Q: isotope of technetium (technetium-99m), has a half-life of 6 hours. A hospital has a sample which…
A: Half life = 6 hoursInitial amount of technetium-99m = 36 mgTime = 12 hours.
Q: The preparations of two aqueous solutions are described in the table below. For each solution, write…
A: a) 0.4 mol of NaOH is added to 1.0 L of a 0.4M HCH3CO2 solution.b) 0.1 mol of NaOH is added to 1.0…
Q: 2) Look at each of the following sets of structures and determine if they are resonance structures…
A: Resonance structures are molecules where there is Delocalisation of charge to attain overall…
Q: ball & stick V + labels What is this base from RNA and DNA?
A: DNA and RNA contain nitrogenous bases.Adenine(A), Guanine(G) , Cytosine(C) and Thimine(T) are…
Q: In the context of chemical reactions, explain the concept of kinetic versus thermodynamic control…
A: The concept of kinetic versus thermodynamic control in chemical reactions explores the factors that…
Q: For n=4, what are the possible sublevels? a. 1,2,3,4 b. 0,1 c. 0,1,2 d. 0,1,2,3 e. 0
A:
Q: A solution is made by dissolving 15.4 g of HCl in 500 mL of water. Calculate the pH of the solution…
A:
Q: Carbon can exist in many forms known as allotropes. Two such allotropes are diamond and graphite.…
A: An element exist in two or more different forms known as allotropy and carbon exhibit allotropy.…
Q: What is the maximum number of electrons that can occupy the 2p subshell? O 3 O 10 O 12 O 6 02
A: -> One orbital can occupy maximum two electrons.-> There is fixed number of orbital in every…
Q: Be sure to answer all parts. In an analysis of interhalogen reactivity, 0.390 mol of ICI was placed…
A: Kc is an equillibrium constant. In its expression all the species involved in the reaction is…
Q: 9. The structure of primary alcohol can be differentiated from other structures in the MS by: (a)…
A: The right solution to the inquiry is (d) MacLafferty reworking.In mass spectrometry (MS), separating…
Q: What is the splitting pattern expected for the proton at C-2 in the following compound? HH He H H Br…
A: 1H NMR spectroscopy is used to determine the structure of unknown compound in organic chemistry. The…
Q: In the Lewis structure for Borane (BH3), how many bonding electrons (not pairs) are near the central…
A: Bonding pair are the pairs of electron that are in a bond.A single bond has one bond pair means 2…
Q: Without rotating the bond, draw a proper Newman structure of the following molecule, looking down…
A: In this question we have given the wedge dash structure of an organic compound and we have to draw…
Q: For a particular redox reaction, MnO₂ is oxidized to MnO and Ag+ is reduced to Ag. Complete and…
A: In balanced chemical equation, the no. of all the atoms involved in a reaction should be equal in…
Q: C1. Subject :- Chemistry
A: The final quantity of reaction's species for any reversible reaction is computed using equilibrium…
Q: An element has three naturally occurring isotopes with the following masses and natural abundances:…
A: In order to calculate relative atomic mass, simply multiply each isotopic mass by the percentage…
Q: In the process 92235U + 01n → 52137Te + 4097Zr + 201n, what can the two neutrons at the end do?
