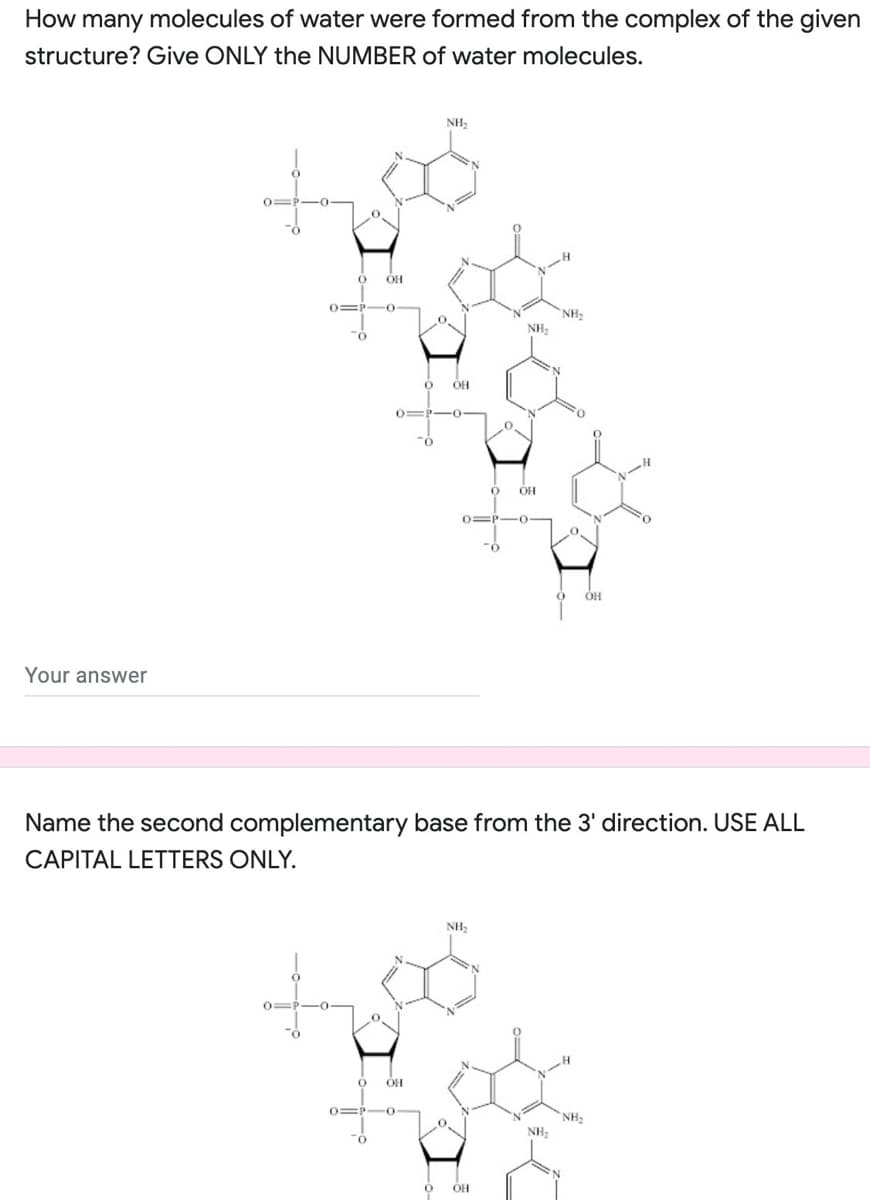
How many molecules of water were formed from the complex of the given structure? Give ONLY the NUMBER of water molecules.
