Q: Using what is/are produced, which reaction or process does not form a new element? O B-10 combining ...
A:
Q: Calculate the number of moles in a 7.89 kg sample of aspirin.
A: No. Of moles = Given mass/molar mass
Q: 4) Consider a solution of 1.4 m of HN3 (molar mass=63.0 g/mol) its density=1.15 g/ml. Calculate the ...
A: Given: The molality of the solution (m)= 1.4 The molar mass of the solute= 63.0 g/mol The density of...
Q: Miller indices
A:
Q: For the reaction below, 0.333M A and 0.500M B are placed in an container. At equilibrium, the concen...
A: We have to calculate concentration of B at equilibrium.
Q: what is the strongest intermolecular forces in CBr4? A. Hydrogen bonds B. Ion-dipole forces C. C...
A: Given Molecule , CBr4
Q: How many grams of Ag would be deposited from an aqueous solution of Ag+ with a current of 2.534 A af...
A: Molar mass of Ag = 108g/mol Reaction takes place:- Ag+ + e- ------> Ag current , I = 2.534 A Ti...
Q: Propose the structure of the following: a. An alkane, C6H14 b. A crylic saturated hydrocarbon, C6H12...
A: Hydrocarbons are organic compounds containing C-H bonds. There will be hetero atoms to form differen...
Q: For each equilibrium, label the stronger acid, stronger base, weaker acid and weaker base. For each...
A: We can differentiate stronger acid, stronger base, weaker acid and weaker base by knowing their pKa ...
Q: 7
A: When a non-volatile solute is added to a solvent, the freezing point of a solvent decreases and this...
Q: 20) Cu²+is an ion of copper. How many nucleons and charged particles are present in the ion? A. Nucl...
A: Nucleon is the collective term for protons and neutrons. Nucleons are the particles found in the nuc...
Q: NaHCO3 + HC2H3O2 o NaC2H3O2 + H2O + CO2 Balanced Equation: Total Ionic Equation: Net Ionic Equati...
A: Since you have asked multiple question, we will solve the first question for you.If you want any spe...
Q: Answer True or False: If an acid has pKa of 2.1, it is a strong acid. ________ HCl and HNO3 are b...
A: pKa = 10-pH, a strong acid or base is defined by its ability to furnish H+ or OH- solution.
Q: How many amperes are required to deposit 0.166 grams of iron metal in 435 seconds, from a solution t...
A: We have 0.166 grams Fe deposit. 435 seconds time is used . We have to calculate the current passed. ...
Q: Name and Draw the structures of all possible chemical (Electrophilic aromatic substitution) reaction...
A: Methyl group is electron donating group which is ortho and para Directing group.
Q: Calculate the energy and the frequency of the red light emitted by neon atom with a wavelength of 68...
A: Answer :- a) The frequency of the red light emitted by neon atom with a wavelength of 680 nm = 4.41...
Q: Without consulting the tables, what sign do you expect for the change in entropy (delta S) for the f...
A: Correct answer is (d) positive. Number of moles of gases are increased with proceed of reaction and ...
Q: A reaction has delta H = –56.3 kJ and delta S = +132 J/K. At 855℃ what is the value of delta G? Gi...
A: Ans
Q: DRAW A COVALENT BOND FOR LiBr AND SI2
A: LiBr ( lithium bromide) SI ( sulfur didiodide ) Covalent bond :- A covalent bond is a chemical bond ...
Q: Calculate the expected pH of a solution created by mixing 30.00 ml of 0.125 M Na(OAc), 20.00 ml of 0...
A: HCl will react with salt and mole of acetic acid will increase .
Q: Consider the Beer's Law equation: A=εbc . Give the correct description of the variable AND units we ...
A: Answer: Beer's law relates the optical attenuation of a physical material containing a single attenu...
Q: You dissolve 30.0g of sodium sulfate (Na2SO4(s)) into 300mL of water. Calculate the moral concentrat...
A: Molar concentration/Molarity: The number of moles of the solute dissolved per liter of the solution ...
Q: A 20.0 L container at 303 K holds a mixture of two gases with a total pressure of 5.00 atm. If there...
A: Volume - 20 L Temperature - 303k Pressure - 5 atm Moles of gas A - 1.64 Moles of gas B ?
Q: What is the pH of a solution that has a OH- concentration equal to 1.3x10-10 M?
A:
Q: What happens if the temperature is too high in the selective non-catalytic reduction (SNR) technique...
A: Selective non catalytic reduction (SNCR) is a combustion which is post emissions control technology ...
Q: Determine the pH when 48.0 mL of 0.100 M NaOH is added to 50.0 mL of 0.100 HCl
A: Whenver an acid and base reacts with each other than they form salt and water and the process is cal...
Q: w is negative when work is done on the system. Select one: True False
A: Using thermodyamic sign convention we can answer this.
Q: A sample of N2(g) has a volume of 16.7 L at STP. What is the pressure of N2(g) when the volume and t...
A:
Q: van der Waals, Berthelot, Dietrici or the virial equation . a. try to represent the...
A: All these three equations relate the behaviour of real gases. They do not obey ideal gas laws.
Q: 4.5 g of LiNO3 from a 15 %(m/m) LiNO3 solution Express your answer to two significant figure...
A:
Q: a) Balance the following redox reaction in acid by separating the skeleton process into half-reactio...
A:
Q: What volume of 0.65 M NaOH is required to neutralize 40.0 mL of 0.25 M HCl?
A:
Q: Calculate the pH of the solution. Assume 1.0 L of solution. You must identify the type of solution b...