A: This is a nuclear fission reaction. A nuclear fission reaction takes place when a neutron is…
Q: Must answer in Handwriting ok. 4. Draw a titration set-up when (a) standardizing NaOH, (b) analyzing…
A: Titration is simple analytical procedure to determine the concentration of the unknown sample by…
Q: A gas at 27 degrees Celsius and 556 mm Hg occupies 12.7L. Determine the molar volume of this gas…
A: Answer:-This question is answered by using the simple concept of calculation of molar volume using…
Q: Consider separate 1.0-L gaseous samples of CH4, O2, and Ar, all at STP and all acting ideally. Rank…
A:
Q: Imagine you collected a master batch (undiluted) wastewater sample as shown in the illustration…
A: A question based on concentration terms. A set of data about Pb in diluted solution is given using…
Q: Give 3 reasons that ligand A is a suitable ligand for lanthanide complexation. O₂C *O₂C N N *O₂C A…
A: It has 8 donor centres= 3 N donor centres + 5 O donor centres
Q: The strength of a covalent bond depends upon the size of the atoms and the bond order. In general…
A: Bond energy is directly proportional to the bond order and inversely proportional to the bond…
Q: Name all the alkyl groups from 1 carbon to 10 carbon using the line structure and angle structure
A: Alkyl group composed of carbon and hydrogen atoms. The alkyl group are defined based on the number…
Q: Ammonia is synthesized from nitrogen gas and hydrogen gas at 400 degrees Celsius and 350 kPa. If…
A: Nitrogen reacts with hydrogen to form ammonia. The equation for the balanced chemical reaction is…
Q: The unknown reaction: A -> P has been measured at the following temperatures: Temp = 298 K Initial…
A: Order of reaction cannot determined theoretically, its a experimental measurement. From the given…
Q: 40.00 mL of 0.300 M Fe3+ solution is titrated with 0.150 M Sn2+ solution. a) Calculate the…
A:
Q: A brain scan uses the radioisotope oxygen-15. The recommended dosage is 50 mCi . A supply of 230 mCi…
A: Recommended dosage = 50 mCisupply of 250mCi correspond to 40ml
Q: The following set up is arranged in a laboratory. Vessel 1 Vessel 2 Assuming the volume of the…
A: Answer:-This question is answered by using the simple concept of calculation of pressure using the…
Q: Cyclopentene is treated with hydrogen gas (H₂) and a platinum catalyst to form the product…
A: A question based on reactions of alkene. A reaction involving conversion of cyclopentene into…
Q: 2 요 NaOH and 2 water
A: For the synthesis of complex compounds and the formation of carbon-carbon bonds, the Aldol reaction…
Q: What is the best explanation for why metals are ductile? Valence electrons carry charge anywhere…
A: Ductility is the property of a material that allows it to be stretched or drawn out into a wire or…
Q: Using just a periodic table (not a table of electronegativities), decide which of these is likely to…
A: Answer:-This question is answered by using the simple concept of polarity of the covalent bond.
Q: Consider the following statements about ionization energy. Which ones are true? 1. It takes energy…
A: Ionization energy is defined as the amount of energy required to remove valence electron from…
Q: Suppose a pair of chemical compounds A and B can react in two different ways: C Reaction 1 gives…
A: Energy diagram of given reactions is to be drawn.
Q: Complete the reactions by drawing the structure of the O a) Ob) Oc) d) CIH O + H₂ :OH OH aOH FOH aoH…
A: -> H2/Pt can reduce aldehyde and ketone to alcohol but it can not reduce carboxylic acid.->…
Q: The electron has the positron as its antiparticle. a True b False
A:
Q: Students in a chemistry lab are making the compound cobalt (II) carbonate. It can be made by…
A:
Q: The volume of the HCI solution is 80.5 mL and the volume of the 0.10 M NaOH titrant at the…
A: Volume of the HCI solution = 80.5 mL volume of the 0.10 M NaOH titrant at the equivalence point =…
Q: hydrocarbon has mass percent composition and molecular formula weight shown below. How hany atomic…
A: Molecule is a hydrocarbonPercentage of C in molecule = 83.6 %Percentage of H in molecule = 100-…
Q: 5. The initial concentration of morphine, CHNO, in a solution is 3.6 x 10³ mol/L. The pOH of the…
A: 5.The initial concentration of morphine is C = 3.6×10-3 M.The pOH of the solution is 4.53.We have to…
Q: The nuclear particle carrying a charge of +1e is the a electron b neutron c proton
A: The neutrons and protons reside in the nucleus of an atom, whereas, the electrons revolve around the…
Q: Which statements are true about the isolated, single p orbital shown below? 8 I. It can hold up to…
A: 1. Orbital is the region where probability of finding electron is maximum. 2. Various orbital are s…
Q: 4. a. The ¹H NMR spectrum of a compound with the molecular formula C-H₁0O2 is shown below (integral…
A: The concept of this question is based on organic spectroscopy which includes HNMR, C13 NMR, UV and…
Q: Consider the following resonance forms (lone pairs and formal charges not shown) of acetamide: O…
A: Following resonance structures are given - Following carbocation also given -
Q: Question 3 Isomer A A B CH 3 I Which isomer is chiral? Isomer B CH3 CH₂
A: Chirality refers to a property of certain objects or molecules that are non-superimposable on their…
Q: how many grams of AsCl3 are produced if 3.9363g of AsF3 are allowed to react with excess C2Cl6.
A: Weight of AsF3 = 3.9363 gm-> C2Cl6 is in excess.
