Q: Explain the structure of benzene (C6H6) in detail using a ubiquitous electron model (the theory of…
A: The most commonly encountered aromatic compound is benzene. The usual structural representation for…
Q: 18 Arrange the hybridizations of carbon based on the highly electronegative * sp > sp3 > sp2 sp >…
A: Hybridization is the concept of mixing atomic orbitals into new hybrid orbitals ( with different…
Q: Match the Lewis dot structures with the bond angles of the central atom. 1. 90⁰, 180° 2. 90⁰, 120⁰,…
A:
Q: с -ОСНЗ || НN- CH —С -NH—СHCН CH, — С— ОН
A:
Q: Use molecular orbital theory to explain the following data (everything must be explicit): Bond…
A: No. of electrons in O2 = 16 electrons No. of electrons in O2+ = 15 electrons No. of electrons in N2…
Q: Which one of the following diatomic species has a bond order of 3/2 (one and a half bond)? hydrogen…
A: Bond order can be calculated as :-
Q: Which species contains three sigma bonds and one pi bond? A. PF3 B. NH4+ C. C2H2 D. CO32-
A: Sigma (σ) bonds are the bonds in which shared hybrid orbital’s electron density are concentrated…
Q: Sketch the bonding molecular orbital that results from the linear combination of two 1s orbitals.…
A:
Q: Describe the bonding in the molecule C15H22N2O3 in terms of sigma (σ) and/or pi (π) bonds and the…
A:
Q: esc 2. Use the molecular orbital diagram shown to determine which of the following is most stable.…
A: Bond Order of a species is determined by the following formula : BO = (No. of bonding electrons -…
Q: 2. Use partial orbital diagrams to show how the atomic orbitals of the central atom lead to hybrid…
A: BF4-undergoes sp3 hybridization.
Q: sund 1: Η Η Η H—C-C-C-0-H Η Η Η lar orbital type: orbitals in the sigma bond: orbitals in the pi…
A: According to the question,we need to determine each of the highlighted bond for the compounds.…
Q: using LCAO-MO theory what is bond order of C2 3 0.5 0 -0.5 1 or 2
A:
Q: Identify the type of bond or the type of orbital the indicated electrons occupy in the diatomic…
A:
Q: Each of the non-hydrogen atoms of this skeletal structure has been randomly numbered. Match the…
A: Hybridization is defined as the phenomenon of intermixing the orbitals of slightly different…
Q: Identify the hybridization states of the indicated atoms. II III O1) sp3; (1I) sp²2; (1II) sp O1)…
A:
Q: Identify all the central atoms that are sp°d2-hybridized in the following molecule: 3- :ö:" XeF2…
A:
Q: Write a hybridization and bonding scheme for each molecule. Sketch the molecule, including…
A:
Q: 27 - Ethylene molecule C2H4 the hybridization is. A-Sp¹ B - Sp² C-Sp³ O A 28 - Acetylene molecule…
A:
Q: Use molecular orbital theory to predict if each molecule or ion exists in a relatively stable…
A: The stability of any molecule is determined by the bond order because it indicates the stability of…
Q: a. Explain Von't Hoff tetrahedral carbon atom theory and asymmetry with examples.
A: van't Hoff suggested two ways of using tetrahedra as models for the carbon atom For drawing one…
Q: 28-Acetylene molecule C2H2 the hybridization is. A - Sp¹ B - Sp² C - Sp³ O A B O C
A:
Q: For each of the following molecules, give the bond order, and describe them as paramagnetic or…
A: “Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Rank the diatomic molecules C2. N2". and 02 in terms of increasing bond orders? Justify your answer…
A:
Q: Fill in the molecular orbital energy diagram for the diatomic molecule C2. carbong MO's carbona 2p…
A: A molecular orbital diagram, or MO diagram, is a qualitative descriptive tool explaining chemical…
Q: Which of the following ions is linear? O NO₂ OSCN O NH₂* O SO₂²- O SO3²-
A: Generally trimolecular compound without lone pair are linear
Q: If a molecule demonstrates paramagnetism, then: The substance can have both paired and unpaired…
A: The given statement has to be matched with correct options.
