Q: Determine the product of the following reaction. OH PCC (excess) CH,Cl2 omo HO, IV %3D
A: PCC is Pyridinium Chlorochromate.
Q: Examine the following reactions, then label each with the mechanism that is mostlikely to occur: ( c...
A: The SN1 reaction is a substitution nucleophilic unimolecular reaction. It is a two-step reaction. In...
Q: What is the pH of a 0.41 M HCl solution? Report your answer with two significant figures.
A: Given: Concentration of HCl = 0.41 M
Q: The table shows four reactions. Reactions Reaction Equation 1 C3H8 + 5O2 → 3CO2 + 4H2O 2 C6...
A: Combination Reaction: Combination reaction is a type of chemical reaction in which two or more eleme...
Q: What is the cell reaction for the following voltaic cell?Al(s) | Al3+(aq) || Cl–(aq) | Cl2(g)| Pt(s)
A: voltaic cell is the electrochemical cell used to produce electrical energy. oxidation occurs at ano...
Q: Consider the balanced equation of KIKI reacting with Pb(NO3)2Pb(NO3)2 to form a precipitate. 2K...
A:
Q: Which statement regarding the hybridisation of carbon in the following compound is correct all Selec...
A:
Q: Determine the number of valence electrons in acetone (CH,C(0)CH3) and then draw the corresponding Le...
A: Lewis structures is used to represent the covalent bonds formed between the atoms using valence shel...
Q: A detergent solution has a pH of 11.84 at 25°C. What is the hydroxide-ion concentration? Concentrati...
A:
Q: Consider the following standard electrode potentials:
A: Under standard state condition, E°cell = Ereduction - Eoxidation = 0.80 V -(-...
Q: How many grams of AgCl will be formed when 60 mL of 0.500M AGNO, is completely reacted according to ...
A: It is given that 60mL of 0.500 M AgNO3 is reacting completely (limiting reagent) to form AgCl and th...
Q: Can you please answer number 6 and show all of the steps to the solution
A: Given: [Ag+] = 1 M [Mg2+] = 1 M at anode : Mg →Mg2+ + 2e- Eo = - 2.37 V at cathode : Ag+ + e- →Ag ...
Q: Calculate the grams of solute needed to prepare 30.0 mL of a 8.00 M HCl solution with appropriate un...
A:
Q: Based on nuclear stability, what is the symbol for the most likely product nuclide when phosphorus-3...
A: e-10
Q: a student prepared a solution by adding 16.75 ml of ethanol (C2H5OH) to obtain a final solution of 1...
A: Ratio of volume of the solute to the volume of the solution multiply by 100 is known as the percenta...
Q: Question 5
A:
Q: Q. Identify the products (from 1 to 10) in the following scheme: HNO, H,SO, AICI, Zn(Hg) HCI KMNO4 (...
A:
Q: Propose a plausible synthetic route using the given reactant to form the desired product. 3 rxns HO.
A: The first step involved the esterification of the given carboxylic acid, followed by enolate formati...
Q: d. Calculate the boiling point of a 0.45 m aqueous Mg(NO3)2 solution (k» = 0.52 for water)
A:
Q: question 4, 5 , 6
A: Lewis structure is the structure in which valence shell electrons are represented in a simplified fo...
Q: 5. Predict the structure of the product formed (with correct stereochemistry) for each of following ...
A:
Q: What is the pOH of a 0.43 M KOH solution?
A:
Q: 6) Chromium can be electroplated from aqueous potassium dichromate, with thereduction shown below. I...
A: The chemical reaction corresponding to electroplating of Chromium is given as:Cr2O72- (aq) + 14 H+ (...
Q: Use the NMR to solve for product B. Show a synthetic scheme where your carbon backbone is created by...
A: i) Two doublet for 4H at the aromatic region suggests that the molecule is 1,4-disubstituted benzene...
Q: Two bulbs of different volumes are separated by a valve. The valve between the 2.00 L bulb, in which...
A: Given, two bulbs of different volumes are separated by a valve,volume of gas of bulb 1st V1 =2.00 Lp...
Q: What is the equilibrium concentration of Br2 if [HBr] 0.35 M and [H2] = 0.22 M at equilibrium? |3D H...
A:
Q: ................... hv 35% 65%
A: Alkanes are usually less reactive but in presence of sun light conditions it undergo free radical s...
Q: Is a system at Equilibrium if the rate constants of the forward and reverse reactions are equal?
