Q: Symbols or atoms of what element(s) are missing from the line drawings?
A: Organic molecules:
Q: What is the IHD for the following molecule? Only enter the numeric answer.
A: Introduction :- the index of hydrogen deficiency IHD (also known as the degree of unsaturation ,…
Q: What is the most likely base peak in the mass spectrum öf the giver
A: Mass spectroscopy: It in an analytical spectroscopic method used to identify mass-to-charge ratio of…
Q: 2. 2. It %3D 不
A: From the sample given above, I can identify only 4 strains which are numbered from 1 to 4 in the…
Q: Which peak (AE) is made from the H's attached to the circled carbon in the molecule below. yo 00000…
A: The splitting of the signals in 1H NMR spectra depends on the no of hydrogens atom present in…
Q: Using a simple chemical test show how to distinguish between the pairs belc of 1) and (A) Reagent:…
A: Given compounds are: A) acetophenone ( phenylethanone) : it's a methyl ketone as carbonyl carbon is…
Q: Which one is the incorrect statement regarding mass spectrum? options: The structure likely…
A: Given are statements regarding mass spectrometry
Q: molecular weight of H2O is 18 but his boilling point 100C, WHY? * 20 points Your answer
A: Boiling point of solvent depends on the intermolecular forces holding them. Stronger the…
Q: How to interept this data to match the structure? It gives the peak/PPM and having trouble matching…
A: The given NMR data is needed to be interpreted and the each signal obtained is needed to be assigned…
Q: Match molecules to the IR spectra
A: A strong peak at ~1751 cm-1 stands for C=O bond stretching. This indicates presence of C=O bond. A…
Q: For the following structure below, select the hydrogen that would display the highest chemical…
A:
Q: Interpret the mass spectra of Methylene chloride. draw atleast 4 possible fragments
A: Mass spectrometry is used to identify the moleculer weight of the given molecule. In mass…
Q: 8. For the mass spec shown below, select the structure that matches. How can you tell? MASS SPECTRUM…
A:
Q: Please identify the compound present in the IR and MS scans. Both scans utilized the same compound.…
A:
Q: Which of the highlighted bonds absorbs at higher in an IR spectrum?
A: a.
Q: CH3 CH3 11 10 .105
A: There is plane of symmetry in the molecule. And we can see the given spectrum ( 1H NMR spectrum )…
Q: Draw a figure to illustrate a SHAPE-probing experiment.
A: the shape is shown below.
Q: Please match each structure to its spectrum. The structures are at the top of the image
A: From IR spectra we can get the idea about the functional group present in the molecule .
Q: MATCH a structure from the list below to the following IR spectra. A. HO
A:
Q: QUESTION 6 What could be the observe rotation of a 0.84 M solution composed of a sample which has a…
A:
Q: Using only the information in the pK, table below, write an estimated pK, value for each labeled H…
A: Acidic strength of a weak acid is measured interms of Ka value , Ka is called acid dissociation…
Q: Br show homotopic protons by numbering the homotopic groups. show homotopic carbon nuclei by…
A:
Q: QUESTION 4 Click the peak that corresponds to the hydrogen on the central carbon (hint: it is…
A:
Q: Tag all the primary sp carbon(s) in the structure below. If there are none, please check the box…
A: Sp3 carbon is single bonded carbon. And primary carbon means carbon has only one carbon attached.
Q: What is the relationship between Ha and H, in the following compound? a Select an answer and submit.…
A:
Q: Answer the following questions based on the mass spectrum shown below: a. What is the molar mass of…
A: a. The mass spectrum of the compound shows two peaks M+ and M+2, of almost equal intensities. The…
Q: IR Peaks for different functional group?
A:
Q: Question is attached
A: It is given a molecule of acetyl eugenol and its NMR peaks associating to which hydrogen needs to be…
Q: Draw two structures with the formula C,H,O that would give a positive iodoform test. Also draw two…
A: Given : We have to draw the structure which give positive and negative iodoform test.
Q: Identify the functional groups in the spectrum below of asprin.
A:
Q: 1 PPM
A: Number of signals ----------> Number of sets of protons in different electronic environment…
Q: Please identify the compound present in the IR and MS scans. Both scans utilized the same compound.…
A: A question based on spectroscopy that is to be accomplished.
Q: Assign each mass spectrum to the molecule on the list. Write down the molecule that corresponds to…
A: An instrumental method by which the structure of an unknown compound by analyzing its interactions…
Q: Sort the alkyl halide used to its substitution pattern: Primary Secondary Tertiary Answer Bank…
A:
Q: If your sample gave you negative absorbance values during the kinetics run, what’s the most likely…
A:
Q: MATCH a structure from the list below to the following IR spectra.
A: Infrared spectroscopy is used in order to find out the functional group present in the spectrum.
Q: When an external magnetic field is applied, what happens to the protons in a sample? a. All protons…
A: When an external magnetic field is applied, what happens to the protons in a sample, some protons…
Q: HC-CH-OH The mass spectrum below most likely belongs to which structure? Explain your answer. 45 100…
A: In mass spectrometry ,the molecule is breaked into fragments and m÷ z value is measured.
Q: Need help on Letter A for #4
A:
Q: Can you help me figure out which graphs make what compound please? Based on IR?
A: A question based on IR spectrum, which is to be accomplished.
Q: 1. For each spectrum below, analyze for the three traits: 1) What is the molecular weight? 2) Does…
A: Four mass spectra are given to be analyzed.
Q: Identify each peak (peak splitting and ppm) and identify the unknown compound. Please draw the…
A: Identify each peak (peak splitting and ppm) and identify the unknown compound.
Q: following localizec lоса
A: In the given compound label all lone pair electron and pi bond electron
Q: Given the parent compound, draw the mass spectrum fragment that is observed at m/z 43. Draw the…
A: In this question, we will draw the fragment which includes all the hydrogen atoms and positive…
Q: a mass spectrometry, hogh energy protons is use to bombard the molecule so that it will break down…
A:
Q: How many hydrogen atoms neighbor the carbon that has the protons that produced peak A? The wording…
A:
Q: Tag all the secondary sp carbon(s) in the structure below. If there are none, please check the box…
A: Carbon may be classified as primary, secondary, tertiary or quaternary depending on the number of…
Q: (image 1) mass spectroscopy: interpretation of peaks: (image 2) Carbon NMR: interpretation…
A: The three peaks 13C NMR spectrum indicates three different carbons in the organic molecule. The mass…
Q: What is the structure of the resulting confirmation after the ring flip?
A: In this question, we will draw the structure of this confirmation after ring flip. You can see…
i need answer fast ill rate up
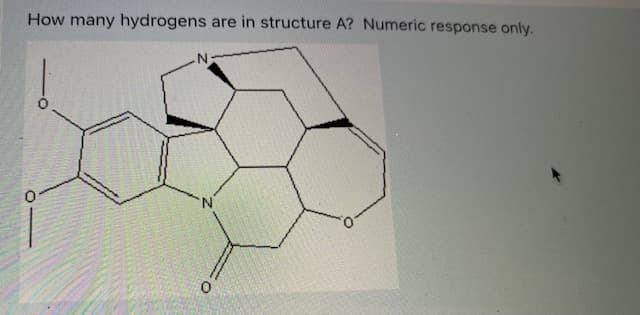
Step by step
Solved in 2 steps with 1 images

- Answer G and HWhich one is the incorrect statement regarding mass spectrum? options: The structure likely contains a chlorine atom if the (M+2)+• peak is approximately one-third as tall as (M)+• The relative heights of (M)+• peak and (M+1)+• peak indicate the number of carbon atoms A signal at M-15 indicates the loss of a methyl group and a signal at M-29 indicates the loss of an ethyl group The structure likely contains a bromine atom if the (M+2)+• peak and (M)+• are almost similar in heights In case of alcohols, a signal at m/z = 18 is produced due to the loss of neutral H2O molecule (Dehydration)Which base peak is expected in the mass spectrum of 2,2,3-trimethylbutane? Question 14 options: M-15 peak M-57 peak M-29 peak M-43 peak M-18 peak

