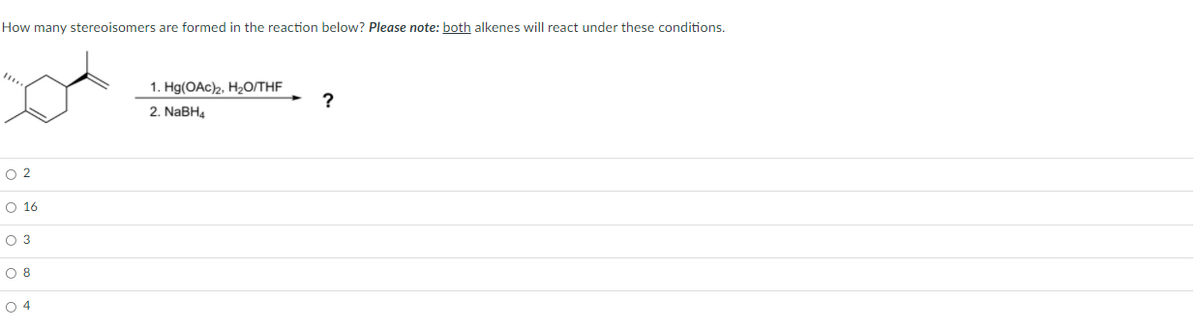
How many stereoisomers are formed in the reaction below? Please note: both alkenes will react under these conditions. 1. Hg(OAc)2, H2O/THE ? 2. NABH4 O 2 O 16 O 3 O 8 O 4
Q: The ammonium ion (pKa = 9.25) has a lower pKa than the methyl ammonium ion (pKa = 10.66). Explain w...
A: Given that pKa of ammonium ion(NH4+) = 9.25 pKa of methyl ammonium ion ( MeNH3+) = 10.66 Concept: A ...
Q: y = log 3.9201 (±0.0006)×10-3
A: Solution - According to the question - Given - Let us calculate the numerator terms: [ simplify] [1....
Q: Which statement is true about the isomerization reaction below? catalyst O The use of the catalyst m...
A: Isomerisation reaction is the conversion of one isomer into another isomer. Trans form of alkene is ...
Q: Dehydrating Cyclohexanol Pre lab Q's A. Which the following is true? I both constitutional isbiners ...
A: Dehydration means removal of water. In dehydration reaction of alcohol, the -OH group is first proto...
Q: Summarize what you have learned nodule separate sheet of paper. You can improve the concept map by a...
A: Atom is the basic building block of all matters whereas ions are charged atoms or molecules.
Q: For the reaction pictured, the experimental enthalpy is -41.3 ± 1.3 kJ mol-1. When using the Gaussia...
A: The correct answer is given below
Q: Complete the following reactions: || C-0-CH,CH, +NAOH- a. b. CH;(CH,),-C-0-CHCH,+H,O CH3
A: Answer of the question given below,
Q: Which of the following compounds can be converted to an alkyl lithium compound in one step by reacti...
A: Alkyl lithium compound formed in one step if there is no acidic hydrogen present .
Q: Which of the following statements is true for a buffer prepared from 2g dihydrogen phosphase and 1.3...
A:
Q: 2. are the percent compositions of the flue (dry basis) if the fuel is burned stoichiometrically? As...
A: We are required to calculate the percent compositions of the flue gas( dry basis) for the combustion...
Q: Describe the difference between solvation of ionic compounds and solvation of covalent compounds, Fo...
A: Ionic compound is made up of ions while covalent compound is made up of molecules. Compound which is...
Q: A 25.00-mL sample of propionic acid, HC3H5O2, of unknown concentration was titrated with 0.145 M KOH...
A: Given, The volume of unknown concentration of propionic acid = 25.00-mL The...
Q: Balance and complete: HNO3 (aq)+Ca(OH)2(aq) Please show your work!
A: Detail equation is given below
Q: For a diatomic vibrating rotor, in vibrational level v =3 and rotational level J, the sum of the rot...
