Q: Fill in the gaps in the following table, assuming each columnrepresents a neutral atom. Symbol…
A: 79Br is the symbol of bromine. Its mass number, as represented by the superscript to the left of its…
Q: hexafluoride. 2.65 In the diagrams shown here, match each of the drawings with the following ionic…
A: The given compounds are Al2O3, LiH, Na2S, MgNO32. The cations are represented by green spheres and…
Q: write the chemical formula for the following compounds g. magnesium phosphide h. potassium sulfide…
A: Chemical formula for the following compounds= g. magnesium phosphide = Mg3P2 h. potassium…
Q: How many electrons does the predictable (mostcommon) ion of fluorine contain? (a) 1 (b) 4 (c) 9 (d)…
A:
Q: 5. How many hydrogen atoms in this compound? OH но OH А. 22 В. 23 Agu С. 21 D. 20
A: The given structure is an organic compound.
Q: 1s 25 2p How many 1s electrons are in the ion?
A: Given : An atomic cation configuration is given and we have to write the electrons present in 1s.
Q: 8 - How many electrons are in the XY3 ion? ( 15X 24Y) a) 88 84 87 91 Boş bırak
A: The given ion is = XY34- The atomic number of X and Y is as follows: atomic number of 15X is =…
Q: Below are some of the interesting (and in some cases toxic) compounds that people will encounter in…
A:
Q: Calcium phosphide consists of two ions chemically bonded together. What are the charges of each ion?…
A: To write the chemical formula of compoundwhich follow the following rule:-(i) write the symbols of…
Q: A sample of CH 2F 2 with a mass of 19 g contains atoms of F. 38 в) 3.3х 1024 c) 2.2 x 1023 D) 9.5 E…
A:
Q: Which ion has 38 protons, 36 electrons? CO a. Kr ОБ. Sr Oc Sr24+ O C. O d. Kr2+ O e. Sr2-
A: Given that: No of protons= 38 no of electrons= 36
Q: How many nuclei can reasonably bear the charge in each of the following ions? b. „CH2 d. :ZI
A: Resonance is delocalization of electron from one atom to another which are coplaner i.e sp2…
Q: The difference between 7°Br and 81Br is their number of: neutrons electrons protons
A: We have to predict the difference between given atoms.
Q: A compound, A,S3, contains three S atoms per formula unit and is 43.71% sulfur. What is the atomic…
A: The compound with element A and sulfur is given by the chemical formula - A2S3 There are three 3 S…
Q: CO Mo Mo OC OC
A: The complex given is,
Q: Name the following ionic compounds: 1) V₂(SO4)3 2) Zn3P₂ 3) Sr(C₂H3O2)2 4)Cu₂0 Could you also please…
A: Name the following ionic compounds: 1) V2(SO 4)3 2) Zn 3P 2 3) Sr(C 2H3O2)2 4)Cu₂O
Q: 4. The compounds given below are all fertilizers and provide nitrogen to the soil. Which of the…
A:
Q: 2. Write the chemical formula for the following compounds a. aluminum oxide, b. lead (IV) chlorite…
A: "The chemical formula of a compound is a symbolic representation of its composition" So lets see…
Q: A newly discovered element has the name Rougen, has the symbol Rg. Rougen can have the charges of…
A: The solution is as follows :
Q: 3. A. B. C. A chlorine atom has 18 neutrons and an atomic mass of 35. How many protons are in the…
A: In an atom three fundamental particles are present. Positively charged particle proton and neutral…
Q: calculate the charge of each particle Pb3(PO4)4
A:
Q: Cu has two isotopes; 63 Cu in 70 % and 65 Cl in 30%. The average atomic mass of Cu atom. Is * O 63 О…
A: Given Natural abundance (%) Atomic Mass 63Cu 70 65 65Cu 30 63
Q: The chromium (IV) ion (Cr4+) has O 52 protons and 48 electrons O 24 protons and 28 electrons O 52…
A: Correct answer is 24 protons and 20 electrons. Atomic number of Cr , Z = 24 Number of protons in a…
Q: Which of these is not a cation A NH4+ B Cl− C Mg2+ D K+
A: Introduction: Atoms are the neutral and smallest unit of element. Each atom consist of some…
Q: 44. A 2.000 g sample of element M reacts with oxygen to form 2.539 g of the metallic oxide, MO2.…
A: Given data mass of element M = 2.000 g mass of metallic oxide , MO2 formed = 2.539 g…
Q: A sample is found to contain 63.65% nitrogen and 36.35% oxygen. What is its chemical formula? O N20…
A: Emperical formula can be determined from the % of different elements present in a molecule. If…
Q: Give the name and formula of the compound formed from thefollowing elements:(a) ₃₇Q and ₃₅R(b) ₈Q…
A: (a) The atomic number of an element is written as a subscript on left side of symbol of the element…
Q: 2.77/The radii of the lithium and magnesium ions are 76 pm and 72 pm, respectively. Which compound…
A: Given, Radii of Li = 76 pm Radii of Mg = 72 pm Which has more ionic attraction = ?