A: Rate constants of forward reaction and backward reaction or reverse reaction is not equal Example :...
Q: Which of the following statements is true concerning the voltaic cell shown below? A) Cu is the anod...
A: Voltaic cell : It is also known as galvanic cell , i.e. electrochemical cell which converts chemical...
Q: Reaction of H2 with ethylene, in the presence of a small amount of palladium catalyst, gives ethane ...
A: The reaction of H2 with ethylene or ethane is a hydrogenation reaction, in the presence of a Pd cata...
Q: How many moles of chlorine gas at 120. °C and 33.3 atm would occupy a vessel of 12.0 L?
A: The ideal gas equation is: PV=nRT where, P is the pressure of the gas V is the volume of the gas n i...
Q: 1. A solution was prepared by dissolving 18.00g of glucose in 150.0g of water. Theresulting solution...
A: When non-volatile solute is added to a solvent the boiling point of the solvent increases and this c...
Q: Classify the reaction as (a) radioactive decay (b) nuclear transmutation, (c) nuclear fusion, (d) n...
A: The given reaction is as follows: Cm+5793239B→ 98245Cf + 01n
Q: Using periodic trends why Nal is an ionic bond and not a covalent
A:
Q: What is the pH of a buffer made from 0.220 mol of HCNO (Ka = 3.5 x 10-4) and 0.410 mol of NaCNO in 2...
A: Buffer is a solution which can resist change in pH when small amount of acid or base is added to it....
Q: question 5
A: 5. PV =nRT pressure at STP = 1 atm Temperature at STP = 273K 1×V =weight Gram molecular weight ×R×T1...
Q: How many grams of nitric acid , HNO,, are required to 31 neutralize (completely react with) 4.30 |g ...
A:
Q: What is the condensed structural formula for methylcyclohexane?
A: The condensed structural formula is a way to represent the molecule’s no. of carbon atoms and hydrog...
Q: Can you please answer number 2 and show all of the steps
A: Gibbs Free Energy is a thermodynamic quantity. It is the amount of energy associated with a chemical...
Q: If the rate of reaction of [0.1 M] sodium cyanide with [0.1 M] 2- bromo-2-methylpropane is 1.2 mole/...
A:
Q: Consider the balanced equation for the reaction of aluminum with hydrochloric acid. 2 Al+6 HCI 2 AlC...
A:
Q: You have a calorimeter with a heat capacity of 0.426 kJ/ºC. You combine 50.0 mL 1.50 M NaA (A- = con...
A: Given data,Heat capacity of calorimeter=0.426kJ/oCInitial temperature=18.6oCFinal temperature=21.1oC
Q: A chemist carefully measures the amount of heat needed to raise the temperature of a 140.0mg sample ...
A:
Q: Compute the weight (g) of solute needed to make the solutions listed below:a. 1250mL of 0.85M AlCl3b...
A: We'll answer the first question since the exact one wasn't specified. Please submit the question spe...
Q: Consider the balanced reaction of magnesium and oxygen. 2Mg+O2⟶2MgO2Mg+O2⟶2MgO What mass, in grams...
A: Given, mass of O2 = 2.81 g Mass of Mg = 1.51 g Molar mass of MgO = 40.3 g/mol Molar mass of Mg = 24....
Q: e. Calculate the osmotic pressure of a 0.25 M CoS04 solution.(R = 0.0821 L.atm/mol.k) at 25ºC
A: osmotic pressure is defined as
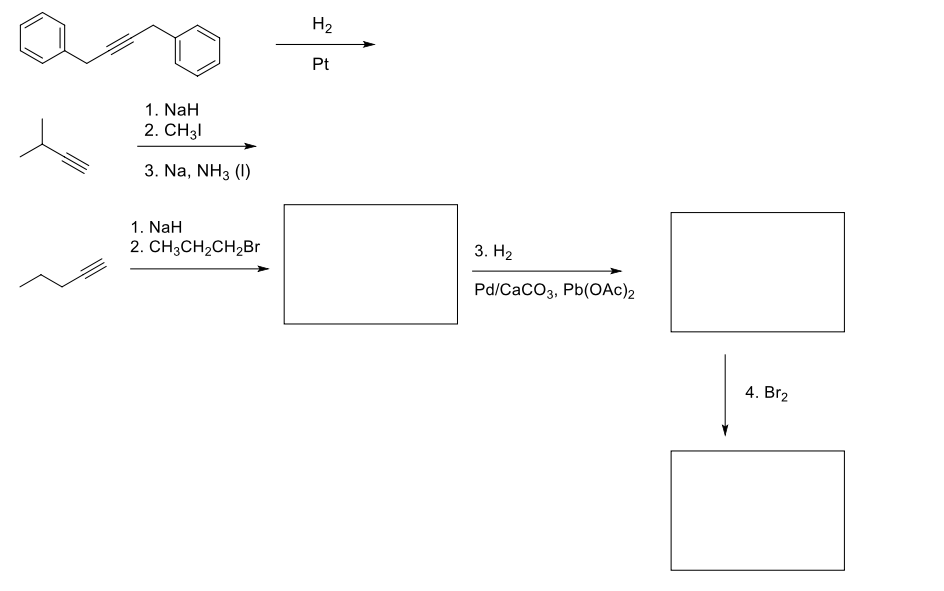
Draw the structure of the major product(s) of the following reactions. Clearly show stereochemistry if appropriate.