Q: Molecules that have the same formula but a different structure are called what? Why does a different…
A: Elements are chemical compounds that have the same number of atoms. Chemical compounds are made up…
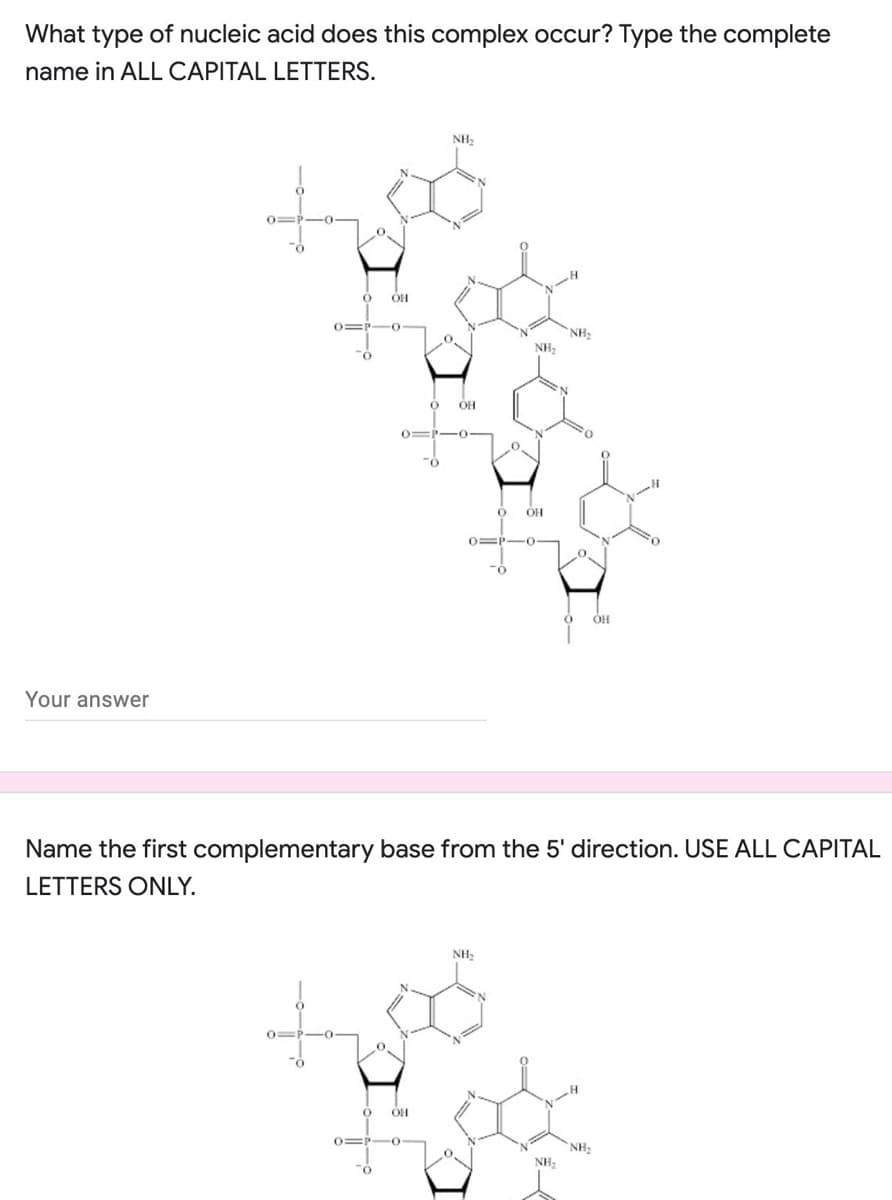
Q: What type of bonds are formed between complementary bases? a triple carbon to carbon bonds b…
A: Two lengthy molecules twist around each other in a double helix in double-stranded DNA. Deoxynucleic…
Q: Н—С—ӧ—н 5-H I-
A: Bond angle : The angle between bonding orbitals formed around central atom . Bond angle can be…
Q: Explain why two identical atoms joined by a covalent bond cannot form a hydrogen bond.
A: Two or more atoms can associate with each other by chemical bonds.
