Q: What makes two-pot synthesis method an appropriate technique for the synthesis of Sudan I?
A: Sudan I is a organic dye.It is used as colouring agents. It is orange red dye used to colourise…
Q: What would this reaaction yleld? 1) Mg /ether Br OCH, 2) H,O/ H* Ma OH NO a)I only b) II + I c) III…
A:
Q: A coffee-cup calorimeter contains 50 mL of water with an initial temperature of 24.8°C 1.972 grams…
A: Given :- Initial temperature of water = 24.8° C Volume of water = 50 mL Mass of metal = 1.972…
Q: A batch of 500 ?? of ??? dissolves in water to form a saturated solution at 350 ? (77 °?) where the…
A:
Q: Vhich of the following gases has the greatest density at 1.0 atm ) CF, (88 g/mol) 3) CO (28 g/mol)…
A: We will determine the highest density among these molecules at standard condition.
Q: Which dissolution process would be most exothermic? a. KCl in H₂O b. CH₃CH₂OH in CH₃OH c. C₂H₆ in…
A: In this question, we will see the most exothermic process when solute dissolved in solvent. We know…
Q: Show how this cyclic ether compound could be prepared from an alkyl halide compound in an SN2…
A:
Q: Air conditioners dry the air while they cool the air. Suppose an air conditioner is used to cool a…
A: Given, Dimensions of the room = 7.73m x 11.47m x 2.30m Outside temperature = 37 °C Vapour…
Q: What would be the curly arrow process for the removal of PPh3
A:
Q: MENO2/ KOH Ph Benzaldehyde
A: Here we are required to predict the product of the reaction
Q: What would this S2 reaction glve as a major product ? KCN H,C- CH (2S,3R) ČN CI a) an optically…
A: A multiple choice question based on introduction to organic chemistry that is to be accomplished.
Q: 1. Determine the % w/w of a solution with 25.0 g of NaCl dissolved in 100 g of water. 2. What is the…
A:
Q: watch molecules cam $om hydoogen bonds with wateE? CH,CHO CHROH Hてつ
A: Given, Which molecules can form hydrogen bonds with water CH3OH C2H6 CH3(CH2)3COOH CH3CHO
Q: Adding a nonvolatile solute to a liquid will cause boiling point depression and freezing point…
A: Given Statement, Adding a nonvolatile solute to a liquid will cause boiling point depression and…
Q: Given
A: ✓1-Breadhave comparatively more gluten and cake have least among the three,so bread have highest…
Q: cell diagram of fluoride ion selective electrode
A:
Q: write out the equilibrium expression for the mineral dissolution of Al(OH)3 in water
A:
Q: 10) To make 50.0 mL 0.0107 M Mgl2 (molar mass 278.11 g/mol) solution, how many grams of Mgl2 do we…
A: Given, Molarity of MgI2 Solution = 0.0107 M = 0.0107 mol/L Volume of the solution = 50.0 mL = 0.05 L…
Q: A student measures the potential of a cell made up with 1 M CuSO4, in one solution and 1 M AgNO3, in…
A:
Q: An aliquot of 10.0mL was removed from the vinegar and diluted to 50mL to prepare what is solution A.…
A: Vinegar contains monoprotic weak acetic acid, CH3COOH(aq) that undergoes neutralization reaction…
Q: Air conditioners dry the air while they cool the air. Suppose an air conditioner is used to cool a…
A: volume of the room = 7.73 m * 11.47m * 2.30 m = 203.92m3 1m3 = 1000 L volume = 203920 L Temperature…
Q: The rate constant of a certain reaction is known to obey the Arrhenius equation, and to have an…
A:
Q: If you are using a NORMAL phase HPLC, predict the order of elution of 1. n-hexane, n-hexanol and…
A:
Q: The boiling point of ethanol, CH3CH2OH, is 78.500 °C at 1 atmosphere. Kp(ethanol) = 1.22 °C/m In a…
A:
Q: 535 mL of HCl(g) at 26.5 °C and 747 torr was dissolved in enough water to make a 625 mL solution.…
A: Given, 535 mL of HCl(g) at 26.5°C and 747 torr was dissolved in water make a 625 mL solution the pH…
Q: 1. The oxidation of nitrogen oxide produces nitrogen dioxide, as shown below: 2NO (g) + O2 (g) →…
A: 1.here we are required to find the number of moles of product formed
Q: What volume ( in mL) of. 0.3050M HBr is required to neutralize 40mL of 0.8000m NaOH?
A: The volume of HBr required is given below
Q: The pK, of CH3CH;CH2OH is than that of CH3CH2CH2SH.
