Q: Quantity 1. Mass of crucible + cover 2. Mass of crucible + cover. Magnesium 3. Mass of Magnesium 4.…
A: Empirical formula: The formula in which the simplest whole number ratio of constituent atoms ( that…
Q: In your initial post to the discussion, include the following parts: Write out two real-life…
A: How to write own scenarios for reaction..
Q: 85.0 mL of 0.375 M HNO3 is titrated by 0.750 M KOH. Calculate the pH of the acid solution before…
A: pH of the solution for the given titration Given, molarity of HNO3 = 0.375 M volume of HNO3 =…
Q: al formula corresponding to each of the fa alcohol p-Diisopropylbenzen
A: Following names of organic compounds given - o-Chlorobenzyl alcohol p-diisopropylbenzene…
Q: When (1-bromoethyl)cyclopentane is heated in water for an extended period of time, SIX products…
A: When (1-bromoethyl)cyclopentane is heated in water for an extended period of time, SIX products…
Q: Calculate the density of 50.0 g of an isopropyl alcohol-water mixture (commercial rubbing alcohol)…
A: Mass of isopropyl alcohol-water mixture = 50.0 g Volume = 63.6 mL Density = ?
Q: 9. "Heavy water," deuterium oxide (D₂O), is sometimes used in nuclear reactors to slow or moderate…
A: Dissociation of Heavy water : D2O(I) ⇌D+(aq) + OD-(aq) Kw= 8.913 ×10-16 at 20°C Lets assume that…
Q: A chemistry graduate student is studying the rate of this reaction: 2H₂PO₂ (aq)-P₂O, (aq) + 3H₂0…
A: In the given, data for the reaction is given and we have to find the rate law and rate constant…
Q: 2. Calculate AH AG and AS for the following reaction given thermodynamic data below. Pay attention…
A: The sign of ∆G0rxn defines the spontaneity of reaction. If the sign of ∆G0rxn <0 , then it is…
Q: The ion [RuCl(NO)2(PPh3)2]+ has N — O stretching bands at 1687 and 1845 cm-1. The C — O stretching…
A: The N-O stretching bands in [RuCl(NO)2(PPh3)2]+ occur at 1687 and 1845 cm-1, which are higher in…
Q: Use the reduction potentials and the unbalanced chemical equation below to answer the following…
A: In chemistry, electrode potential is a measure of the energy required to transfer electrons from a…
Q: Consider the molecule given below. How many products are possible from Free Radical Chlorination of…
A: Allylic radicals are the stable radicals formed in step 1.
Q: 7. Give a Mechanism for the following reaction; show formal charges and significant intermediates.…
A:
Q: 2.) A newly designed superconductor material has 3 different types of atoms in its molecular formula…
A: As A resides in the corners of the cube. so there are 8 vertices in the cube and there are 8A in 8…
Q: function represents the minimum amount of energy to observe ejected electrons via the photoelectric…
A: Given, Titanium has a work function = 6.94 x 10-19 J. The maximum kinetic energy of ejected…
Q: Calculate the pH of a 0.0428 M sodium acetate solution. Ka (HC2H3O2) = 1.8 × 10−5. Report your…
A: pH calculation of Salt:- Salt of weak acid (WA) and a strong base (SB) Consider, weak acid…
Q: H3C -CH3 CH3 CH3 excess Na2Cr2O7 H₂SO4 COOH A COOH H3C COOH -CH3 CH3 В COOH с COOH
A:
Q: - Consider the reaction below to answer the following questions: S Write the complete stepwise…
A: The nucleophile and electrophile are present on the same molecule and it involes in the formation of…
Q: 17. The following compound can be prepared by a mixed aldol condensation reaction. Give the…
A:
Q: Which of the following would make a good buffer system. Group of answer choices HI/BaI2 HNO3/NaOH…
A: A buffer system is a solution that can resist pH changes when small amounts of an acid or base are…
Q: A chemistry graduate student is studying the rate of this reaction: 2NH3 (g) N₂(g) + 3H₂(g) She…
A: In the given question, a data of reaction is given and this relates to chemical kinetics. We have…
Q: E2 elimination takes place under the same conditions as SN2 substitution and the reactions compete…
A: When E2 elimination takes place under the same conditions as SN2 substitution, the reactions compete…
Q: Predict the reactants of this chemical reaction. That is, fill in the left side of the chemical…
A: Answer:- This question is answered by using the simple concept of writing the balanced molecular…
Q: A solution in which the pOH is 12.6 would be described as very acidic slightly acidic neutral
A: As we know ph scale
Q: Calculate the volume (mL) of 0.100 M H3PO4 required to neutralize 25.0 mL of 0.010 M Ca(OH)2.
