Q: ů. + Zn(Hg) + HCI Fo? AICH HCI ?HC
A:
Q: Write the conjugate acid of each of the following bases: C. HCO3
A: Bronsted-Lowry Conjugate acid-base pairs : The pairs consists of two substances that differ only by…
Q: or rotation about its C2-C3 bond? Energy (kcal/mol) 5 3 N 0.9 -180 5.0 -120 1.8 5.0 0 torsional…
A: Question 11: According to the question, we need to determine which organic compound which give the…
Q: CH3 CH3 H HAI H Н Edit Drawing CH₂ CH3
A: Lithium aluminum hydride is used as a reducing agent.
Q: CH3CH₂OH (shown below) is soluble in water. Draw a molecular level picture that represents after…
A: The alcohols are soluble in water because they form hydrogen bonding with water molecules. Due to…
Q: QUESTION 20 A sample run through a GC gave a peak with retention time of 2 431 minutes, a peak…
A:
Q: A beaker contains 100. mL of the buffer solution. What is the resulting pH when 50.0 mL of 0.4 M…
A: Given the total volume of the buffer solution, V = 100 mL Molarity of H2PO4-(aq) in the buffer…
Q: What is the pH at the equivalence point when Jeremiah titrates the NaOH and HCIO3? HCIO3 + NaOH →…
A: Here, we have to find the pH at the equivalence point when NaOH is titrated with HClO3.
Q: Rimantadine was among the first antiviral drugs to be licensed in the United States to use against…
A:
Q: Which of the following solutions of strong electrolytes contains the largest number of ions? O 150.0…
A:
Q: A 1 gallon bottle of vinegar is 4.8% (v/v) acetic acid. How many mL of acetic acid are in the…
A: Given, The volume of acetic acid present in 1 gallon bottle of vinegar that contain 4.8% (v/v)…
Q: 4 CI IS - Consider the following structure. Convert the Newman projection to a bond-line formula…
A: -> In bond line formula there are not seen any carbon and hydrogen in the structure . -> For…
Q: Balance the following equations and classify the reactions as decomposition, combination,single-…
A: We will use hit and triel method to balance the chemical equations.
Q: What is the IUPAC name of this compound? H Handwritten only Otherwise down vote, Explain very well.
A: In nomenclature double bond gets lowest numbering and branching gets lowest numbering.
Q: An example of a heterogeneous catalyst is: a. Wilkinson’s catalyst for the hydrogenation of…
A: heterogeneous catalysis is that catalysis where the phase of catalyst is different from that of…
Q: Buffer : HC₂0/C₂02- You are trying to create a pH 4.35 buffer using HC2O4- and C2O4-2. The Ka of…
A:
Q: Be sure to answer all parts. The dissociation of molecular iodine into iodine atoms is represented…
A: Kc = 3.80× 10-5 mol of I2 = 0.0461 Volume = 2.28 L Temperature = 1000 K
Q: A benzoic acid and sodium benzoate solution has the concentrations listed below the reaction. What…
A: Given, Concentration of the benzoic acid = [HC7H5O2] = 0.25 M Concentration of the sodium benzoate =…
Q: How many of the following statements are true. 1) Aromatic compounds are especially stable. 2)…
A: Please see explanation
Q: Predict the major product(s) for the following reaction: ? H 1) PhMgBr 2) H₂O+
A:
Q: Consider the following addition reaction below. Can the organic starting material and the product be…
A: IR spectroscopy is used to study and identify chemical substances or functional groups in all three…
Q: The density of gasoline is 0.774 g/cm3. How much does 25.5 gallons of gasoline weigh, in kilograms?
A:
Q: What is the IUPAC name of this compound? H Other Exp
A: Write IUPAC name of the given structure ?
Q: Nitrite concentration is commonly determined by interpolation method using spectrophotometry (an…
A:
Q: Given the parent compound, draw the mass spectrum fragment that is observed at m/z 43. Draw the…
A: In this question, we will draw the fragment which includes all the hydrogen atoms and positive…
Q: are these enantiomers, diastereomers, constitutional isomers, or not isomeric. I F II
A: In this question, we will see the relationship between the given two alkene compounds means these…
Q: Balance the following aqueous reactions: a) Se2C12 → H2SeO3 + Se + HCI
A:
Q: 9:20 am HW4, Ch14 Q1. at: Initial Сваде Equil H₂(g) + [2(g) = 2 HI(g), K₂ = 54.3 at 430°C 0.40…
A: H2(g) + I2(g) <------> 2HI(g) Kc = 54.3 Initial concentration of HI = 0.40 M
Q: Question 7 of 25 13.06 a PMBO PMBO > Tertiary halide Tertiary alcohol Secondary halide Primary…
A:
Q: Consider the following rotational-vibrational line spectrum: a. D b. G Vight 22 which peak…
A: The given problem is from the rotational vibrational spectroscopy. From the concept of P , R branch…
Q: A 0.8798 -g sample of Al2(CO3)3 decomposed with HCI; the liberated CO2 (MW = 60.0g) was collected on…
A: Here we are required to find the percentage of Al in the sample.
Q: A 0.7875 -g portion of manganese dioxide (MnO2, MW 86.94 g/mol) was added to an acidic solution in…
A:
Q: What is the mass percent of potassium chloride when 9.35 grams of potassium chloride is mixed into…
A: Mass percent = (mass of solute/ mass of solution) x 100%
Q: Is the answer to a.) 1?
A: We have to answer the given options. The required answers are given below a) The required product is…
Q: Which peak (AE) is made from the It's attached to the circled carbon in the molecule below. yo PPH
A: -> splitting of signal is depends upon number of surrounding non equivalent hydrogen nuclei.…
Q: The line-angle formula for uric acid is shown below. What is the geometry relative to the atom…
A: We have to predict the geometry of indicated atom.
Q: Bond tool, Multiple bonds tool, Dative Bonds tool and Wedged bonds tool are used to add shapes to…
A: The object tool has both lasso mode and a rectangle tool which is used to draw a selection around…
Q: What is the H30+ concentration in a solution that has a pH of 2.43?
A: Given data the pH of the solution= 2.43 the concentration of H3O+ ion, [H3O+]=
Q: You measured the pH of a 0.01M NaOH solution (strong base) and it was 13.0. The, you diluted this…
A:
Q: What is the IUPAC name of this compound? H Handwritten only Otherwise downvote 1 Explain very well.