A: Here, we have to find whether the pKa of CH3CH2CH2OH is greater or lesser than that of CH3CH2CH2SH.…
Q: What is the product of the reaction of HBr with (Z)-2-hexene Oa. H. Br CH3-C-C-CH,CH,CH3 нн Ob. H.…
A:
Q: 1. O3 (excess) 2. DMS
A: Given,
Q: 2. In a titration of 50.00 mL of 0.05000 M formic acid, HCOOH with 0.1000 M potassium hydroxide,…
A:
Q: Deteremine the enthalpy of the following reaction: 1. NH + Oza) N + H,O) O-241.80 kJ O-1267.2 kJ…
A:
Q: Write an expression that gives the equilibrium concentration of H,O in
A:
Q: 3. An unknown NiCl2 solution is diluted by mixing 2.00 mL of the original sample with 3.00 mL of…
A: In the given question we have to calculate the original concentration of NiCl2 Given data: Optical…
Q: What mass of carbon dioxide is produced from the complete combustion of 3.20×10−3 g of methane?…
A: Given, Mass of methane (CH4) gas react = 3.20 × 10−3 g mass of carbon dioxide (CO2) gas produced =…
Q: PPH3 OC, H в + R H. PPH3 A CI
A:
Q: A student measures the volume of her bag as 1274.7 mL. Room temperature is 21.0 °C. The atmospheric…
A: Dear student, As you have asked multiple parts questions but according to guidelines we will solve…
Q: How did you get 2.69 x 10-12 ? When I did the caculation I got -5.16 x 10-22
A:
Q: Similar to teams of ballplayers, elements have symbols (abbreviations or logos) and are arranged in…
A: Element name Element symbol Group number Period number Chlorine Cl 7 3 Calcium Ca 2 4 Radium…
Q: Determine the pH given the following values: a. [H* ] = 1 x 10-13 b. [H*] = 1.58 × 10-9 c. [OH] = 2…
A: 3.) pH can be calculated using formula : pH = - log[H+] If [OH-] is given , then we first…
Q: Explain per step. Given the MS spectra below, show the fragmentation pattern responsible for m/z…
A: Triethylamine, (CH3CH2)3N has a molar mass of 101 u.
Q: 9: Which of the following is an alkali metal? A) Zr B) Rb C) Cs D) Te 10: The symbols for Potassium,…
A:
Q: Explain why Hinsberg’s method would fail to distinguish among the three classes of amines if the…
A: This is an Nucleophilic Addition Reaction in which amines attach hinsberg reagent .
Q: PPH3 OC, `R В + CI PPH3 A R
A:
Q: CH 3- CA 3 C. Br C. and Br H Br H Br di astere omers I'
A: Stereisomers are those which have same molecular formula but different arrangement of atoms in…
Q: Q.2 Choose the correct answer from the following? 1- In all chromatographie separations the sample…
A: Introduction :- Chromatography technique is a process used to seperate mixtures. In this technique…
Q: Exercises: Solve the following problems. Show a complete solution. 1. Determine the % w/w of a…
A: How much sugar should be added in 2000 mL water to have 30% (w/v) sugar solution?
Q: A buffer solution is a good example of the common ion effect in solution. True or False?
A: A buffer is formed by the mixing of a weak base with its conjugate acid or mixing of weak acid with…
Q: What is the pH of a buffer solution containing 0.50 M acetic acid and 0.50 M sodium acetate? The…
