H3C, ОН CH3 СООН CH3 H OH Cholic acid is a component of human bile whose function is to aid in the absorption and digestion of dietary fats. Draw the three-dimensional structure of cholic acid, and then determine the orientation of the following groups: #1: OH group on ring A #2: OH group on ring B #3: OH group on ring C
H3C, ОН CH3 СООН CH3 H OH Cholic acid is a component of human bile whose function is to aid in the absorption and digestion of dietary fats. Draw the three-dimensional structure of cholic acid, and then determine the orientation of the following groups: #1: OH group on ring A #2: OH group on ring B #3: OH group on ring C
Biochemistry
6th Edition
ISBN:9781305577206
Author:Reginald H. Garrett, Charles M. Grisham
Publisher:Reginald H. Garrett, Charles M. Grisham
Chapter25: Nitrogen Acquisition And Amino Acid Metabolism
Section: Chapter Questions
Problem 13P
Related questions
Question
Options are equatorial or axial.
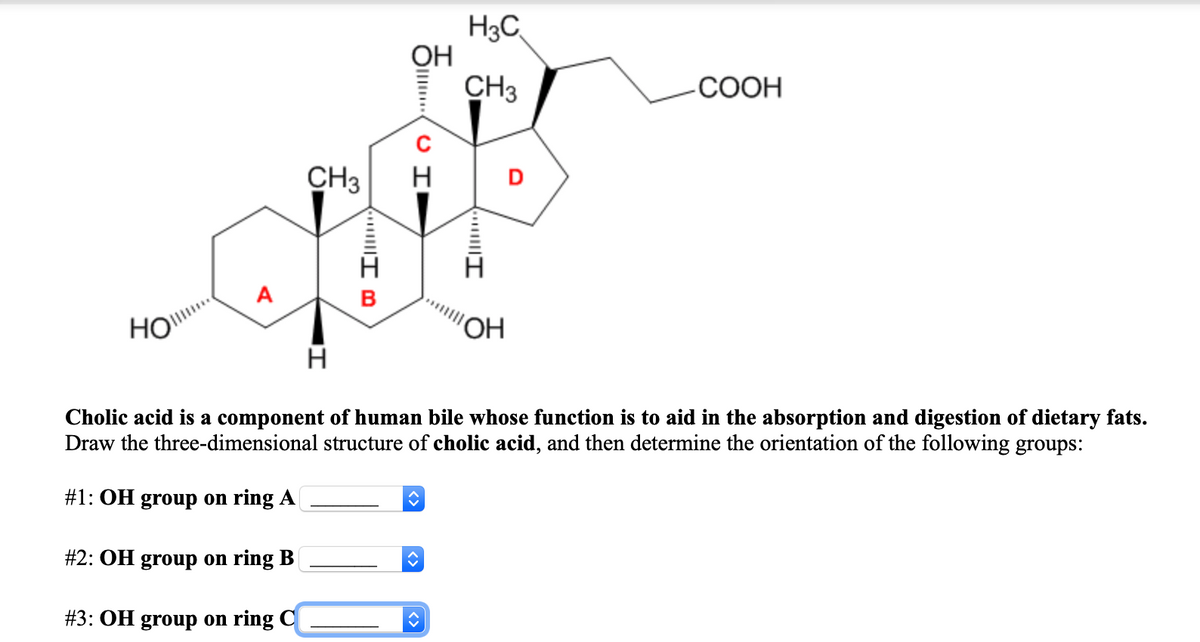
Transcribed Image Text:H3C
ОН
CH3
-СООН
CH3
H
D
A
B
HO"
H.
Cholic acid is a component of human bile whose function is to aid in the absorption and digestion of dietary fats.
Draw the three-dimensional structure of cholic acid, and then determine the orientation of the following groups:
#1: OH group on ring A
#2: OH group on ring B|
#3: OH group on ring C
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biochemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning

Biochemistry
Biochemistry
ISBN:
9781305577206
Author:
Reginald H. Garrett, Charles M. Grisham
Publisher:
Cengage Learning