The molecule 2-butene, C4H8, can undergo a geometric
change called cis-trans isomerization:As discussed in the “Chemistry and Life” box on the chemistry
of vision, such transformations can be induced by
light and are the key to human vision. (a) What is the hybridization
at the two central carbon atoms of 2-butene?
(b) The isomerization occurs by rotation about the central
C¬C bond. With reference to Figure 9.29, explain
why the p bond between the two central carbon atoms isdestroyed halfway through the rotation from cis- to trans-
2-butene. (c) Based on average bond enthalpies (Table 8.3),
how much energy per molecule must be supplied to break
the C¬C p bond? (d) What is the longest wavelength of
light that will provide photons of sufficient energy to break
the C¬C p bond and cause the isomerization? (e) Is the
wavelength in your answer to part (d) in the visible portion
of the
of this result for human vision.
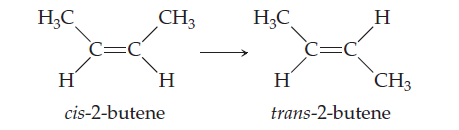
Trending now
This is a popular solution!
Step by step
Solved in 2 steps









