Q: THERMODYNAMICS: Given the following hypothetical thermochemical equations: 2QJ3 (g) + 3Q2X (g) → 4Q...
A:
Q: THERMODYNAMICS: Use the given standard enthalpies of formation to calculate the AH° (in kJ) for the ...
A:
Q: ELECTROCHEMISTRY: Calculate the required voltage (V) if an unknown metal (M) was electrodeposited fr...
A: Let voltage required is V volt . And we are given 91%effeicient current = 6.82 ,so nett effective cu...
Q: THERMODYNAMICS: Use the given standard enthalpies of formation to calculate the AH° (in kJ) for the ...
A:
Q: QUESTION 8 Which IR frequency range can differentiate a carboxylic acid from an alcohol? 3200 cm-1 1...
A: The IR frequency range of carboxycic acid (-COOH) = 2500 - 3200 cm-1 the IR frequency of alcohol ...
Q: THERMODYNAMICS: Given the following hypothetical thermochemical equations: 2QJ3 (g) + 3Q2X (g) → 4Q2...
A:
Q: Consider the nuclei 11750Sn. Determine its mass defect in kilograms if its atomic mass is 116.928 a...
A:
Q: THERMODYNAMICS: The combustion of 1.00 mol of glucose (C6H1206) liberates 2820 kJ of heat. If 1.14 g...
A: We will first calculate moles of glucose, then calculate hear capacity of calorimeter.
Q: a. Hydrogen cyanide is used in the manufacture of clear plastics such as Lucite and Plexiglas. It is...
A: A reaction is said to be endothermic reaction if ,to carry out the reaction, heat is required Where...
Q: ELECTROCHEMISTRY: Calculate the required voltage (V) if an unknown metal (M) was electrodeposited fr...
A: Answer: If current efficiency is 96% then effective current passed I=6.69×96100AI=6.69×0.96AI=6.4224...
Q: Use the given standard enthalpies of formation to calculate the ΔHo (in kJ) for the hypothetical rea...
A:
Q: Which of the following will produce ammonia upon hydrolysis? `NH2 H
A:
Q: ELECTROCHEMISTRY: Calculate the required voltage (V) if an unknown metal (M) was electrodeposited fr...
A: Given: Current = 5.05 A Time passed = 12 hour Power = 0.1734 KWh. Current efficiency = 86 %
Q: 3. o Q CH3CO CCH3 + НОН
A: Hydrolysis of anhydrides gives carboxylic acids.
Q: he strength of Lewis bases follows the order of... A. NH3 > Me2NH > Me3N B. Me3N > Me2NH > MeNH2 C. ...
A: Given : We have to arrange in the correct Lewis order.
Q: Question 4 THERMODYNAMICS: Calculate the value of AH (in kJ) for the reaction, 2AR3 (g) + Q (s) → A2...
A: The given reaction is, 2AR3(g) + Q(s) ---> A2R4(g) + QR2(s) The reactions involved to give the ...
Q: Which of the following will give an N-nitroso-N,N-dimethyl amine? „NH2
A: This is the question of Diazotization reaction.
Q: A chemist adds 900.0 mL of a 3.98 g/dL barium chloride (BaCi,) solution to a flask. Calculate the ma...
A: To solve this problem we will calculate the mass of BaCl2
Q: ELECTROCHEMISTRY: Copper was electroplated from a CuzSO4 solution onto an electrode with an active s...
A: The electroplating thickness formula = gm Deposited =( it/96500) × (FM/n) i=current in amp t=time ...
Q: Give the chemical name of the products A and B in the following sequence of reactions: CH;CH,OH CH;C...
A:
Q: ELECTROCHEMISTRY: Copper was electroplated from a Cu2SO, solution onto an electrode with an active s...
A: Given: Cu2SO4 is being used for electroplating Area = 3.11cm2 Current = 4.45A Time = 1.2715h
Q: QUESTION 15 Which of the following will NOT produce an ester with an acid chloride? O CH3CH2OH O (CH...
A: Question 15 Correct option : CH3CH2OCH2CH3
Q: THERMODYNAMICS: The combustion of 1.00 mol of glucose (C6H1206) liberates 2820 kJ of heat. If 1.38 g...
A: Given: Heat released on combustion of 1 mol glucose = 2820 KJ Mass of glucose = 1.38 gm Mass of w...
Q: THERMODYNAMICS: Use the given standard enthalpies of formation to calculate the AH° (in kJ) for the ...
A:
Q: A solution of sodium hydroxide is standardized to a concentration of 0.600 M NaOH. If 20.05 mL of th...
A:
Q: Determine the molar mass (amu) of Metal (M) if it took 1.68 minutes for a current of 1.08 A to plate...
A: The metal will come out as M3++3e-= M . We can conclude from here that 3 moles of electrons are requ...
Q: ELECTROCHEMISTRY: Determine the molar mass (amu) of Metal (M) if it took 1.24 minutes for a current ...
A:
Q: -COOH (7a)_ + 2 NaOH _(7b)_ NazCOs. +. H2O
A: An acid is a chemical substance that can furnish the hydrogen ions in an aqueous solution and can be...
Q: THERMODYNAMICS: A 1.21 g sample of acetic acid (HC2H3O2) was burned in excess oxygen in a bomb calor...
A:
Q: ELECTROCHEMISTRY: Given the line notation: Pt(s) | X+(aq), X2+(aq) || Y3+(aq) | Y(s) ...
