Chapter12: Gravimetric Methods Of Analysis
Section: Chapter Questions
Problem 12.29QAP
Related questions
Question
![x OWLV2| Online teaching and I X
Final Exam List of Equations.po X PeriodicTable (1).pdf
FER
x sulfur trioxide molar
mass - Bi
o://east.cengagenow.com/ilrn/takeAssignment/takeCovalentActivity.do?locator=Dassignment-take
[References]
Use the References to access important values if needed for this question.
How many grams of Cu(OH)2 will precipitate when excess NaOH solution is added to 72.0 mL of 0.707 M Cul, solution?
Cul2(aq) + 2NaOH(aq) Cu(OH)2(s) + 2NaI(aq)
Try Another Version
2 item attempts remaining
Submit Answer
pt
pt](/v2/_next/image?url=https%3A%2F%2Fcontent.bartleby.com%2Fqna-images%2Fquestion%2Ff04dfd8c-1a56-4c24-8b87-b19a0073a8c5%2F70309ccd-f1d0-47f9-937a-978b1faf2015%2F8dv3791_processed.jpeg&w=3840&q=75)
Transcribed Image Text:x OWLV2| Online teaching and I X
Final Exam List of Equations.po X PeriodicTable (1).pdf
FER
x sulfur trioxide molar
mass - Bi
o://east.cengagenow.com/ilrn/takeAssignment/takeCovalentActivity.do?locator=Dassignment-take
[References]
Use the References to access important values if needed for this question.
How many grams of Cu(OH)2 will precipitate when excess NaOH solution is added to 72.0 mL of 0.707 M Cul, solution?
Cul2(aq) + 2NaOH(aq) Cu(OH)2(s) + 2NaI(aq)
Try Another Version
2 item attempts remaining
Submit Answer
pt
pt
Expert Solution
Step 1

Step 2
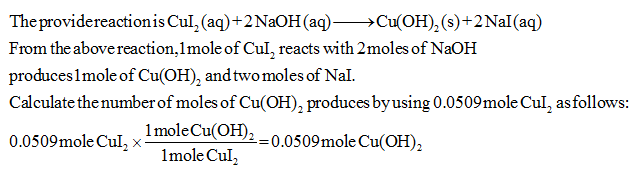
Step by step
Solved in 3 steps with 3 images

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Recommended textbooks for you


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning


Chemistry: Principles and Reactions
Chemistry
ISBN:
9781305079373
Author:
William L. Masterton, Cecile N. Hurley
Publisher:
Cengage Learning

Principles of Modern Chemistry
Chemistry
ISBN:
9781305079113
Author:
David W. Oxtoby, H. Pat Gillis, Laurie J. Butler
Publisher:
Cengage Learning

Chemistry by OpenStax (2015-05-04)
Chemistry
ISBN:
9781938168390
Author:
Klaus Theopold, Richard H Langley, Paul Flowers, William R. Robinson, Mark Blaser
Publisher:
OpenStax

Chemistry: An Atoms First Approach
Chemistry
ISBN:
9781305079243
Author:
Steven S. Zumdahl, Susan A. Zumdahl
Publisher:
Cengage Learning

Chemistry
Chemistry
ISBN:
9781305957404
Author:
Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:
Cengage Learning