Q: Which of the following associations between R groups are the strongest? (a) hydrophobic interactions…
A: An amino acid consists of an R group, which is attached to the αC atom. The Cα amino group joined to…
Q: Which of the following bonds are high energy? (select all that apply)
A: Bond energy - it is the requirement of the energy to break the a covalent bond formed between two…
Q: Which of the following is responsible for the cohesive property of water? A Hydrogen bonds between…
A: Water is directly and intimately involved in all detail of animal physiology, but because it is so…
Q: Which of the following is a representation of a covalently bonded molecule that shows the relative…
A: Structural formula is generally used to describe the molecular structure of a chemical compound…
Q: What is the basic units of matter and the defining structure of elements? a. Cell b. Atom c.…
A: Matter- It is any substance that takes up space, has volume and has mass. It exists in various…
Q: For a covalent bond to be polar, the two atoms that form the bond must have differing…
A: Polar bond A polar bond is a covalent bond which have unequal distribution of electrons in the bond.…
Q: NH3 OH NH: NH: OH
A: The given structure is a chain of nucleotides joined by phospshodiester linkage. A nucleotide is…
Q: When two atoms share a pair of valence electrons with different levels of electronegativity, what…
A: Covalent Bond: A chemical link involving the sharing of electron pairs between atoms is known as a…
Q: What does the atomic number tell us about an atom
A: The atomic number which is denoted by Z of a chemical element uniquely identifies the chemical…
Q: Complete the following diagram illustrating ionic bonds. Atom X gives one electron to each atom Y.…
A: The atom is made up of three parts neutron, electrons, and proton. The neutrons are neutral and the…
Q: Based on their electronegativity values, which of the following atom pairs will form an ionic bond?
A: Electronegativity refers to the ability of an atom to pull or attract electrons toward it.…
Q: 12
A: Most of the Atoms don't exist in atomic form but they exist in the molecular form. So to understand…
Q: Given below is a macromolecule HO Near 1. Based on the functional groups shown what type of molecule…
A: Note: As Per Guidelines, We Can Answer One Question At A Time. Ask Again To get rest answers.…
Q: A carbon atom contains six protons and six neutrons.A. What are its atomic number and atomic…
A: CARBON - Carbon is a chemical element with the symbol C . It is nonmetallic and tetravalent—making…
Q: Nucleus acids are organic compounds. In addition to hydrogen, what element is an essential component…
A: Nucleic acids are organic compounds. They are of two types in our cells. DNA- Deoxyribonucleic acid.…
Q: What is the difference between a molecule and a compound? Give an example of each
A: The branch of science which is concerned about the chemical, physicochemical processes, and…
Q: Monomer 1 Monomer 2 ОН НО- Monomers linked by covalent bond H,0 Monomer 1 Monomer 2 Monomers linked…
A: Biological macromolecules are crucial cellular components that execute a variety of functions that…
Q: of the bonds in the molecule below are polar. explain why.
A: The chemical bond is an attraction between atoms, ions or molecules that enables the formation of…
Q: At which pH would the structure shown be predominate? H₂N-CH- - H OpH 1 pH 3 pH 8 pH 11 0 0
A: Glycine has the simplest structure among all the amino acids. This side chain of amino acid has most…
Q: what is a weak bond between hydrogen atom with a partial positive charge(usually O or N) and…
A: Chemical bonds. In biology, there so many types of bond which help to bind atoms, molecules,…
Q: Which of the following statements correctly explains why a water molecule is polar? A. Water is…
A: A water molecule is made up of two hydrogen atoms and one oxygen atom. Water is considered as a…
Q: Compute the formal charge (FC) on each atom in the following structures [H2CNH2]+
A: The formal charges are determined by counting the electrons that take part in each atom and compare…
Q: If you are given the structure of a molecule, explain how you would determine if it’s chiral or…
A: The property of a stereoisomer of a molecule by which it is non-superimposable on its mirror image,…
Q: ________ atoms give organic molecules their overall shape; ________ atoms determine the overall…
A: Compounds can be classified as an organic and inorganic compound on the basis of the material from…
Q: What is the maximum number of hydrogen bonds a water molecule can form with neighboring water…
A: Cohesiveness means the strong attraction of molecules for other molecules of the same kind. Water…
Q: A covalent bond consists of a pair of shared electrons between two atoms. How does this shared pair…
A: Covalent bonds are formed by the mutual sharing of the electrons between two atoms having…
Q: Why is the 3-dimensional structure of naturally occurring molecules important
A: The 3-D structure of naturally occuring molecules is important because :- The three-dimensional…
Q: Water molecules have one oxygen atom that is more electronegative than the two hydrogen atoms bound…
A: In biological molecules, two main types of bonds are found that is covalent and non-covalent bonds.…
Q: Match the structure formula to the chemical formula for this substance
A: According to the question, a molecular structure is given. Now, we have to match the structural…
Q: Atomic Structure W Label the parts of an atom on the diagram below.
