Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter13: The Chemistry Of Solutes And Solutions
Section: Chapter Questions
Problem 83QRT
Related questions
Question
Transcribed Image Text:云:
00
#3
Chrome
File
Edit View
History Bookmarks
Profiles
Tab
Window
Help
O Ass X
A ALE X
G Nar x C Nar X C Nar X C Usi x C Cor X b Ans X
Ho X G familiar
www-awu.aleks.com/alekscgi/x/lsl.exe/1o_u-IgNslkr7j8P3jH-Ivdr8RUjiNBZSaKW4JswZj9xS7uMs6c1gBjbNya89YwNIE
Apps 9 OpenCCC: Interna...
O Form N-648
O New Tab
I Downloads
P lapd
G My Account
Apply Now | Join...
Los Ar
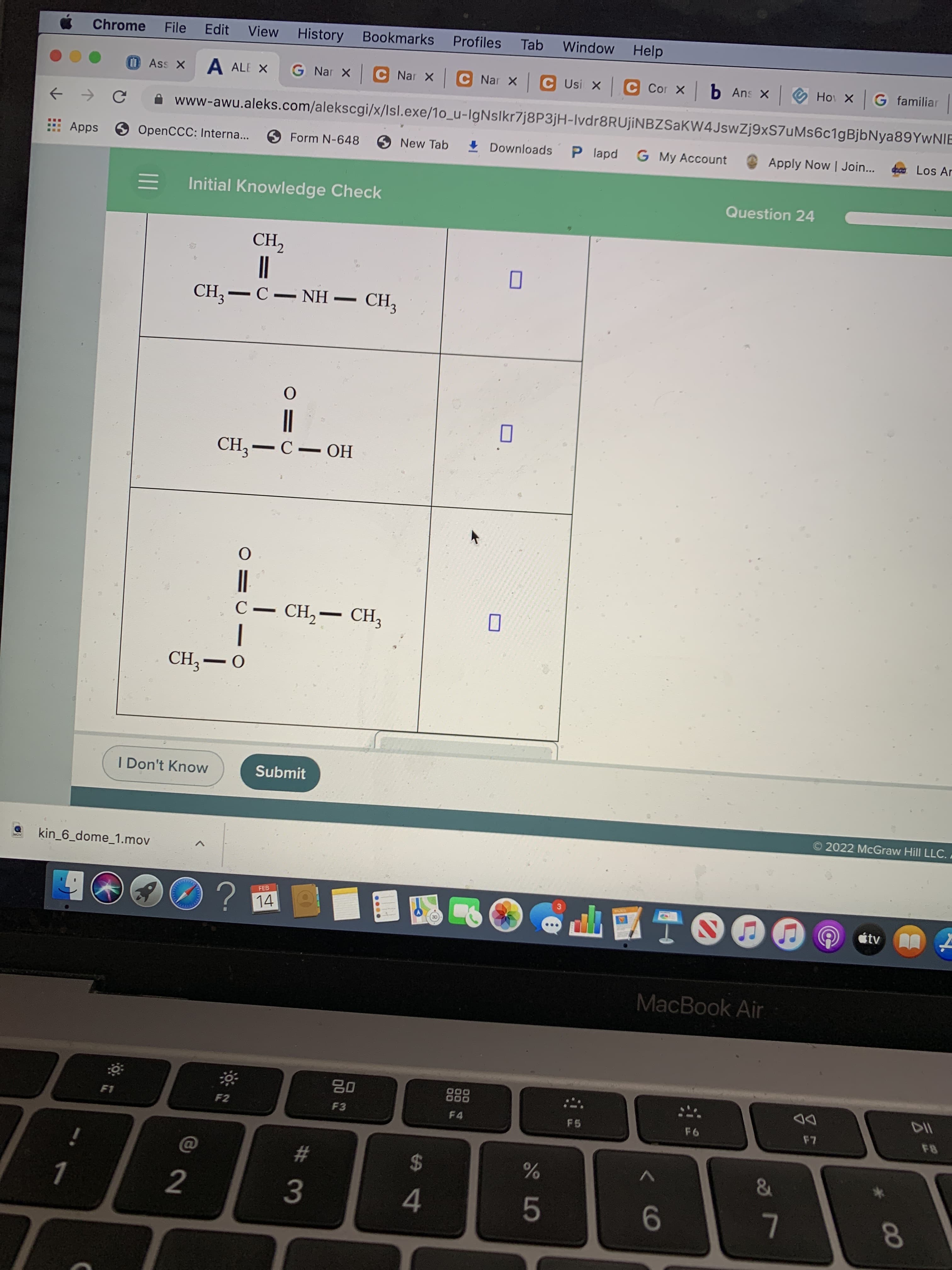
Initial Knowledge Check
Question 24
CH,–C- NH
-
|
CH,-C- OH
|
||
C– CH,- CH,
|
-
I Don't Know
Submit
© 2022 McGraw Hill LLC..
kin_6_dome_1.mov
D?
FEB
14
tv
MacBook Air
000
000
F4
F2
DD
F3
F5
FB
24
2.
