Q: how many grams of AsCl3 are produced if 3.9363g of AsF3 are allowed to react with excess C2Cl6.
A: Weight of AsF3 = 3.9363 gm-> C2Cl6 is in excess.
Q: If the recommended dose for an adult is 350 mg of Guaiphenesin. How many moles of Guaiphenesin does…
A: Weight of Guaiphenesin = 350 mg = 0.350 g(1 g = 1000 mg)Molar mass of Guaiphenesin= 198.21 g/mole
Q: When ammonia (NH) is allowed to flow past copper(II) oxide at a high temperature nitrogen gas (N) is…
A: The mass of ammonia gas is 12.3 g.The mass of CuO is 45.6 g.The balanced reaction is 2NH3(g) +…
Q: Determine the pH-value and the degree of dissociation of sodium hydrogensulfate at 1.00 mol-1¹¹, at…
A: Sodium Hydrogen sulfate is a non proton releasing acid.Pka= 1.99
Q: The following set up is arranged in a laboratory. Vessel 1 Vessel 2 Assuming the volume of the…
A: Answer:-This question is answered by using the simple concept of calculation of pressure using the…
Q: In the context of chemical reactions, explain the concept of kinetic versus thermodynamic control…
A: The concept of kinetic versus thermodynamic control in chemical reactions explores the factors that…
Q: ify each of the molecules as one of the central electron geometries and molecular shapes. Molecule:…
A: -> electron geometry refers to arrangement of electron density around the central atoms. Geometry…
Q: Suppose you wanted to increase the buffer capacity of the formic acid/sodium formate buffer solution…
A: In this question we have to found what we have to do to increase buffer capacity.
Q: demic Upgrading Chemistry 2 6. Solid potassium chlorate, KCIO4, decomposes to form solid potassium…
A:
Q: 0.754 mol sample of hydrogen gas at a temperature of 19.0 °C is found to occupy a volume of 25.2…
A: The idea gas equation is,
Q: In the reaction of sulphuric acid and sodium hydroxide, what is the name given to the salt (ionic…
A:
Q: 6) Which of the following sets of reactions accomplish the synthesis shown below? Br H3C-C=CH Br
A:
Q: The reaction below leads to more than one product. Please write a step-by-step mechanism and ovide…
A: The addition of hydrobromic acid (HBr) to an alkene follows Markovnikov's rule. According to…
Q: Which of the following salts is the least soluble in a given volume of water? PbCl₂ Al(OH)3 Agl…
A: The question is based on the concept of solubility product principle. It that states that when a…
Q: for the following reaction mechanism: 1) H2(g) 2NO(g) -> N2O(g)+H2O(g) 2) N2O(g)+H2(g) -> N2(g)…
A:
Q: . What product(s) would you obtain as the following reactions proceed (a) (b) Br CH3CH₂CHCH3 (c) Br…
A: (a): This reaction followed SN2 reaction. Reaction is given below.
Q: A molecule is connected H₂CC (O) CH₂ where the oxygen is connected to the middle carbon and the…
A: Lewis’s bonding theory is based on the octet rule. The Lewis structure is a simplified…
Q: 7) What name is commonly given to the elements of Group 1 of the periodic table? 8) True or False…
A: 7.The name commonly given to the group 1 Elements in the periodic table is alkali metals.The name…
Q: What type (redox, acid-base, precipitation) are the reactions below? For the redox reactions,…
A: a. FeCl3 + Cu FeCl2 + CuClb: CuSO4 + Pb(NO3)2 PbSO4 + Cu(NO3)2c: NH4NO3 N2O +…
Q: A total of 1.558 F of electricity (1 F = 1 mol e) was required to electrodeposit all of the Zn and…
A: ZnSO4 and CoSO4 both have divalent metal ions as, Zn2+ and Co2+. (Here M= metal)So, 2F electricity…
Q: C1. Subject:- Chemistry A solution is made by mixing 120 mL of 1.5 M disodium oxalate and 75 mL of…
A: Volume of disodium oxalate = 120 mlMolarity of disodium oxalate= 1.5 MVolume of sodium oxalate= 75…
Q: name the following compound: Ag2S
A: Naming of compound is done in such a way that the name of the cation is written first then the name…
Q: Calculate the molar solubility of CaCO3, Ksp 8.7×10 ⁹.