A: An atom is the basic unit of matter.
Q: Which kind of bond holds atoms in a water molecule together? What attracts water molecules to each…
A: A water molecule has two hydrogen atoms and one oxygen atom. The water molecules present in a liquid…
Q: What would be the simplest way to tell the difference in non polar covalent bonds, polar covalent…
A: The complete transfer of electrons leads to ionic bonds formation, whereas, sharing of electrons…
Q: What type of chemical bond is present in a single water molecule?
A: An attraction between two atoms, molecules or ions is termed as a chemical bond. Due to the presence…
Q: a. Complete the bond table below. Bond Type Short Description Types of Atoms involved Example POLAR…
A: Chemical bonds are established between atoms, molecules, and ions, which results in the formation of…
Q: The triple point of an element is at 75 torr and 125°C. This means that liquid is more dense than…
A: Various biochemical and physiological processes occurs in the cells resulting in various activities…
Q: fill the following table
A: Please note, keeping with the site regulations and in order to provide a good quality answer, the…
Q: Which of the four emergent properties of water is the most important one for supporting life on…
A: Water has 4 essential emergent properties that can support life on Earth. The 4 major emergent…
Q: Use the following diagram to answer the question: Which of the molecules have hydrogen bonding? (B…
A: In the given images, we are shown different biomolecules. There are many biomolecules present in the…
Q: A polymer made of 24 monomers would have lost ______________ molecules of ________.
A: The examples of polymers in biological system are the proteins, polysaccharides etc. They are made…
Q: If the pH is 2, what is the %HA at of a molecule has a pKa = 6? Report the answer to one decimal…
A:
Q: Considering only the side chains of the amino acids in the molecule shown above, how many can…
A: Hydrogen bonding is the bonding between the Hydrogen in a molecular dipole and an electronegative…
Q: Draw and label a simplified model of an atom. Explain how this model misrepresents our understanding…
A: An atom is the smallest component of an element present everywhere in the universe. It is made up of…
Q: Which element is a metal? A. C B. Rn C. CI D. Cr
A: The classification of chemicals into different groups and understanding their properties individual…
Q: What are differences between covalent bonds (both polar and nonpolar) and ionic bonds. Recognize…
A: In a molecule or compound, two or more atoms combined together by bonds called chemical bonds or…
Q: 4
A: Ice as well as hot water are less denser than cool water that leads the ice to float on top of…
Step by step
Solved in 3 steps with 3 images

- What kind of hydroxyl groups do carbohydrates have? (More than one answer is possible.) A. secondary B. tertiary C. primary D. quaternaryWhat is the isoelectric point of casein? Please choose one correct answer only. A. pH 6.6 B. pH 5.6 C. pH 4.6 D. pH 3.6 E. None of the given optionsWhy is it unlikely that two neighboring water moleculeswould be arranged like this?H HH HO O
- ______________ are nitrogen bases with two rings.Which bond should be the strongest? Group of answer choices Sodium (Na) - Fluorine (F) Lithium (Li) - Chlorine (Cl) Carbon (C) - Oxygen (O) Ammonia (NH3) - Ammonia (NH3) Water (H2O) - Ammonia (NH3)Breaking the hydrogen bonds that hold the bases together requires energy. Which of these two will be the most difficult to separate?a. C – Tb. T – Gc. G – Td. A - T
- ___________ are nitrogen bases with one. They include __________ and ___________.Can you explain the various options and how did you come to the answer? How to determine when something is a weak acid or base or strong acid or base? Is there are any trick?What type of chemical bond forms between hydrogen and oxygen atoms within a water molecule? Which type of chemical bond forms between water molecules, binding them to one another?
- what is the parent shape of the molecule calledDefine the following terms:a. acidb. basea. weak acidd. weak basee. conjugate base.Which of the following substances would be considered basic? A. hydrochloric acid (pH 1.0) B. milk (pH 6.6) C. baking soda (pH 8.1) D. water (pH 7.0) E. Dr. Pepper (pH 3.0)