A: Given that the sum of rotational and vibrational energies is 11493.6cm-1. Equlibrium frequency is 29...
Q: AICI3 CI
A: This is intramolecular F. C. Acylation reaction. They undergoes cyclisation to form cyclic ketone
Q: S° rxn for
A:
Q: In the synthesis of aspirin experiment, why do you think it is important to wait until the crystals ...
A: To take yield measurement , we usually weigh complete dry product.
Q: Balance
A:
Q: Why is it needed to cover the buffer solutions until their pH is determined? For a buffer prepared ...
A: Generally we cover a buffer solution untill the pH is determined because-
Q: write the line notation of the cell ii.) describe in detail how this galvanic cell is able to produc...
A: I) Line notation of galvanic cell is Oxidation half cell is written in the left side of salt bridge(...
Q: Consider the following redox reaction with nitrogen (N): NO3 + H* + 8e +→ _NH4* a. Balance this redo...
A:
Q: Which of the following have S configuration?
A:
Q: Consider the combustion of propane: C3 Hs (g) +502 (9) + 3CO2 (9) + 4H2O(1) AH = -2221 kJ A balloon ...
A:
Q: Use the phase diagram for water below to answer the following questions. 12. What is the state of wa...
A:
Q: For the successful synthesis of triphenylmethanol , 0.05 moles of methyl benzoate would require mole...
A:
Q: Draw the major product of this reaction. Include any relevant stereochemistry. Ignore inorganic bypr...
A: Phosphonium ylides are used in the Wittig reaction, a method used to convert ketones and especially ...
Q: The Kgp for Ag,CrO, is 1.2 x 10-12. a) Complete writing the balanced equilibrium equation for the di...
A:
Q: Explain how you would draw XeF4 and determine its shape, hybridization, polarity and intermolecular ...
A: The molecule given is Xenon tetrafluoride XeF4.
Q: Which one of the following compounds will be least reactive towards a Friedel-Crafts reaction? O phe...
A:
Q: Provide the correct systematic name for the compound shown here. N-eth yl-N-meth yl butan amine N- 2...
A: Systatic name for give compound is, N,N- dimethyl Butamide.
Q: cide if the equilibrium favours the starting materials or the products.
A:
Q: of K. for t
A:
Q: A total of 665.19 Joules of heat are observed as 49.79g of lead is heated from 12.0°C to 78.12°C. Fr...
A:
Q: Calculate AG° at 599 K for H20(g) + 1/2 02(g) = H202(g) using the following data: H2(g) +02(g) 5 H2O...
A: In the given question equilibrium constant of the reaction is determined with the help of equilibriu...
Q: Calculate the total energy in J of an electron in a hydrogen atom in the n=3 state
A:
Q: Calculate the pH of a 0.877 M solution of NaF. The K, for the weak acid HF is 6.8 x 10*. pH =
A:
Q: draw 5-(1-methylpent-2-ynyl)-1-(2-methylprop-2-enyl)cyclohex-1-ene
A: Given :- IUPAC name of compound = 5-(1-methylpent-2-ynyl)-1-(2-methylprop-2-enyl)cyclohex-1-ene To ...
Q: Draw the major product of this reaction. Ignore inorganic byproducts. TSOH, H2O Type here to search
A: p-Toluenesulfonic acid and H20 is used for deprotection of ketal derivative. And convert into ketone...
Q: The reaction 2H2 + O2 → 2H2O is studied and the concentration of O2 is measured over time. The data ...
A:
Q: 3. A solution that may contain any of the group III cations. Treatment of the solution with CIO in a...
A: Group 3 cation involve Cr3+ , Al3+ , Ni2+ and Fe3+.
Q: HBrO will act as a Bronsted-Lowry acid or base when reacting
A:
Q: A 1.46 L buffer solution consists of 0.171 M butanoic acid and 0.341 M sodium butanoate. Calculate t...
