
Chemistry
10th Edition
ISBN: 9781305957404
Author: Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher: Cengage Learning
expand_more
expand_more
format_list_bulleted
Concept explainers
Question
Transcribed Image Text:Question 31 of 49
Submit
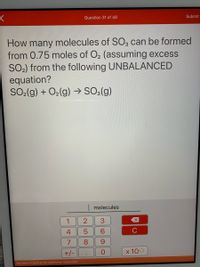
How many molecules of SO3 can be formed
from 0.75 moles of O2 (assuming excess
SO2) from the following UNBALANCED
equation?
SO2(g) + O2(g) → SO3(g)
| molecules
1
3
C
7
8.
+/-
x 100
Tap here or pull up for additional resources
LO
4.
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution
Trending nowThis is a popular solution!
Step by stepSolved in 2 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, chemistry and related others by exploring similar questions and additional content below.Similar questions
- Butane, C4H10C4H10, is a component of natural gas that is used as fuel for cigarette lighters. The balanced equation of the complete combustion of butane is 2C4H10(g)+13O2(g)→8CO2(g)+10H2O(l)2C4H10(g)+13O2(g)→8CO2(g)+10H2O(l) At 1.00 atmatm and 23 ∘C∘C, what is the volume of carbon dioxide formed by the combustion of 2.00 gg of butane?arrow_forward6. Consider the following chemical reaction: 2 Cr(OH)3(aq) >Cr₂O3(s) + 3H₂0(1) 12.00 moles of chromium (III) hydroxide is decomposed. Calculate moles of the water produced. O 18.00 moles O 1.500 moles O 4.00 moles O 12.00 molesarrow_forwardOxygen is a product of the decomposition of mercury(II) oxide as shown below. 2 HgO(s) → 2 Hg(ℓ) + O₂(g) In the last question, you determined that you started with 0.0152 moles of HgO. How many moles of O₂ gas will be formed in this reaction?arrow_forward
- Consider the following balanced equation. 3 Ag(s) + 4 HNO3(aq) → 3 AgNO3(aq) + NO(g) + 2 H2O(l) Calculate the number of grams of NO produced as a byproduct of the reaction of 118.17 grams of Ag with excess HNO3. Give your answer to the correct number of significant figures without unit. Molar mass of Ag: 107.87 g/mol Molar mass of HNO3: 63.01 g/mol Molar mass of AgNO3: 169.87 g/mol Molar mass of NO: 30.01 g/mol Molar mass of H2O: 18.02 g/molarrow_forwardBefore the use of CFCs, sulfur dioxide was used as a refrigerant. A sample of sulfur dioxide undergoes a simple decomposition reaction as follows: 8 SO2(g) --> S8(s) + 8 O2(g) Calculate the number of moles of oxygen gas produced in addition to the production of 1.18 mol of sulfur.arrow_forward
arrow_back_ios
arrow_forward_ios
Recommended textbooks for you
 ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning
ChemistryChemistryISBN:9781305957404Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCostePublisher:Cengage Learning ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education
ChemistryChemistryISBN:9781259911156Author:Raymond Chang Dr., Jason Overby ProfessorPublisher:McGraw-Hill Education Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning
Principles of Instrumental AnalysisChemistryISBN:9781305577213Author:Douglas A. Skoog, F. James Holler, Stanley R. CrouchPublisher:Cengage Learning Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education
Organic ChemistryChemistryISBN:9780078021558Author:Janice Gorzynski Smith Dr.Publisher:McGraw-Hill Education Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning
Chemistry: Principles and ReactionsChemistryISBN:9781305079373Author:William L. Masterton, Cecile N. HurleyPublisher:Cengage Learning Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY
Elementary Principles of Chemical Processes, Bind...ChemistryISBN:9781118431221Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. BullardPublisher:WILEY

Chemistry
Chemistry
ISBN:9781305957404
Author:Steven S. Zumdahl, Susan A. Zumdahl, Donald J. DeCoste
Publisher:Cengage Learning

Chemistry
Chemistry
ISBN:9781259911156
Author:Raymond Chang Dr., Jason Overby Professor
Publisher:McGraw-Hill Education

Principles of Instrumental Analysis
Chemistry
ISBN:9781305577213
Author:Douglas A. Skoog, F. James Holler, Stanley R. Crouch
Publisher:Cengage Learning

Organic Chemistry
Chemistry
ISBN:9780078021558
Author:Janice Gorzynski Smith Dr.
Publisher:McGraw-Hill Education

Chemistry: Principles and Reactions
Chemistry
ISBN:9781305079373
Author:William L. Masterton, Cecile N. Hurley
Publisher:Cengage Learning

Elementary Principles of Chemical Processes, Bind...
Chemistry
ISBN:9781118431221
Author:Richard M. Felder, Ronald W. Rousseau, Lisa G. Bullard
Publisher:WILEY