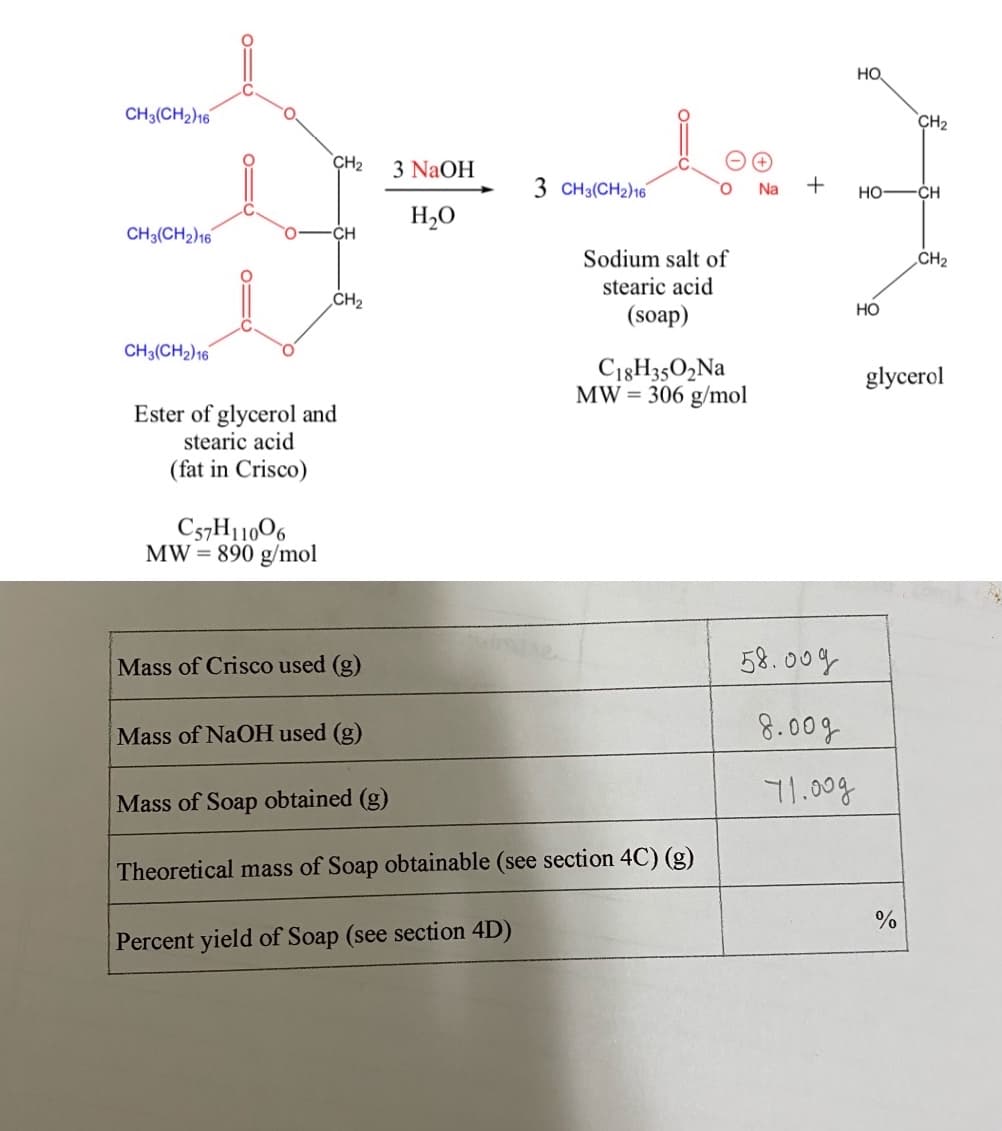
HO CH3(CH2)16 CH2 CH2 3 NaOH 3 CH3(CH2)16 + Na но CH H,O CH3(CH2)16 CH Sodium salt of CH2 stearic acid CH2 (soap) CH3(CH2)16 C18H3502NA MW = 306 g/mol glycerol Ester of glycerol and stearic acid (fat in Crisco) C57H11006 MW= 890 g/mol %3D Mass of Crisco used (g) 58.009 Mass of NaOH used (g) 8.00g 71.00g Mass of Soap obtained (g) Theoretical mass of Soap obtainable (see section 4C) (g) Percent yield of Soap (see section 4D) `우
HO CH3(CH2)16 CH2 CH2 3 NaOH 3 CH3(CH2)16 + Na но CH H,O CH3(CH2)16 CH Sodium salt of CH2 stearic acid CH2 (soap) CH3(CH2)16 C18H3502NA MW = 306 g/mol glycerol Ester of glycerol and stearic acid (fat in Crisco) C57H11006 MW= 890 g/mol %3D Mass of Crisco used (g) 58.009 Mass of NaOH used (g) 8.00g 71.00g Mass of Soap obtained (g) Theoretical mass of Soap obtainable (see section 4C) (g) Percent yield of Soap (see section 4D) `우
Chemistry: The Molecular Science
5th Edition
ISBN:9781285199047
Author:John W. Moore, Conrad L. Stanitski
Publisher:John W. Moore, Conrad L. Stanitski
Chapter4: Energy And Chemical Reactions
Section: Chapter Questions
Problem 123QRT
Related questions
Question
100%
need help with calculating theoretical mass and percentage yield
Transcribed Image Text:HO
CH3(CH2)16
CH2
CH2
3 NaOH
3 CH3(CH2)16
+
Na
но
CH
H,O
CH3(CH2)16
CH
Sodium salt of
CH2
stearic acid
CH2
но
(soap)
CH3(CH2)16
C18H35O2N
MW = 306 g/mol
glycerol
Ester of glycerol and
stearic acid
(fat in Crisco)
C57H11006
MW= 890 g/mol
%3D
Mass of Crisco used (g)
58.00g
Mass of NaOH used (g)
8.00g
Mass of Soap obtained (g)
71.00g
Theoretical mass of Soap obtainable (see section 4C) (g)
%
Percent yield of Soap (see section 4D)

Transcribed Image Text:First, the theoretical mass of soap obtainable will be calculated. This will be compared to the
experimental mass you obtained, by calculating the percent yield. Percent yield = (experimental
mass of soap/theoretical mass of soap)x100.
4A. Divide the mass of Crisco used by the molecular weight (MW) of fat in Crisco. The answer
is the number of moles of fat used.
4B. Multiply the number of moles of fat used, by three. The answer is the number of moles of
soap theoretically obtainable. (Remember that one mole of fat yields three moles of soap.)
4C. Multiply the theoretical number of moles of soap obtainable, by the MW of soap. The
answer is the theoretical mass of soap obtainable.
4D.
Divide the experimental mass of soap you obtained, by the theoretical mass of soap
obtainable, and multiply the answer by 100. The answer is the percent yield of soap.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 2 steps with 1 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning

Chemistry: The Molecular Science
Chemistry
ISBN:
9781285199047
Author:
John W. Moore, Conrad L. Stanitski
Publisher:
Cengage Learning

Introductory Chemistry: A Foundation
Chemistry
ISBN:
9781337399425
Author:
Steven S. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning

Chemistry & Chemical Reactivity
Chemistry
ISBN:
9781337399074
Author:
John C. Kotz, Paul M. Treichel, John Townsend, David Treichel
Publisher:
Cengage Learning