Q: Q2) A- What is the bond order and the magnetic properties of 02 molecule.
A: Determine bond order and magnetic property of O2 molecule ?
Q: 65. This is an example of, hybridization." 109.5° A sp sp2 O sp3
A:
Q: :The hybridization of the central atom in XeF 5 * is sp2.a O sp3.b P sp3.c dsp3.d sp.e
A: The ion is XeF5+, in this ion, a xenon atom is attached with five fluoride atoms. Hybridization in…
Q: Draw the Lewis structures and name the geometry shapes of the following molecules: Do not forget to…
A: Given: To draw Lewis structure of following species:
Q: Determine the molecular structure abs hybridization of the central atoms X in the poly atomic ion…
A: The ion given is XY3+ where X is the element with number of electrons 36 in neutral atom and Y forms…
Q: Give the electronic configurations and bond orders for N2, N2+, N22+, N2-, N22-, 02,
A: Bond order = 12Nb-Na, where, Nb=no. of electrons in bonding molecular orbital and Na= no. of…
Q: Predict the hybridization of the central atom in a molecule with ideal bond angels of 120° and 90°
A:
Q: 07. Present the linear structure formula of the following compounds, including non- genetic pairs,…
A: The hybridisation(mixed orbital function) on the central atom can be identified by using the…
Q: 20) The hybrid orbitals used for bonding by Xe in the unstable XeF2 molecule are, orbitals. A) sp3d2…
A: Given that : We have to identify the hybrid orbitals used for bonding by Xe in the unstable XeF2…
Q: Figure out the valence electron configurations & bond orders in the these molecules: : C+ 2, C2, C-…
A: C+2 Total number of electrons = 4 Electronic configuration: 1s2 2s2 Diamagnetic C2 Total number…
Q: 13) Give the shape that describes the hybrid orbital set sp³d² : A) trigonal planar B) octahedral C)…
A: Hybridization : It is introduced by pauling to explain equivalent nature of covalent bonds in a…
Q: Write a hybridization and bonding scheme for each molecule. Sketch the molecule, including…
A:
Q: Which of the following are predicted by the molecular orbital model to be stable diatomic species?…
A: By looking at the bond order, we can configure out the stable diatomic species
Q: Fill in the Molecular Orbital Energy Diagram for the diatomic molecule 02. E 2p oxygenÅ 2s MO's 0*2p…
A: In MOT diagram we fill Electron according to their energy in increasing order.
Q: Describe the hybridization state of Se in SeF6.
A:
Q: 8.) What is the hybridication of the corbon atom below H-C :0-H bo) sp2 C6) sp3 6.1 spt
A: We are given a structure having various atoms, we have to predict the hybridisation of the carbon…
Q: The overlap of atomic orbitals shown here will give rise to a molecular orbital with what type of…
A: Concept : Atomic Structure 1) positive part of wave function + positive part of wave function leads…
Q: the second and third molecular orbitals in a triple bond are: a) orbital s b) orbital p c) orbital…
A: Orbital: The place where electrons can spin is recommended as orbital. It provide 3D graphical…
Q: Of the following, the central atom is sp 3d 2 hybridized only in A) XEF4 В BeF2 C Br3- PH3 E PCI5
A: A) XeF4 : This compound structure is planar because Four F lies in on square plane and two lone pair…
Q: How many valence-shell electrons are there in NO? ▲ Figure 9.46 The energy-level diagram for atomic…
A: N has 5 valence electrons, and O has 6 valence electrons.
