Chemistry
10th Edition
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Chapter5: Gases
Section: Chapter Questions
Problem 129E
Related questions
Question
Transcribed Image Text:100% 7
Tue 5:5
File
Edit
View
History
Bookmarks
Profiles
Tab
Window
Help
Chrome
$550 mustang x
O S550 Mustanc X
(2 unread) - d x
ALEKS - Davi
ALEKS
WPAL 101_ 23
Purple/Black
A Gloss Purple
D Paused
A www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-IvdWKW_BBZZ16tTytly4Fcfu6zOtOf80MM9sefhQp9QAYI8idN-02kVSt27lmROEJa_z..
O CHEMICAL REACTIONS
David
O 1/5
Using a chemical equation to find moles of product from moles ...
with
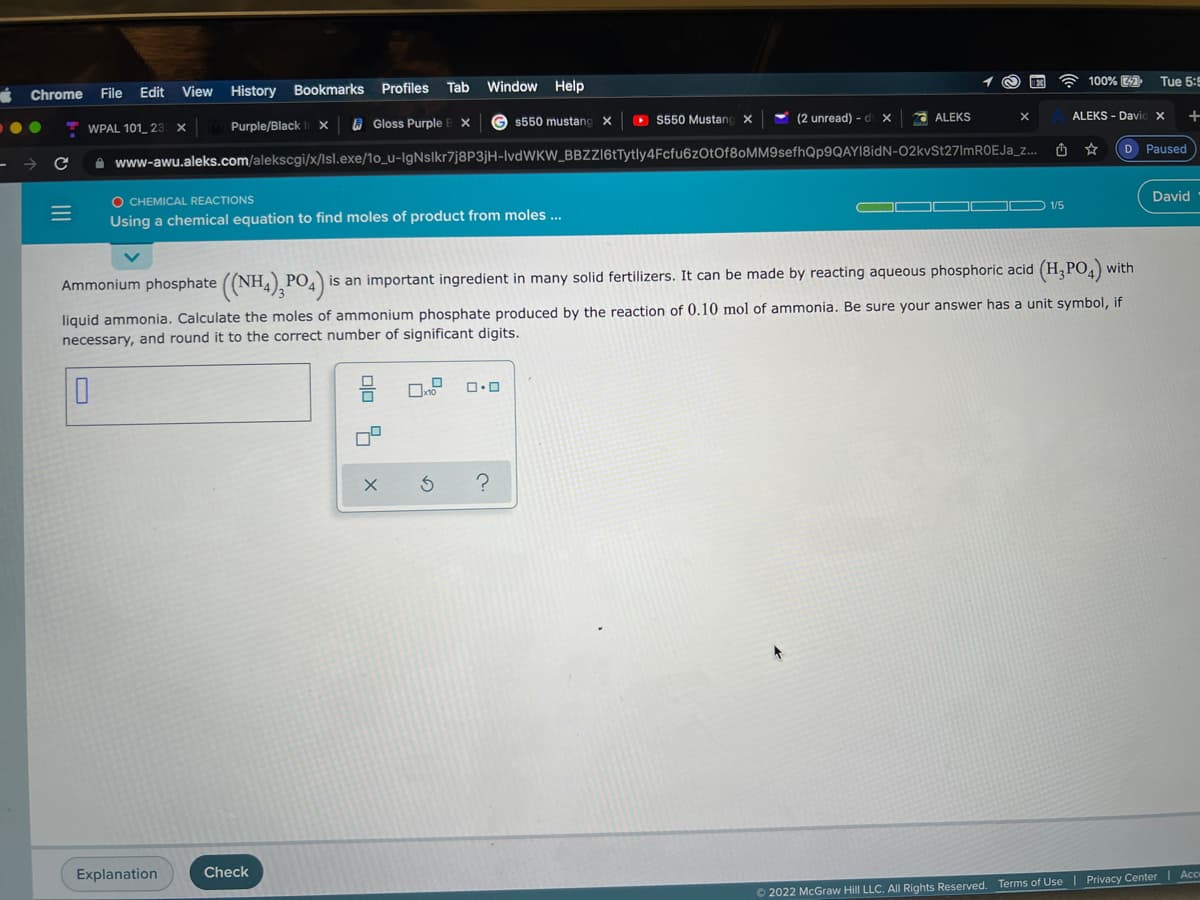
Ammonium phosphate ((NH4), PO4)
is an important ingredient in many solid fertilizers. It can be made by reacting aqueous phosphoric acid (H, PO,)
liquid ammonia. Calculate the moles of ammonium phosphate produced by the reaction of 0.10 mol of ammonia. Be sure your answer has a unit symbol, if
necessary, and round it to the correct number of significant digits.
Explanation
Check
Acc
O 2022 McGraw Hill LLC. AlI Rights Reserved. Terms of Use | Privacy Center
olo %
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning


Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning
