Chapter13: Titrations In Analytical Chemistry
Section: Chapter Questions
Problem 13.10QAP
Related questions
Question
How do I solve for the freezing point depression?
Transcribed Image Text:Afr
R Fou
b firs b firs firs , hist
b
Ь Ans
> cou
lab
Lab
ho h
Ans
Pas
gm
Ne
+
Ô https://newconnect.mheducation.com
Dashboard UF Application for.
H Mail - Garmar Sutto..
A Macmillan Learning.
O Other favorites
Lab Data
SUBMIT
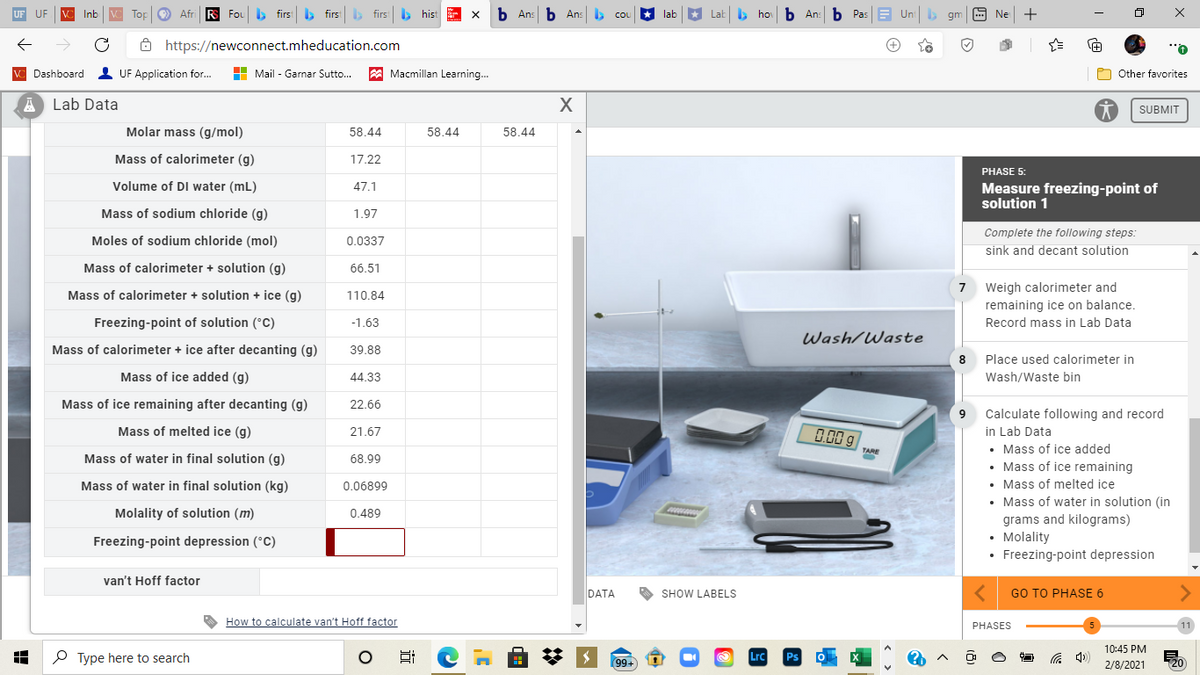
Molar mass (g/mol)
58.44
58.44
58.44
Mass of calorimeter (g)
17.22
PHASE 5:
Volume of DI water (mL)
47.1
Measure freezing-point of
solution 1
Mass of sodium chloride (g)
1.97
Complete the following steps:
Moles of sodium chloride (mol)
0.0337
sink and decant solution
Mass of calorimeter + solution (g)
66.51
Weigh calorimeter and
remaining ice on balance.
Record mass in Lab Data
7
Mass of calorimeter + solution + ice (g)
110.84
Freezing-point of solution (°C)
-1.63
Wash/Waste
Mass of calorimeter + ice after decanting (g)
39.88
Place used calorimeter in
Mass of ice added (g)
44.33
Wash/Waste bin
Mass of ice remaining after decanting (g)
22.66
Calculate following and record
in Lab Data
Mass of melted ice (g)
0.00 g
21.67
• Mass of ice added
• Mass of ice remaining
• Mass of melted ice
• Mass of water in solution (in
TARE
Mass of water in final solution (g)
68.99
Mass of water in final solution (kg)
0.06899
Molality of solution (m)
0.489
grams and kilograms)
• Molality
• Freezing-point depression
Freezing-point depression (°C)
van't Hoff factor
DATA
SHOW LABELS
GO TO PHASЕ 6
How to calculate van't Hoff factor
PHASES
10:45 PM
P Type here to search
99+
2/8/2021
20
立
Transcribed Image Text:O Afr
R Fou
> first b firs firs b hist
b Ans
b Ans
b COu
lab
Lab
ho h
Ans
Pas
gm
Ne
+
Ô https://newconnect.mheducation.com
Dashboard UF Application for.
H Mail - Garnar Sutto.
A Macmillan Learning.
O Other favorites
CHEMISTRY • DETERMINE VAN'T HOFF FACTOR
SUBMIT
Lab Data
PHASE 5:
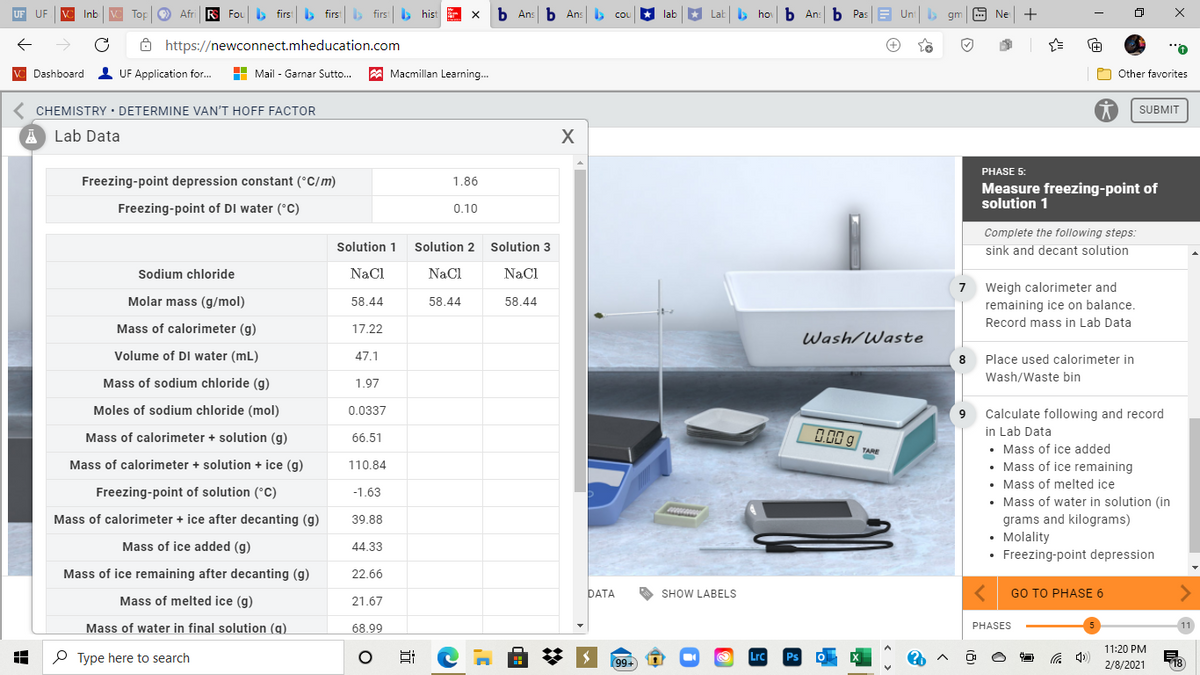
Freezing-point depression constant (°C/m)
1.86
Measure freezing-point of
solution 1
Freezing-point of DI water (°C)
0.10
Complete the following steps:
Solution 1
Solution 2
Solution 3
sink and decant solution
Sodium chloride
NaCl
NaCl
Nacl
Weigh calorimeter and
remaining ice on balance.
Record mass in Lab Data
7
Molar mass (g/mol)
58.44
58.44
58.44
Mass of calorimeter (g)
17.22
Wash/Waste
Volume of DI water (mL)
47.1
Place used calorimeter in
Wash/Waste bin
Mass of sodium chloride (g)
1.97
Moles of sodium chloride (mol)
0.0337
Calculate following and record
in Lab Data
Mass of calorimeter + solution (g)
0.00 g
66.51
• Mass of ice added
• Mass of ice remaining
• Mass of melted ice
• Mass of water in solution (in
TARE
Mass of calorimeter + solution + ice (g)
110.84
Freezing-point of solution (°C)
-1.63
Mass of calorimeter + ice after decanting (g)
grams and kilograms)
• Molality
• Freezing-point depression
39.88
Mass of ice added (g)
44.33
Mass of ice remaining after decanting (g)
22.66
DATA
SHOW LABELS
GO TO PHASE 6
Mass of melted ice (g)
21.67
Mass of water in final solution (q)
68.99
PHASES
11:20 PM
P Type here to search
99+
2/8/2021
近
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you