Q: Classify these orbital descriptions by type. Atomic orbital Hybrid orbital Molecular…
A: An atomic orbital is defined as the region around the nucleus where the chances of finding an…
Q: Determine the molecular structure and hybridization of the central atom X in the polyatomic ion XY3+…
A: Determination of molecular structure. Molecular structure of the polyatomic ion is T- shaped with 2…
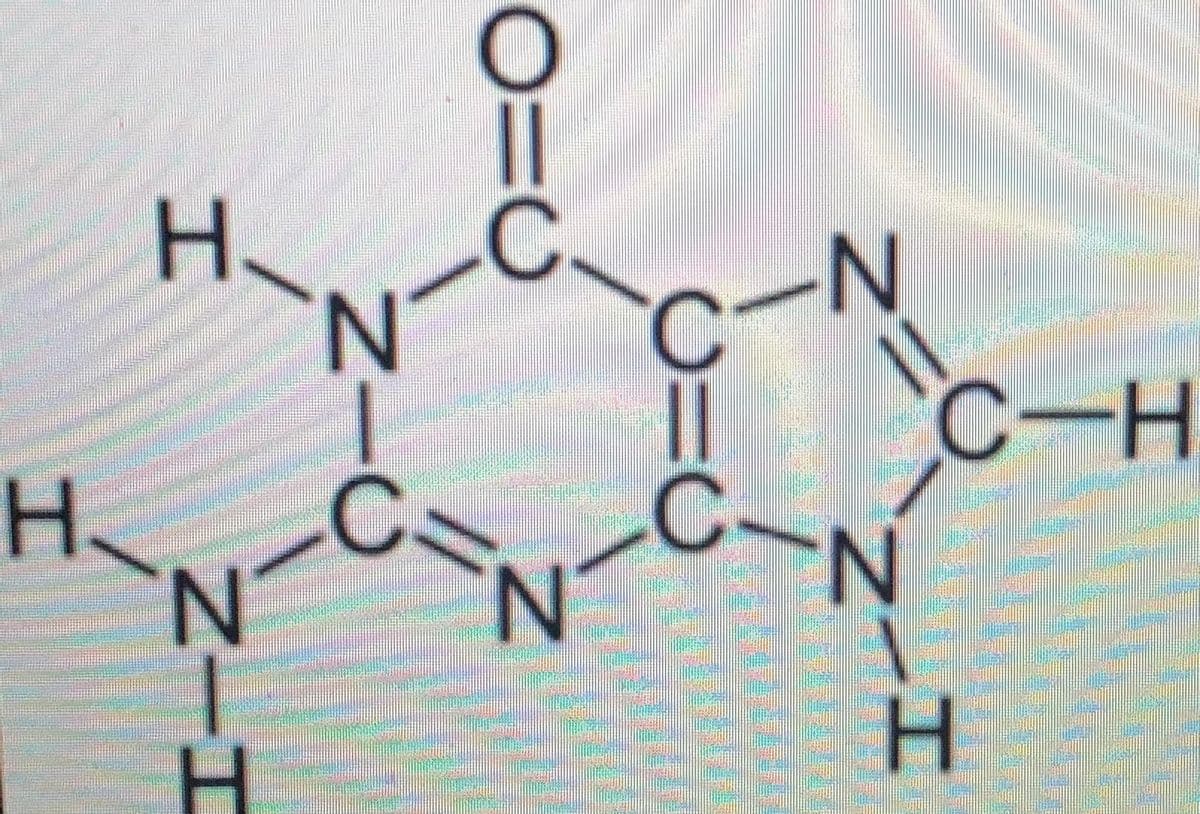
how many pi bonds are in the following molecule
7
4
5
11
16
17
21
3
20
or
14
Step by step
Solved in 2 steps with 1 images

- Can you help me to explain step by step with number 1 part A, number 1 part B, and number 1 part C? Can you explain it to me?Can you help me step by step with number 9 part A, part B, part C? Can you explain it to me?Rectified spirit is:(a) 50% ethanol(b) 80% ethanol(c) 95% ethanol(d) 40 to 50% ethanol