Q: Write an acceptable IUPAC name for the compound below. (Only syster Keep the information page open…
A: Note: Since you have asked multiple questions, we will solve the first question for you. If you want…
Q: Boron nitride is the second hardest substance known (next to diamond). It has a very high melting…
A: A substance made of boron and nitrogen atoms is called boron nitride (BN). A substance's…
Give answer asap..I will give you upvote
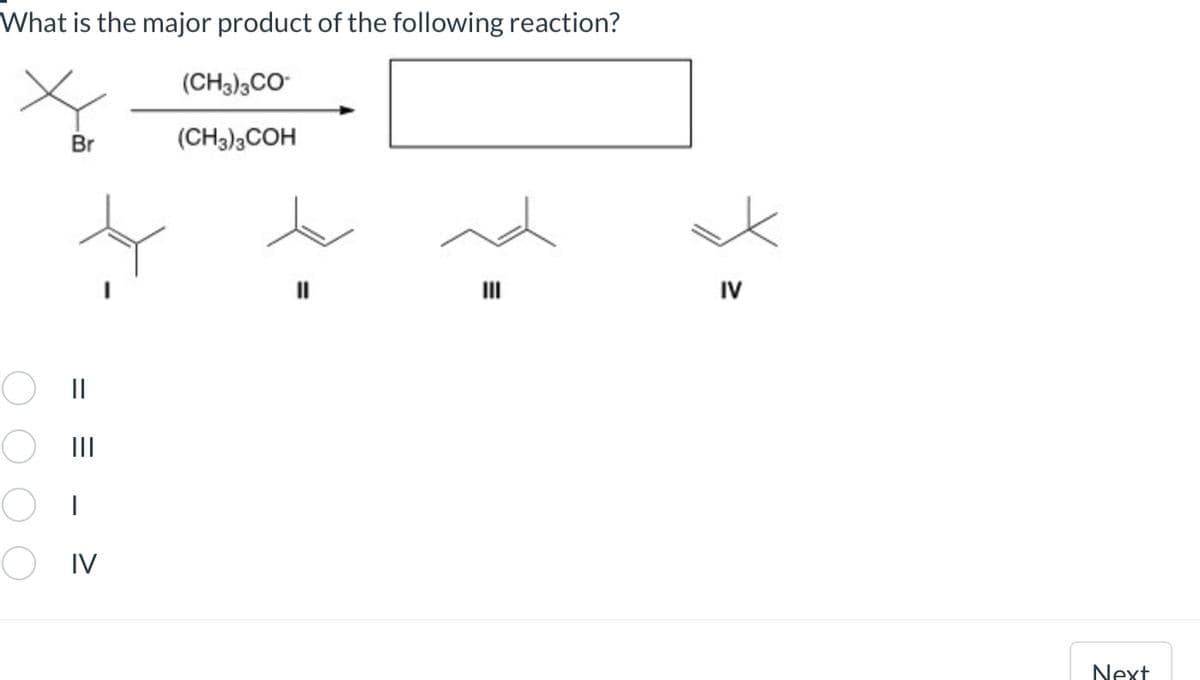
Trending now
This is a popular solution!
Step by step
Solved in 4 steps with 3 images

- 2) What is the main product for each of the following reactions? (d) Styrene + CH3CH2COCl AlCl3, then Br2, Fe -----> ? (e) Benzene + 1-chloro-2,2-dimethylpropane, AlCl3 -----> ? (h) Benzene + HCOCl, AlCl3 then 1-chlorobutane, AlCl3 -----> ?The following product can be synthesized from the reaction of an alkene with Hg(OAc)2 followed by reduction with NaBH4. Draw all possible alkenes that will give the product shownRank the following groups in order of decreasing priority. −COOH, −CH2OH, −H, −CHO
- Thank you for the answer, would it be possible to follow the path I have attached as well srarting from methanol ---> CH3Br -----> CH3MgBr-------> (H3C)CH-CH2OH ----> E1 IsobutaleneRange order from lowest to highest leaving group TosO- OH- I- F-For each compound, give the product(s) expected from (1) HgSO4>H2SO4@catalyzedhydration and (2) hydroboration–oxidation.(a) hex-1-yne
- Which of the following is the product of the reaction shown in Image 17? a. (C6H5)3COH b.C6H5CH2OH c.(C6H5)3CH d.(C6H5)2CHOHPredict the major products formed when 2- methyl-1-butene reacts with: H2, Pt/25°C. Show the reaction mechanism the given alkene reactionsdedcue the major organic product from the following reations. be ablw to draw hydration halogenation hydrohalogenation, halohydrin formation, oxymercuratiom demercuration qnd hydroboration oxidation.