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 1 images

- ?⟶2? [A] (M) 1.33 × 10−2 2.66 × 10−2 3.99 × 10−2 Rate (L mol−1 h−1) 3.80 × 10−7 1.52 × 10−6 3.42 × 10−6Cocaine(C17H21O4N) is a natural substance found in the leaves of the coca plant, which has been used for centuries as a local anesthetic and stimulant. Illegal cocaine arrives in the USA either as a pure compound or as the hydrochloride salt(C7H21O4NHCl). At 25 degrees celsius, the salt is very soluble in water(2.59kg/L), but pure cocaine is much less so(1.70g/L) a. What is the maximum amount of salt in grams that can dissolve in 50.0mL of water? b. When a 25mL aqueous solution that contains 62.50 g of the salt is treated with NaOH, the salt is converted to pure cocaine. How much water in liters is needed additionally to dissolve the formed pure cocaine,How many moles of sodium bicarbonate (NaHCO3) are in 1/8 tsp of NaHCO3? Show your work, including units. 1/8 tsp NaHCO3 = 0.6 g NaHCO3
- ?CaSO4+AlCl3→CaCl2+Al2(SO4)3 Whats ?In one sentence explain what is wrong with them, and rewrite them so that the coefficients are correct. 2 Mg(OH)2(s)+4HCl(aq)->2MgCl2(aq)+4H2O(l)3 Fe(s) + 4 CO2(g) -------Fe3O4(s) + 4 CO(g)Keq = .(b) Fe2O3(s) + 6 HCl(g) ------2 FeCl3(s) + 3 H2O(g)Keq = .(c) 2 NO(g) + Cl2(g) ----- 2 NOCl(g)Keq = .
- 1.50 g of CaCl2 is sissolved in water and combined with an excess of NaCO3 CaCl2 + Na2CO3 ---> CaCO3 (s) +2NaCl mistakenly you used LiCO3 instead of NaCO3, explain the effects on your experiment?Calcium ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) Ca (s) 0 0 41.4 Ca (g) 178.2 144.3 158.9 Ca2+ (g) 1925.9 CaC2 (s) -59.8 -64.9 70.0 CaCO3 (s, calcite) -1206.9 -1128.8 92.9 CaCl2 (s) -795.8 -748.1 104.6 CaF2 (s) -1219.6 -1167.3 68.9 CaH2 (s) -186.2 -147.2 42.0 CaO (s) -635.1 -604.0 39.8 CaS (s) -482.4 -477.4 56.5 Ca(OH)2 (s) -986.1 -898.5 83.4 Ca(OH)2 (aq) -1002.8 -868.1 -74.5 Ca3(PO4)2 (s) -4126.0 -3890.0 241.0 CaSO4 (s) -1434.1 -1321.8 106.7 CaSiO3 (s) -1630.0 -1550.0 84.0 Carbon ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) C (s, graphite) 0 0 5.7 C (s, diamond) 1.9 2.9 2.4 C (g) 716.7 671.3 158.1 CCl4 (l) -135.4 -65.2 216.4 CCl4 (g) -102.9 -60.6 309.9 CHCl3 (l) -134.5 -73.7 201.7 CHCl3 (g) -103.1 -70.3 295.7 CH4 (g) -74.8 -50.7 186.3 CH3OH (g)…If I have 5 mol Br2, how much KBr will I make ? 2K + Br2 --> 2KBr