Q: A sample of iron sulfide is found with the formula Feo.87S. What percentage of the iron ions are…
A: The given formula of the compound is Fe0.87S1.00. Iron(Fe) can be present in +2 or +3 oxidation…
Q: Q11/ Calculate he equivalent weight of K2SO4, if you know the atomic weight of K=39, S=32, O=16.
A: Potassium sulphate = K2SO4 --> 2K+ + SO42- molecular weight = 174 (2x39 + 32 + 4x16)
Q: calculate the charge of each particle'' Pb(CrO4)2
A: Given Pb(CrO4)2 To find The individual charges of each element in the given formula.
Q: 14. Given the information below, calculate the weighted average atomic mass (amu) of the element X.…
A:
Q: How many nuclei can reasonably bear the charge in each of the following ions? a. b. ČH2 с. d.
A: Resonance is said to occur whenever for a molecule we ccan write two or more Lewis structures which…
Q: why is that in Pb(CrO4)2, the charge of CrO4 is -2
A: The species lead Pb can have the potential to show +2 and +4 oxidation state because the outermost…
Q: The chemical analysis of a binary hydrate shows that it is composed of 27.76% Mn, 35.82% Cl and…
A: Given: Mass composition of Mn = 27.76 % Mass composition of Cl = 35.82 % Mass composition of H2O =…
Q: b(NO2)2 is an ionic compound. What are the ions of which it is composed?
A:
Q: An unkown element is a mixture of isotopes 120X and 122X. Theaverage atomic mass of X is 121.58 amu.…
A: • Two Isotopes 120X and 122X average atomic = 121.58 amu.Suppose the amount of 122X = y…
Q: 6. Verify the law of conservation of mass by using relative atomic masses for the following chemical…
A: Given reaction is : BaCl2 + H2SO4 ----------> BaSO4 + 2HCl Verification of law of conservation…
Q: 3) a) Based on the properties of molecular compounds explain why they are the main components of…
A:
Q: 4. Count the atoms for each element in the formulas. (C:N.):N NaHCO, 2H.SO. Bas(PO.). 3AI(OH). 5KNO.…
A: Welcome to bartleby !
Q: What is the charge of the copper in the compound CuCl2?