A: For H3PO4 solution, Concentration of H3PO4 solution = 0.100 M = 0.100 mol/L Volume of H3PO4 solution…
Q: 2.48×105 Pa Express your answer using three significant figures.
A: Pressure = 2.48×105 Pa 2.48×10^5 Pa expressed using three significant figures is 2.48 x 10^5 Pa.
Q: The concentration-based equilibrium constant for the reaction shown below is Kc = 1.37 at 137 ℃.…
A:
Q: Grignard reagent is a nucleophile. Which electrons does it give up in the reaction? a) Electrons in…
A: Grignard reagent is alkyl magnesium halide ( i.e RMgX). Let's R= CH3 and X = Cl So, the reagent is…
Q: 29. Calculate the minimum concentration of Mg2+ that must be added to 0.10 M NaF in order to…
A:
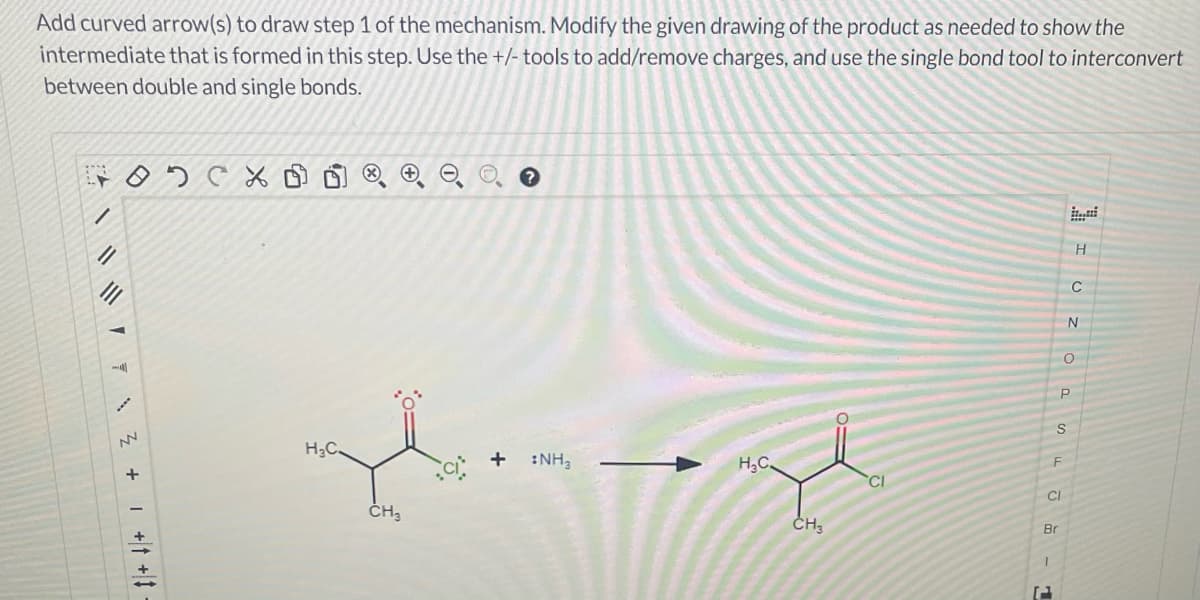
Step by step
Solved in 3 steps with 3 images

- Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- ΔH=-75 kJ/mol Rate = k [CH3CH2Br][CH3COO-] Which reaction energy profile would be the best representative of the data provided?Please give major organic products along with mechanism. Please also identify what type of reaction it is (SN1, SN2, E1, E2)Below is the equation for a nucleophilic substitution reaction and some experimental data. CH3CH2Br + CH3COO- ⇌ CH3CH2CO2CH3 + Br- Rate = k [CH3CH2Br][CH3COO-] Which mechanism would best fit the data?
- Please show me the mechanism for O3+2-methyl-2-butene, stoping at the ozonide?Fill in the boxes with reagents or products and show the mechanism.-Name the correct mechanism via arrow pushing -name each mechanism -name each minor and major products (1R,2S)-1-iodo-2-methylcyclohexane + ethanol/DMSO/stirred 3 days
- Identify which substitution mechanism best fits the following statement: The reaction proceeds through a concerted mechanism. A) SN1 B) SN2Please help with the following ochem mechanisms.... 1. Provide the stepwise mechanisms for the following reactions (see attached picture)Circle the molecule tht would form in the following reaction and show the mechanism