A:
Q: Be sure to answer all parts. A quantity of 0.29 mole of carbon dioxide was heated to a certain…
A: Given : Moles of CO2 = 0.29 mole Average mole mass of gases at equilibrium= 34.5 g/mole Total…
Q: A solution of 25.0 mL of 0.40 M H2SO4 is titrated with 0.50 M of LiOH. H₂SO4 + 2LiOH →>> Li₂SO4 +…
A: Given, H2SO4 + 2LiOH → Li2SO4 + 2H2O Molarity of H2SO4 solution = 0.40 M = 0.40 mol/L Volume of the…
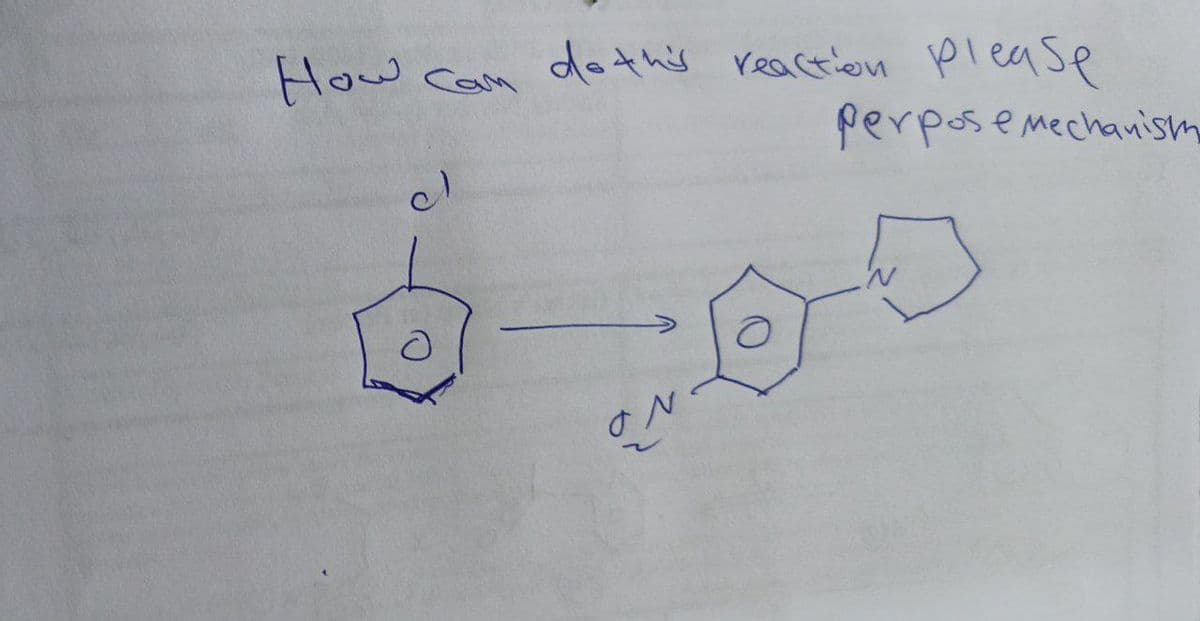
Q: 2. How to do the following reactions. Please propose mechanism. 14 OH 5) o 0₂N
A: Note- The given question is a multiple question, hence I solved first question according to rules of…
Q: Which of the following can complete the reaction below? O Bry/Febry O NBS/PMC₂2 Hy, Pt. Ethanol at…
A: The given reaction is hydrogenation of benzene moiety. Hydrogenation can be done by hydrogen in…
Q: QUESTION 14 What is the [H3O+] in a solution with [OH] = 1 × 10-12 M? O A) 1 x 10-12M OB) 1x 10+2M…
A: Question 14 pH = - log [H3O+] [H3O+] = 10-pH pOH = - log [OH-] [OH-] = 10-pOH pH + pOH = 14
Q: a cube of ice has mass= 0.0051 lb , determine the volume of this cube in mL if the density of ice is…
A: Given, Mass of a cube of ice = 0.0051 lb. The density of ice = 0.920 g/cm3 The volume of ice cube…
Q: Crie Camie the moderntary Gine gurmaay with lines [AEN] = [E₁-E₂] Polor and mon polar with reason…
A:
Q: How many of the following statements are true )Aromatic compounds are especially stable 2)…
A: Yes ,aromatic compounds are very stable. Antiaromatic compound do not follow Huckel's rules so there…
Q: the initial temperture was 32.5 degrees celsius, what is its final temperture? {density of…
A: Specific heat capacity is amount of heat required to raise temperature of one gram of a substance by…
Q: MAN If you want to consume 75 mg of vitamin C, how many mL of cranberry juice n you drink? Cranberry…
A:
Step by step
Solved in 2 steps with 2 images

- Solve correctly please. (Gpt/Ai wrong ans not allowed)Plastic photochromic sunglasses are based on the following reversible rearrangement of a dye inside the lenses that occurs when the lenses are exposed to sunlight. The original dye absorbs UV light but not visible light and is thus colorless, while the rearrangement product absorbs visible light and is thus darkened. (a) Show the mechanism of the rearrangement. (b) Why does the rearrangement product absorb at a longer wavelength (visible light) than the original dye (UV)?youn didn't explain how the mechanism effects the product formation.