4.
9-
Transcribed Image Text:File
Edit
View
History
Bookmarks Profiles
Tab
Window Help
Chrome
G Nar x C Nar x C Nar x C Usi x C Cor x b Ans x
O Ho X G familia
O Ass X A ALE X
A www-awu.aleks.com/alekscgi/x/Isl.exe/1o_u-IgNslkr7j8P3jH-Ivdr8RUjiNBZSaKW4JswZj9xS7uMs6c1gBjbNya89Yw
OpenCCC: Interna..
O Form N-648 6 New Tab
I Downloads P lapd G My Account
Apply Now | Join... Los
sdd
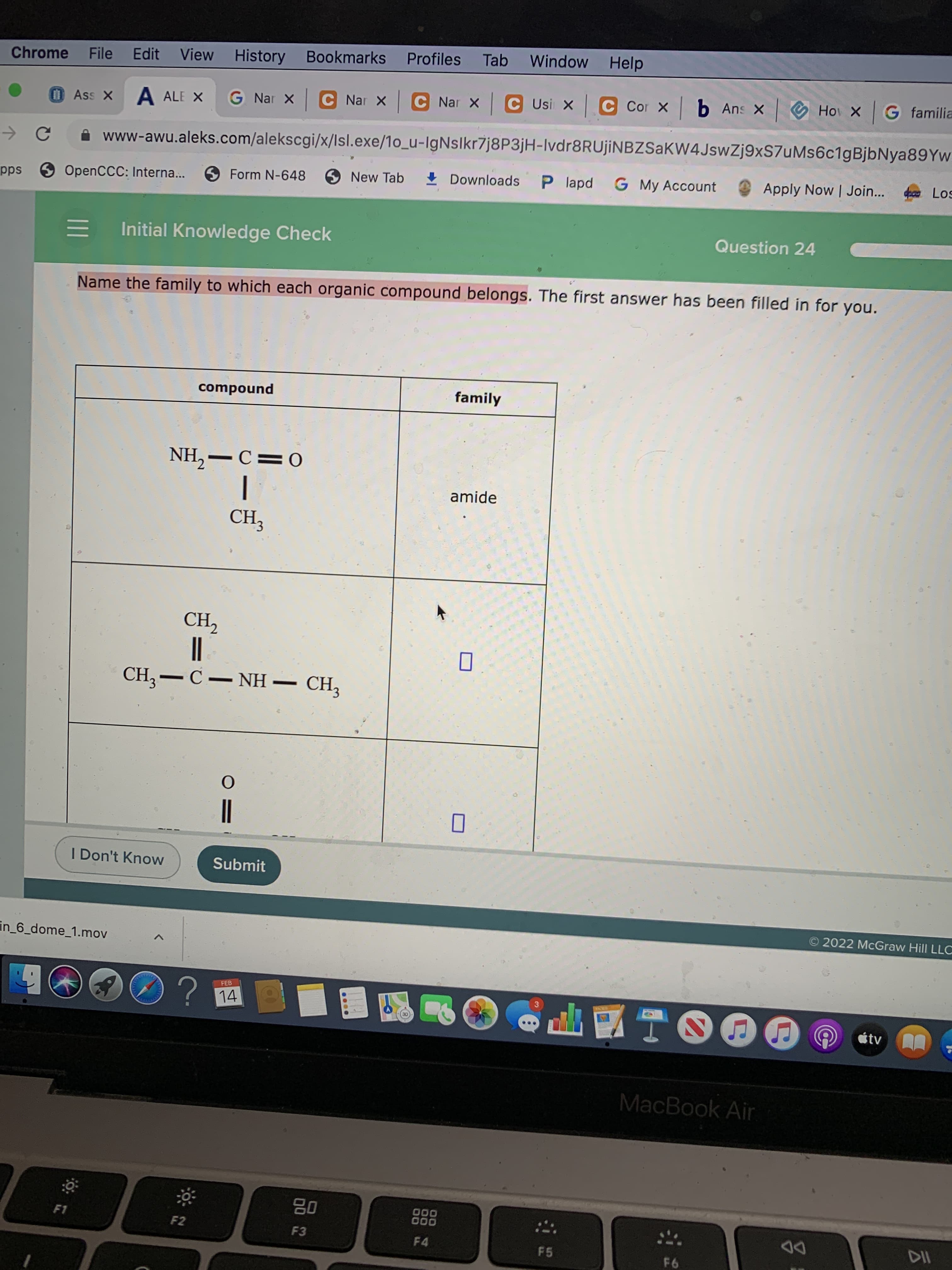
Initial Knowledge Check
Question 24
Name the family to which each organic compound belongs. The first answer has been filled in for you.
compound
family
0=ɔ- HN
amide
||
CH,-C- NH – CH,
|
|
||
I Don't Know
Submit
O 2022 McGraw Hill LLC
in_6_dome_1.mov
FEB
14
átv
MacBook Air
08
F3
F1
000
000
F2
F4
F5
DD
F6
110
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 2 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning