Q: Spectrum 10: LOD 3000 1000 soo 4000 2000 HAVENUMBERI -
A: A question based on IR spectroscopy, which is to be accomplished.
Q: A sample of water pf unkown mass undergoes a temperature change from 86.52 degrees celsius to 30.15 ...
A: Given :- initial temperature of water = 86.52 °C final temperature of water = 30.15°C q = -17405 ...
Q: Identify the formal charge in the case below by indicating the magnitude, sign, and location of the ...
A: Given structure is : Magnitude and sign of formal charge on the molecule = ? Location of formal cha...
Q: The molar concentration of 290 ppm of Cs+ (132.91 g/mol) in a solution that has a specific gravity o...
A: Given: Concentration of Cs+ in ppm = 290 ppm And specific gravity of solution is 1.08. Since density...
Q: JO JOUIU 7) When propionic anhydride is dissolved in water, it forms propionic acid according to the...
A:
Q: Which of the following sentence is correct:
A: The term error is referred to as the uncertainty in a measurement. The three possible errors are : R...
Q: Iron was analyzed by dissolving 1.1324 g sample in concentrated HCL. The solution was diluted in H2O...
A: Given: Mass of iron sample = 1.1324 g Mass of Fe2O3 produced = 0.2697 g
Q: Use the information provided below to calculate the quantity of heat (in kJ) required to convert 63....
A: Given the mass of the substance = 63.01 g Molar mass of the substance = 27.1 g/mol => moles of th...
Q: Applications of NMR in Pharmaceutical, Fine Chemistry, University level, Medical Sciences, Petrochem...
A: Every nuclei will have spin and if an external magnetic field is applied, there will be an energy tr...
Q: Calculate the number of mmoles of solute that are present in 4000 cm of an aqueous solution that con...
A: Given, Volume of the solution = 4000 cm3 ppb (part per billion) of AgNO3 = 5000 ppb Density of th...
Q: dentify the spectra. All pertinent peaks must be assigned on spectrum (5-6 peaks). Label the peaks o...
A:
Q: A 50.0 ml of acidic solute (Ka = 1.20 x 10-2) is titrated with a %3D 2.50 mL of 1.00 x 10-5 M NAOH s...
A: Given: Ka of acid solute = 1.20 X 10-2 Volume of acid solution = 50.0 mL = 0.050 L ...
Q: = proposed Lewis structure below is reasonable.
A:
Q: O Standardization is required when the titrant solution molarity is approximate primary standard sho...
A: The incorrect statement is: A standard solution should react completely with the analyte without ...
Q: Does this molecule have both cis and trans isomers?
A:
Q: Titration of 0.2342 g of pure Na,C,0, (134.0 g/mol) required 33.45 ml of KMNO, solution according to...
A:
Q: Given the standard enthalpy changes for the following two reactions: (1) 2Pb(s) + O2(g)2PBO(s) AH° =...
A: Given that - (1) 2Pb(s) + O2(g) ----> 2PbO(s) , ∆H°1 = -434.6 kJ (2) Pb(s) + Cl2(g) ----> ...
Q: in titrating 40.0 mL of 0.10 M of HCI solution with 0.15 M of KOH solution, what would be the pH of ...
A: The balanced chemical reaction is HCl + NaOH ------> NaCl + H2O Number mmoles of HCl = molarity ...
Q: Ine pH of a 500.00 ml buffer solution containing sodium mandelate and mandelic acid (ka- 3.89x10 )(a...
A: Given, Ka = 3.89 x 10-4 ao = 0.90 Formula Used, pKa = - log (Ka) pH is calculated by Henderson–Hasse...
Q: Titration of 0.2342 g of pure Na,C,O, (134.0 g/mol) required 13.45 ml of KMNO, solution according to...
A: Number of moles = mass/molar mass Molarity = number of moles/volume of solution (in L)
Q: If the bond length in HF is 95 pm and its dipole moment is 1.53D, the charge distribution in this mo...
A: Given: For HF: Bond length = 95pm Dipole moment = 1.53 D
Q: The largest gem-quality diamond ever found is the Cullinan diamond, found in 1905 in South Africa. T...
A: #32.d Given the weight of the diamond = 3106.75 carat Cost per diamond = $ 4 million per carat
Q: A foot powder sample containing Zn was dissolved on 200.00 mL water and was titrated to the end poin...
A:
Q: A sample of water of a mass of 417.9g and an intial temperature of 61.79 degrees celsius loses 35705...
A:
Q: Iron was analyzed by dissolving 1.1324 g sample in concentrated HCL. The solution was diluted in H2O...
A:
Q: A 50.0 mL of basic solute (Kb = 1.58 x 109) is titrated with a 25.0 mL of %3D 0.100 M HCI solution. ...
A: Depending upon the PH of the solution, one can determine which indicator is the best for this acid ...
Q: 10. For each of the following compounds, draw the important resonance forms. Indicate which structur...
A:
Q: What is the major product produced in the following reaction? HO Br CH,SNa CH,OH Но HO но 1) 2) 3) S...
A: A multiple choice question based on nucleophilic substitution reaction, which is to be accomplished.
Q: please find the percent error not for each one but for the three trials together:)
A: In the given question we have to calculate the percent error for the density of water. we know that...
Q: How many grams of Ba(103)2 (487 g/mol) can be dissolved in 500 mL of water at 25°C. (Ksp of Ba(103)2...
A:
Q: Spectrum 1: LOD NH, 4000 3000 2000 1s00 1000 500 HAVENUMBERI - TRANSMETTANCEI
A:
Q: A hot cup of tea placed near an open window to cool. Which of the following statements best describe...
A: Given: Hot cup of tea placed near an open window to cool.
Q: Box the functional groups present in the following compounds and name them
A:
Q: Density problem to find the grams or mass.
A: Example 1 Density = Mass/Volume Mass = Density x Volume Example 2 From ideal gas equation, PV = ...
Q: In acid-base titration one of the following is incorrect: The end point will be sharper if the react...
A: The question is based on the concept of titrations. we have to identify the incorrect statement rega...
Q: Which of the following sentence is correct: O To increase the particle size of the precipitate the r...
A: 5 statements are given . Out of these 5 statements , one statement is correct. We have to choose tha...
Q: A 113.25 gram sample of gold is initially at 100.0 °C. It gains 20.0 J of heat from its surroundings...
A: Heat formula will be used to calculate the final temperature of gold. Here it is: Heat = Mass of gol...
Q: Which of the following is incorrect: General steps in qualitative analysis include: Method selection...
A: Chemical analysis can be of two types, qualitative analysis, and quantitative analysis. Qualitative ...
Q: Calculate the 95% confidence interval for these set of data: 3.3,3.4,3.1 and 3.6 ppm
A:
Q: Which of the following sentence is correct: Gross errors result from carelessness and the limitation...
A: The solution is given below -
Q: Which of the following is incorrect: a. General steps in quantitative analysis include: Method selec...
A: Statement a is correct as general steps is quantitative analysis includes all given steps.
Q: Five blood samples got these results: 0.752, 0.756, 0.752, 0.751, 0.760 ppm The standard deviation o...
A:
Q: Five blood samples got these results: 0.752, 0.756, 0.752, 0.751, 0.760 ppm The RSD (in ppt ) of the...
A:
Q: All of the following regarding Argentometric titration are correct except a. Chromate indicator is u...
A: fajan method An adsorption indicator shows two different colour in adsorbed or desorbed from is requ...
Q: The Pauling electronegativity of Cl is 3.2 while that of Br is 3.0. Estimate the bond dissociation e...
A: Given : EN of Cl : 3.2 EN of Br : 3.0 BDE of Cl-Cl = 160 KJ mole-1 BDE of Br- Br = 224 KJ mole-1
Q: Identify the following spectra. Assign all pertinent peaks (~5-6 peaks). Place the structure of the ...
A: The question is based on the concept of spectroscopy. we have to analyse the spectral data provide...
Q: In each box, write the number of H's that each labeled carbon has. For example, if the carbon next t...
A: We know that, carbons have four bonds. In a and b carbon atoms each carbon have three bonds and carb...
Q: Based on Kapustinskii equation, calculate the lattice energy of AIF3. Given that: W =1.079 x 105 kJ....
A: W=1.079×105 KJ.pm/mol and ionic radii areAL3+=53pm and F=119pm.
Q: Iron was analyzed by dissolving 1.1324 g sample in concentrated HCL. The solution was diluted in H,O...
A:
Q: In titrating 40.0 mL of 0.10 M of HCI solution with 0.15 M of KOH solution, what would be the pH of ...
A: Answer :- pH = 2.44 -------------------------------------------------------------------------------...
Step by step
Solved in 3 steps with 2 images

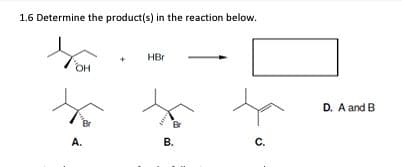
- What steps are needed to convert A to B?I know its not A or D but I don't know how to rule out either C or BAnswer each part for the reaction below. a. Draw in ALL missing lone pairs b. Draw appropriate curved arrows to complete the acid-base reaction c. Draw the corresponding products for the reaction