A: There are two types of bases/acids. One is strong base/acid which is completely dissociable in into ...
Q: A buffer contains 0.175 moles C2H5NH3NO3 and 0.225 moles C2H5NH2 in 800.0 mL water.Kb (C2H5NH2) = 4....
A: Given: 0.175 moles C2H5NH3NO3 0.225 moles C2H5NH2 V = 800.0 mL Kb (C2H5NH2) = 4.3 x 10-4
Q: Friedel crafts Acylation of P-cresol?
A: The structure of the p-cresol is as follows:
Q: 21. Calculate the percent atom economy for the following catalytic hydration reaction. Hint: you can...
A: Atom economy has been calculated below
Q: Predict the compound that is expected to be the most soluble in octane, C3H18? О КОН O CH3CH3 33 O H...
A: We are to check which of the given compounds is most soluble in octane. Given options are KOH, CH3C...
Q: Determine the value of AH° in kJ for the following reaction: 2SO2(g) + O2(g) 2SO3(g) Select one: O a...
A:
Q: Identify the base and conjugate acid in the equation. Explain in detail how you came to your conclus...
A: According to the Brønsted-Lowry concept of an acid-base :- An acid is a proton donor A base is a ...
Q: After 42 years, what mass remains of a 200.0 g sample of a radioactive isotope with a half-life of 8...
A:
Q: 2-) You may have noticed that the "solvent of choice" for many organometallic compounds such as Grig...
A: Following are the reason of solvent choice in case of Grignards reagent .
Q: Given: F2(2) + 2 e° → 2F (aq) E° = +2.87 v Hg²+ + 2 e → (aq) Hga E° = +0.87 v Calculate AG° for: Hga...
A: Given is F2 + 2e– ——> 2F– E° = + 2.87 v ......eq(1) Hg+2 + 2e– ——> Hg E° = 0....
Q: Balance the following redox reaction (Give the final overall balanced equation): CrO42- + I- → Cr3...
A: Given reaction is ., CrO42- + I- ---------> Cr3+ + I2
Q: How many moles of nitrogen monoxide (NO) are produced from 28.0g of NH3 and 45.0g of O2? 4 NH3 + 5 O...
A: Given 4 NH3 + 5 O2 → 4 NO + 6 H2OMass of NH3= 28 gMolar Mass of NH3 = 17 g/moleMoles of NH3 = Ma...
Q: Identify the major product(s) for the following reaction. Select all that apply. If the reaction doe...
A: When Toluene is treated with N - Bromo succinimide ( NBS ), in presence of sunlight, then there is b...
Q: Write the oxidation number (same as oxidation state) for the designated atoms (in boldface and under...
A: To find oxidation state.
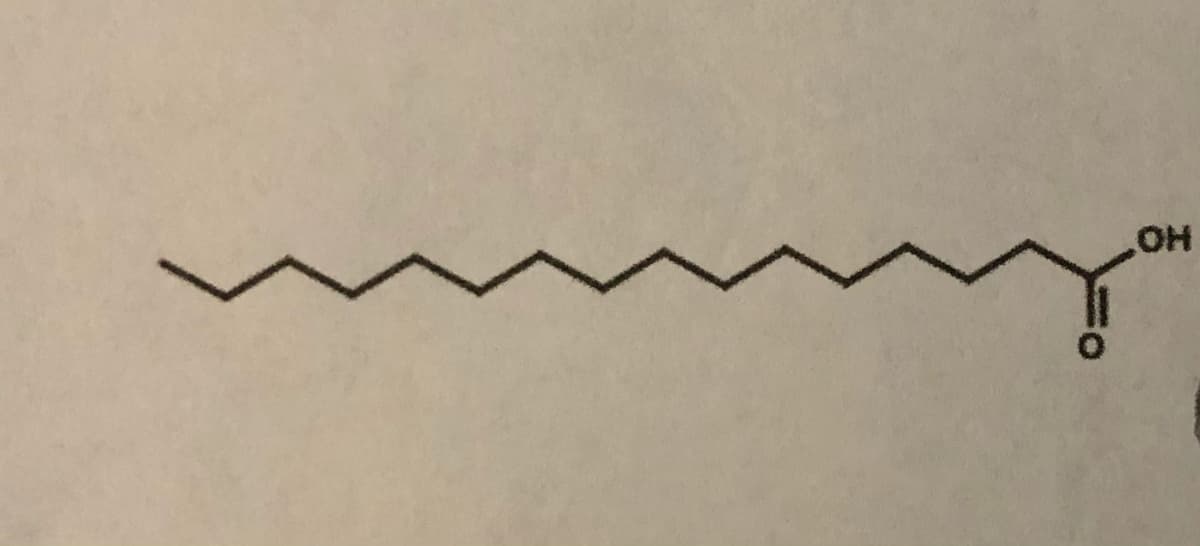
Q: Chemistry Question
A:
Q: W what is q? in biolosy a.) indicates the direction Of heat flow b.) indicates thapy en C.) indicate...
A: The term "q" is one of the variables of thermodynamics. When a thermodynamic process is going on the...
Q: Determine the shortest frequency of light required to remove an electron from a sample of Ti metal i...
A: Given; The binding energy of Ti=3.14×103 KJ/mol=3.14×106 J The binding energy of 1 electron of Ti=3....
Q: experiment on the table below. Calculate the H* and OH concentrations of these solutions, and indica...
A: Since you have asked a question with multiple sub-parts, we will solve first three sub-parts for y...
Step by step
Solved in 2 steps with 1 images