A: Charge on a atom in a molecule is determined by considering the charge of the other atoms and…
Q: A sample of iron sulfide is found with the formula Feo.87S. VWhat percentage of the iron ions are…
A: Co-ordination compounds are formed when the metal ion is bonded to the ligand which donates its…
Q: An element's most stable ion forms an ionic compound with bromine, having the formula XBr3. If the…
A: Given: 1) Ionic compound with bromine is=XBr3 2) Mass number of an element is = 225 3) Number of…
Q: An unknown element X has the following isotopes: 12°X (22.00% abundant), 128X (34.00% abundant),…
A: Given that element = X, With three isotopes. X-126, X-128 and X-130. The percentage abundance are:…
Q: calculate the charge of each particle Sn(MnO4)2
A: Charge on each particle in Sn(MnO4)2 is determined as
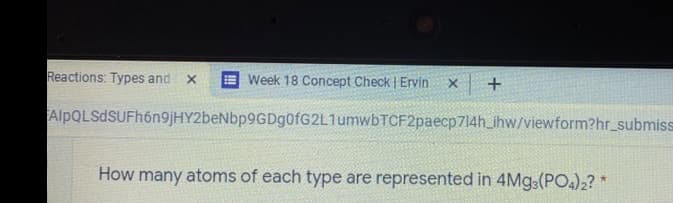
Q: atoms of each type are represented in 4Mg:(PO)2? *
A:
Q: How many atoms of each kind are represented by: 3(Hg2)3(PO4)
A: Molecular formula is a type of chemical formula that is presented and expressed by counting and…
Q: Determine the charge of manganese in MnO3
A: Oxidation state indicates the degree of oxidation for an atom in a chemical compound; it is the…
Step by step
Solved in 2 steps

- Calcium ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) Ca (s) 0 0 41.4 Ca (g) 178.2 144.3 158.9 Ca2+ (g) 1925.9 CaC2 (s) -59.8 -64.9 70.0 CaCO3 (s, calcite) -1206.9 -1128.8 92.9 CaCl2 (s) -795.8 -748.1 104.6 CaF2 (s) -1219.6 -1167.3 68.9 CaH2 (s) -186.2 -147.2 42.0 CaO (s) -635.1 -604.0 39.8 CaS (s) -482.4 -477.4 56.5 Ca(OH)2 (s) -986.1 -898.5 83.4 Ca(OH)2 (aq) -1002.8 -868.1 -74.5 Ca3(PO4)2 (s) -4126.0 -3890.0 241.0 CaSO4 (s) -1434.1 -1321.8 106.7 CaSiO3 (s) -1630.0 -1550.0 84.0 Carbon ΔHof (kJ/mol) ΔGof (kJ/mol) So (J/mol K) C (s, graphite) 0 0 5.7 C (s, diamond) 1.9 2.9 2.4 C (g) 716.7 671.3 158.1 CCl4 (l) -135.4 -65.2 216.4 CCl4 (g) -102.9 -60.6 309.9 CHCl3 (l) -134.5 -73.7 201.7 CHCl3 (g) -103.1 -70.3 295.7 CH4 (g) -74.8 -50.7 186.3 CH3OH (g)…Tu Re-order each list in the table below, if necessary, so that the atoms or lons in it are listed in order of decreasing size. Please don't provide the handwriting solutionpotassium ferrioxalate find and solve these and only show math work for Mass of C2O42-and Average Mass % of C2O42- so i can understand 12Mn0-4 + 5C2O4-2 + 16H+--> 2Mn2+ + 10CO2 +8H2O
- 5. In a mass spectrum, the peak of greatest abundance is referred to as the_________________________. Relative abundance is the unit along the y-axis ina mass spectrum. What are the units on the x-axis? _____________________.Fe ves at the ie = 0-225 Vo Por Cord. amber 4 in Ceram IC Cpt oe tiide e eT. ue aeI need help with the mass spec problome I linked a sampe example of how the queshtion needs to be answered
- Hydrogen sulfide is a gas with the odor of rotten eggs. The gas can sometimes be detected in automobile exhaust. It is a compound of hydrogen and sulfur in the atomic ratio 2:1. A sample of hydrogen sulfide contains 0.707 g H and 11.24 g S. What is the atomic weight S of relative to H? (Show Steps)Chemistry Common Isotopes and Mass spectrum of element Mo are given below Most common isotopes: Mo-98; Mo-96 Mo-95; Mo-92; Mo-97 ; Mo-100; Mo-94 Calculate the average atomic mass of Mo How the MS determined the relative intensity (Scale of 100) and Isotope abundance (%)? Why absolute intensity was not used? Can detector sensitivity, ICPMS temperature influence the relative intensity? Explain1. Sketch and label the main parts of the mass spectrometer. 2. Give detail description about how it works.