A: Given, Volume of the buffer = 1.46 L Molarity of butanoic acid = [Butanoic acid] = 0.171 M Molari...
Q: 1. Which statement about the following chemical reaction is not correct? 3H2 + N2 2NH3 For every thr...
A: 1. In a balanced chemical equation, the coefficients of chemical species represents the molecules/mo...
Q: buffer solution?
A: Buffer solution is the mixture of weak acid Or weak base and it's salt. Called buffer which resist t...
Q: Potassium has three stable isotopes, potassium-39, potassium-40, and potassium-41. calculate the ave...
A: Potassium exhibit three stable isotopes, Potassium- 39, potassium - 40 and potassium - 41. Here, we ...
Q: Shown below is a two ion pair cluster of 1-methyl-3-methyl-imidazolium tetrafluoroborate, including ...
A: The three possible interactions between a two ion pair cluster of 1-methyl-3-methyl-imadazoliumtetra...
Q: 261.28 mL of 4.0°C water is heated until its temperature is 48.24°C. If the specific heat of water i...
A:
Q: Consider the ionization of HBrO3 and identify each species listed as a Bronsted-Lowry acid, Bronsted...
A:
Q: What series of reagents are necessary to carry out the following conversion? CH3 `Br CH,ca / AICa Br...
A: Mechanism is given below in next step
Q: In your own words what is the main reason why lactose is harder to break than sucrose?
A: Lactose is composed of a molecule of galactose joined to a molecule of glucose by a β-1,4-glycosidic...
Step by step
Solved in 2 steps with 2 images

- Please identify priorities of functional groups and name following molecules based on thier stereochemistry as R or SIs the following alkene E,Z, or neither? a. E b. Z c. neither d. It is one or the other, but which is impossible to determine from the information givenConsider a reaction where cis-but-2-ene is treated with OsO4 followed by NaHSO3/H2O. Draw the structure of one product that is formed in the reaction, including correct stereochemistry.
- Which alkene from the choices can react with excess Hbr to yield the major product shown in the first photo?OChem HELP... Draw the conformational structures (chair or boat conformations) for the MAJOR product formed when 1-tert-butylcyclohexene reacts with each of the following reagents. Also, indicate if the product obtained is racemic form. a) Br2, CCl4 b) Br2, H2O c) OsO4, then aqueous NaHSO3 d) ICl e) mCPBA, then H3O+, H20 f) O3, then Me2S (conformational structure not required) g) BH3:THF, then H2O2, HO- h) D2, Pt i) Hg(OAc)2 in THF-H2O, then NaBH4, HO- j) BD3 :THF, then CH3CO2TAddition of HBr to allene (CH2=C=CH2) forms 2-bromoprop-1-ene ratherthan 3-bromoprop-1-ene, even though 3-bromoprop-1-ene is formed froman allylic carbocation. Considering the arrangement of orbitals in theallene reactant, explain this result.
- Stereochemistry: Assign each of the following alkenes as E or Z or unassignable. Also assign each of the following alkenes as cis or trans or not applicable.Which alkene would give the product alcohol as shown as themajor product? A) I B) II C) III D) Both I and IIAddition of HBr to allene (CH2=C=CH2) forms 2-bromoprop-1-ene rather than 3-bromoprop-1-ene, even though 3-bromoprop-1-ene is formed from an allylic carbocation. Considering the arrangement of orbitals in the allene reactant, explain this result.
- Addition of HBr to allene (CH2 = C = CH2) forms 2-bromoprop-1-ene rather than 3-bromoprop-1-ene, even though 3-bromoprop-1-ene is formed from an allylic carbocation. Considering the arrangement of orbitals in the allene reactant, explain this result.What is the product (A, B, C, or D) of the reaction shown in Image30? a. C b.D c. B d. A1. i.What are the various ways by which alkenes may be synthesized? ii. Give two examples each of Unsymmetrical alkenes and reagents. iii. Give two examples of reactions of alkenes that result in Anti-Markonikov’s addition products