A: When sparingly soluble salt is dissolve in water most of the salt remain undissolved in the…
Q: O LOH H₂SO4, A & 1) 2 equiv 2) H₂O MgBr
A: In a Grignard reagent, an alkyl group is attached to the electropositive metal, magnesium, which…
Q: Imagine you collected a master batch (undiluted) wastewater sample as shown in the illustration…
A: A question based on concentration terms. A set of data about Pb in diluted solution is given using…
Q: H₂O(g) + Cl₂O(g) = 2HOCI(g) K = 0.0900 @ 298 K The sealed 2.00 L reaction vessel was charged with…
A:
Q: Use the following concentrations to calculate your experimental Ksp value for Ca(OH) 2 [Ca^ 2+…
A: We have to calculate the solubility product for Ca(OH)2
Q: Which molecule is more acidic? A HO C HO CI B HO D Di HO Br F
A: The electron-withdrawing or electron-donating properties of the substituents linked to the carboxyl…
Q: Which of the following has the same number of significant figures as the number 10.3100? 8.66…
A: In the given question we have to find the number which has same number of significant figures as the…
Q: Answer ASAP please! :) Thank you During the corrosion of Magnesium, the anode reaction is ... a) Mg…
A: In electrolytic cell, the reaction at cathode is the reduction reaction (accepting electrons).…
Q: 12.44 Classify each of the following alcohols as primary (1°), secondary (2°), or tertiary (3°):…
A: Note: Since you have posted a question with multiple sub-parts, we will solve the first three…
Q: Give 3 reasons that ligand A is a suitable ligand for lanthanide complexation. O₂C *O₂C N N *O₂C A…
A: It has 8 donor centres= 3 N donor centres + 5 O donor centres
Q: Draw the most stable chair conformation of β-D-galactopyranose. Name and draw the major product that…
A: We have to draw the most stable chair conformer of β-D-galactopyranose.
Q: X 58 dakika kaldıØ JUIST Which one is false about the reaction? OH Son kaydetme 14:01:57 CrO₂ acetic…
A:
Q: Decide which element probably forms a compound with chlorine that has a chemical formula most and…
A: Potassium is an element which is a metal. It has symbol 'K'.Chlorine is an element which is…
Q: Interpret the pictures below in terms of nanoparticles and nanotechnology.
A: Nanoparticles (NPs; 1–100 nm in size) have a special place in nanoscience and nanotechnology, not…
Q: Question 14 Structure A CO₂H H H CH 3 Br Br Structure B CH 3 H H A and B are the same molecule A and…
A:
Q: A medical technician is working with the four samples of radionuclides listed in the table below.…
A: we have to complete the given table with respect to the radioactivity of the given samples
Q: Rank the following bonds from most polar to least polar? Cl-O, Mg-O, O-O, C-O
A: -> polarity of bonds can be identified using electronegativity of constituent atoms.
Q: Describe the difference between the Rutherford model of the atom and the Bohr- Rutherford model of…
A: Atomic models that were proposed to describe the structure of atoms include the Rutherford model and…
Q: Q1. Which of the following alkyl halides form a substitution product in an SN1 reaction that is…
A: SN1 is unimolecular nucleophilic substitution reaction which involves formation of carbocation as…
Q: how many grams of nitrogen would be formed if 12.3g of ammonia was passed over 45.6g of copper(II)…
A: From the given equation and given data we have to estimate the amount of nitrogen .
Q: 2) Look at each of the following sets of structures and determine if they are resonance structures…
A: In this question we have given some organic compounds and we have to tell that wheather those…
Q: Which of the following would be classified as cephalins? (Select all that apply.) CH₂ 0-…
A:
Q: Boron metal has two naturally occurring isotopes: boron-10 (19.90%; isotopic mass-10.01) and…
A: The atomic mass is calculated by the summation of atomic mass of atomic mass times fraction of the…
Q: What is the major product of the following reaction? (CH3)3CO- (CH3)3COH Br O O O O = = _ > IV IV
A:
Q: Using the information from the table below, what should be the unit for k? Total reaction order 0 1…
A:
Q: calculate the mass of plutonium-238 remaining after 350.0 years.
A:
Q: For the equilibrium system described by 2 SO₂(g) + O2+2 SO at a particular temperature the…
A:
Q: Please draw the HOMO and LUMO of the superoxide ion O2^-. Please draw the overlap of the constituent…
A: The molecular orbital theory describes the electrons' energy states and spatial distribution where…
A chemist has been asked to make the chemical shown below. Draw and name the one initial organic chemical used and devise a synthesis for the chemical below. Ensure you include all reagents and conditions.
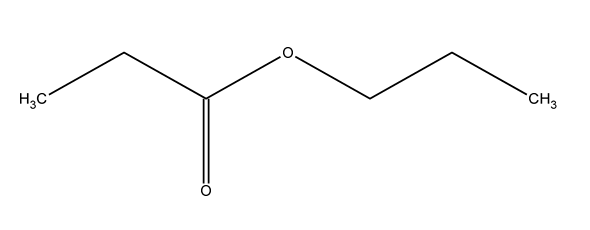
Step by step
Solved in 3 steps with 1 images