A: Given the line notation: Pt(s) | X+(aq), X2+(aq) || Y3+(aq) | Y(s) Giv...
Q: ELECTROCHEMISTRY: Given the line notation: Pt|Xa), X7+ (ag) Where: x2+ + e → X+ y3+ + 3e → Y Pt++ + ...
A:
Q: What will be the major product for the following reaction?
A: Carboxylic acid react with alcohol form ester
Q: How many grams of neon occupy a volume of 26 L at STP?
A: At Standard Temperature and Pressure (STP) a gas occupies a volume of 22.4L per mole. Number of mol...
Q: C9H14N4O3 Draw a complete lewis structure with bond angles and shapes
A: We have to draw a complete Lewis structure with bond angles and shapes for the following given compo...
Q: Il Beedéd for this question. Taking logarithms and antilogarithms is necessary to solve many chemist...
A: Interpretation: We have to fill the table.
Q: ELECTROCHEMISTRY: Calculate the cell potential of a voltaic cell given the following standard potent...
A: E cell is given by Ecathode - Eanode . Cell with higher reduction potential is cathode and the other...
Q: ELECTROCHEMISTRY: Determine the molar mass (amu) of Metal (M) if it took 1.12 minutes for a current ...
A: Given: Time = 1.12 minutes = 1.12×60 sec Correct = 1.22 A Mass of the metal M = 0.0896 g
Q: ELECTROCHEMISTRY: Determine the molar mass (amu) of Metal (M) if it took 1.57 minutes for a current ...
A: Using the formula for mass deposition to solve this.
Q: How much heat in kJ is required to convert 916 g of ice at -11.0°C to liquid water at 99.0°C?
A: Ice undergo various transition from -11°C to 99°C and releases heat at each stage
Q: THERMODYNAMICS: Calculate the value of AH (in kJ) for the reaction, 2AR3 (g) + Q (s) → A2R4 (g) + QR...
A: To solve this problem we will use the given thermochemical equation
Q: Which compound would be the weakest acid?
A: The acids given are,
Q: 'hich of the following is expected to have the highe *NH2
A: According to the question, we need to determine which organic compound will have the highest boiling...
Q: ELECTROCHEMISTRY: Given the line notation: Pt(s) | X+(aq), X2+(aq) || Y3+(aq) | Y(s) where: X2+ + e-...
A:
Q: ELECTROCHEMISTRY: Copper was electroplated from a Cu2SO, solution onto an electrode with an active s...
A:
Q: A gas compressor releases 1,500 kJ of heat while doing 500 kJ of work. Calculate AEsurroundings. A) ...
A: According to the first law of thermodynamics, the energy of the universe remains constant. For a the...
Q: NUCLEAR CHEMISTRY: The mass defect of nuclei (9/x) is 1.072 amu. Estimate the total binding energy p...
A: Given : mass defect = 1.072 amu Speed of light = 2.998 x 108 m/sec
Q: SUPPLY THE MISSING DATA.WRITE THE CORRECT CONDENSED STRUCTURAL FORMULA AND GIVE THE IUPAC NAME.ANSWE...
A: Since you have posted questions with multiple sub-parts, we are entitled to answer the first 3 only....
Q: NUCLEAR CHEMISTRY: Consider the nuclei 1150 Sn. Determine its mass defect in kilograms if its atomic...
A:
Q: Which of the following will NOT produce an ester with an acid chloride? O CH3CH2OH O (CH3)3COH O CH3...
A: Let an acid chloride R-CO-Cl As we know that Cl is a leaving group .So when nucleophile will attack...
Q: Which of the following will give an N-nitroso-N-methyl amine?
A: N-Cyclohexyl-N-methyl amine reaction with HNO2 (NaNO2 + HCl) gives N-nitroso-N-methyl amine.
Which of the following will undergo hydrolysis the fastest?
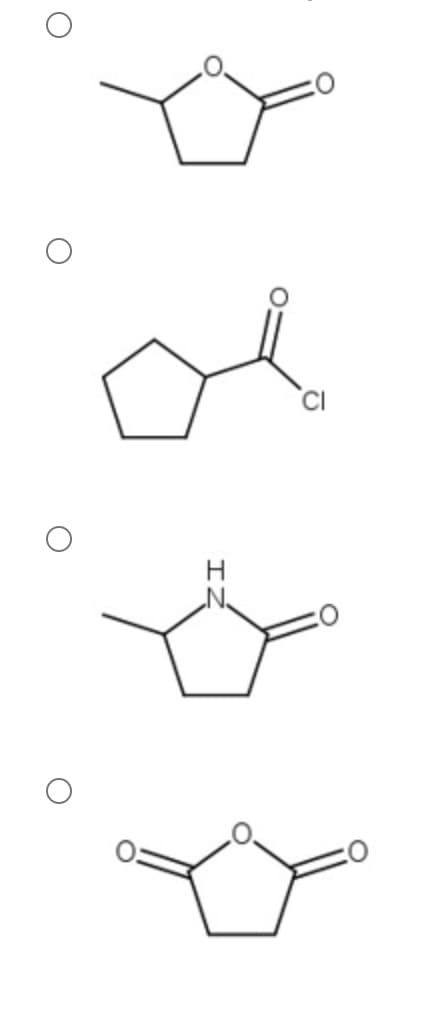
Step by step
Solved in 2 steps with 1 images