A:
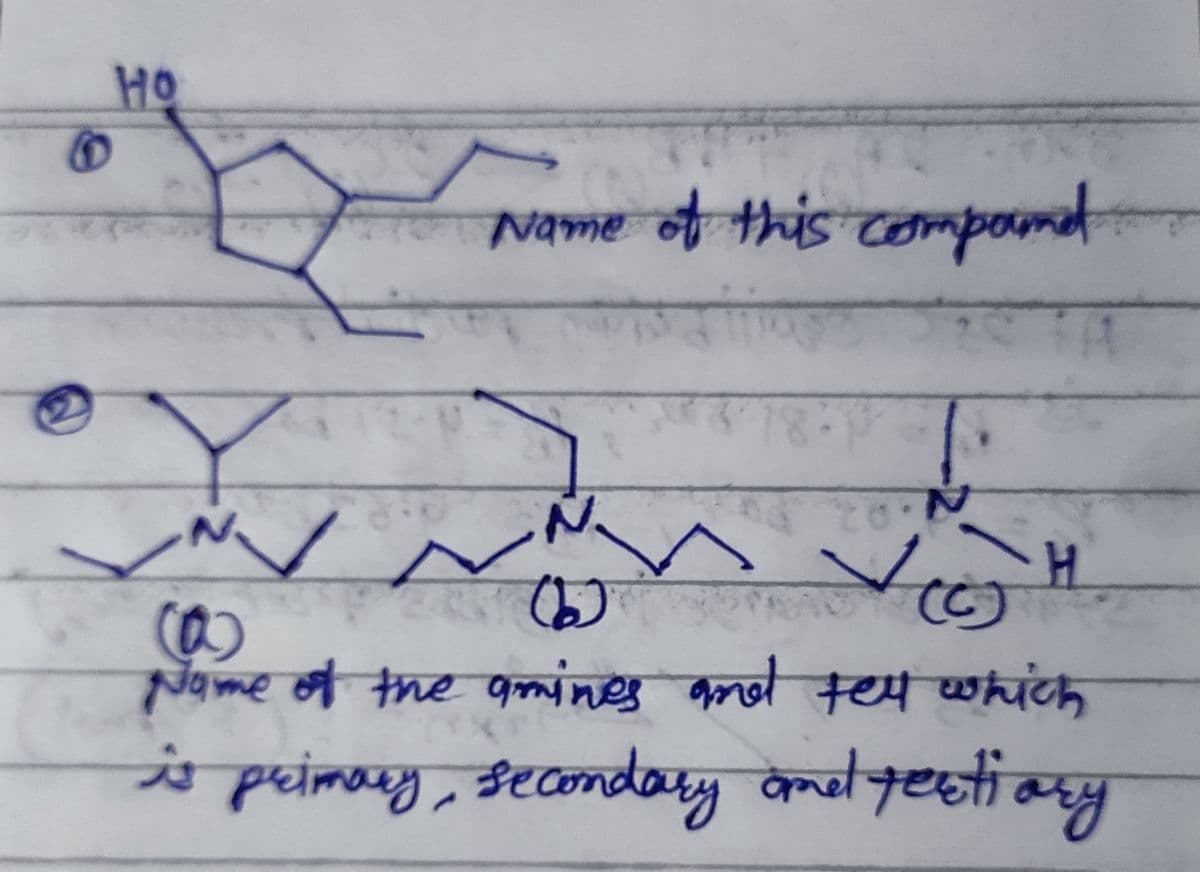
Q: Give the IUPAC name for each of the following compounds: 1. Br a) (CH3);C-COOH b) CH,-СH-CH,-соОН…
A:
Good hand written explanation
ASAP
Step by step
Solved in 3 steps with 3 images

- Calculate the index of hydrogen deficiency of this compound Q.) Ascorbic acid (vitamin C), C6H8O6molecular weight of H2O is 18 but his boilling point 100C, WHY? *The dipole moment of bromobenzene is 5.17 × 10−30 C m and its polarizability volume is approximately 1.5 × 10−29 m3. Estimate its relative permittivity at 25 °C, when its mass density is 1491 kg m−3.
- The NMR spectrum of bromocyclohexane indicates a low field signal (1H) at δ 4.16. To room temperature, this signal is a singlet, but at -75 ° C it separates into two peaks of unequal area (but totaling one proton): δ 3.97 and δ 4.64, in ratio 4.6: 1.0. How do you explain the doubling in two peaks? According to the generalization of the previous problem, what conformation of the molecule predominates (at -75 ° C)? What percentage of the molecules does it correspond to? Solve all parts otherwise down vote and hand written solutionCan you give an example of homotopic, heterotopic, enantiotopic, and diastereotopic for compounds?Oxidation of citronellol, a constituent of rose and geranium oils, with PCC in the presence of added NaOCOCH3 forms compound A. A has amolecular ion in its mass spectrum at 154 and a strong peak in its IRspectrum at 1730 cm−1, in addition to C—H stretching absorptions.Without added NaOCOCH3, oxidation of citronellol with PCC yieldsisopulegone, which is then converted to B with aqueous base. B has amolecular ion at 152 and a peak in its IR spectrum at 1680 cm−1, inaddition to C—H stretching absorptions. a.) Identify the structures of A and B.b.) Draw a mechanism for the conversion of citronellol to isopulegone.c.) Draw a mechanism for the conversion of isopulegone to B.
- The proton resonance of the OH group is found at δ 5.80 for phenol and at δ 10.67 for 2- nitrophenol, both in dilute solution of deuterated chloroform CDCl3 . Explain.Which of (a)-(d) indicates the multiplicities for hydrogens on C1, C3, and C4 of butanone attributable to spin-spin coupling in its 1H-NMR spectrum.N-propylbenzene, C6H5CH2CH3, contains C (sp3) -H and C (sp2) -H bonds. Its IR spectrum shows strong or medium absorptions at 3085, 3064, 3028, 2960, 2931 and 2873 cm ^ -1, as well as bands below 1600cm -1. Which statement is wrong? A.) Stretching of the C (sp3) -H bonds results in absorptions at lower wave numbers than the stretching of the C (sp2) -H bonds. B.) The absorptions at 2960, 2931 and 2873 cm ^ -1 are assigned to stretching of the C (sp3) -H bonds. C.) The absorptions at 3085, 3064 and 3028 cm ^ -1they are assigned to stretching of the C (sp2) -H bonds. D.) Each absorption can be assigned to the stretch mode of a particular bond in the propylbenzene molecule.

