Q: Predict the approximate chemical shifts of the protons in the following compounds. (a) benzene…
A: In benzene, all of the protons are aromatic protons. They are present in identical environment. Thus…
Q: Determine the structure of the compound C10H14 whose 13C NMR spectrum is shown here. C10H14 200 150…
A: The double bond equivalent of the compound can be calculated as follows:
Q: Identify each compound below from its molecular formula and its 13C NMR spectrum.
A: The 13C NMR always helps in recognizing carbon atoms that exist in given organic molecule. Each…
Q: Draw the structure that corresponds to the following information: Molecular formula: C₂H₂O…
A:
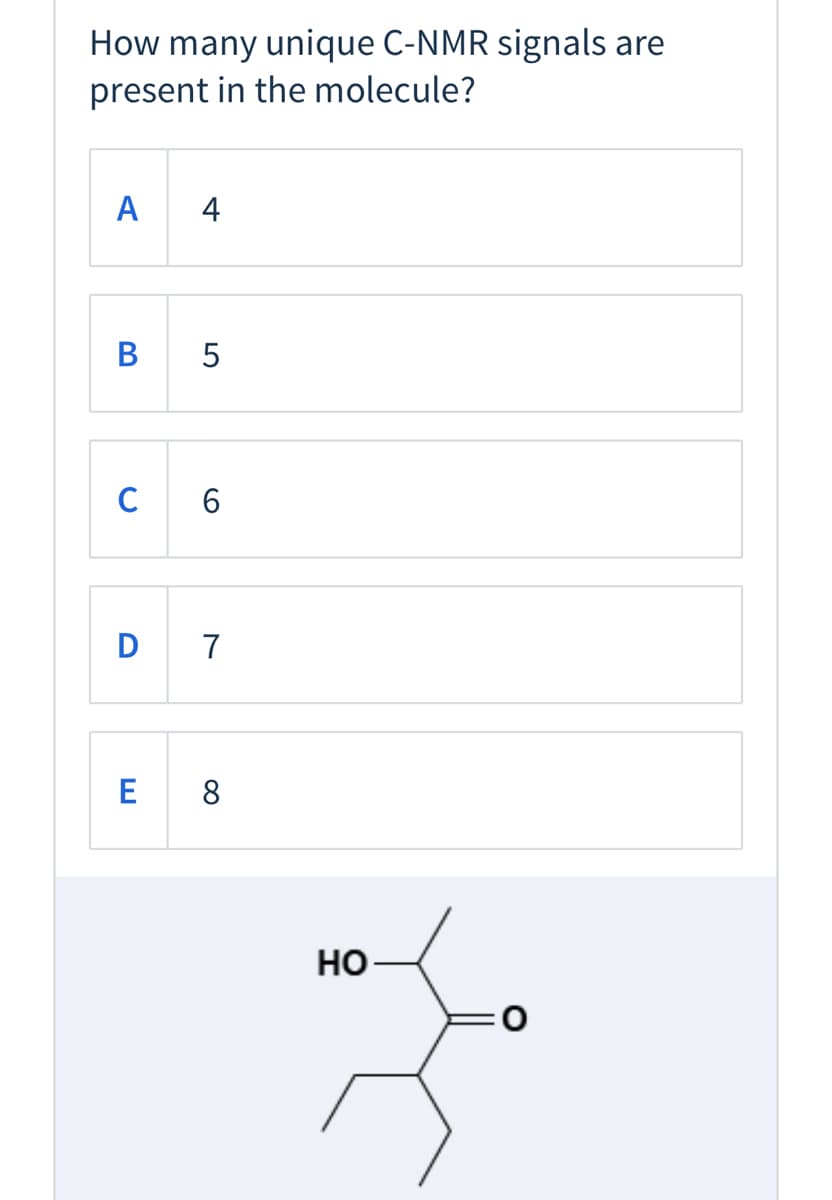
Q: How many 'H NMR signals do you expect to see for the following compound? Br B) 5 C) 6 D) 7 E) 8 4,
A: The number of NMR signal in a compound is equal to the number of chemically non-equivalent protons…
Q: Predict the number of signals and the splitting pattern of each signal in the 1H-NMR spectrum of…
A: It is a part of nuclear magnetic resonance spectroscopic technique (NMR) out of which we primarily…
Q: Draw the structure of C4H8O2. Indicate which carbon atoms correspond to which signal in the C NMR…
A: Note : Ethyl acetate is the compound. 4 signals observed in the 13C NMR of ethyl acetate.
Q: Considering the C-NMR below, choose the compound from the list provided that gave rise to the…
A: The carbons with different environment will give different signal in C-13 spectrum. This shows the…
Q: Identify the number of signals for the following compound in 'HNMR spectrum. OH H2 H2 H3C-C -CH3 (A)…
A: Proton nuclear magnetic resonance exploits the magnetic properties of the hydrogen nuclei to…
Q: Which of (a)-(d) indicates the correct order of chemical shifts of the four carbons in the 13C-NMR…
A:
Q: Choose the compound with the greatest number of 13C-NMR signals: A C D
A:
Q: How many signals will the following compound display in the carbon NMR spectrum? Compound:…
A:
Q: Predict the approximate chemical shifts that would exhibit the signals in the following compounds.…
A:
Q: Identify compound below from its molecular formula and its 13C NMR spectrum.
A: Calculation of double bond equivalent:
Q: i) ii) i) NH2 0 OH `NH2 NH
A: Note: According to our guidelines we are supposed to answer only first three subpart
Q: 6. Predict the numbers of signals, integration ratios, and splitting pattems of the following…
A: Answer is in second step
Q: Identify the carbon atoms that give rise to the signals in the 1°C NMR spectrum of each compound. a.…
A: a.
Q: Label each set of chemically equivalent protons, using a for the set that will be at the lowest…
A: Since you have asked multiple subparts, we will solve the first three subparts for you, for the rest…
Q: 14) The intensity of a signal in a ¹H NMR spectrum is determined by A. The number of neighboring C.…
A: In the given question we have to select the correct option from the following. the intensity of…
Q: How many 'H NMR signals would appear as singlets in the following molecule? Select one: O a. 4 O b.…
A: The splitting of an NMR signal is according to the N+1 rule, where N is the number of hydrogens on…
Q: Which compound is the correct structure for the given NMR? A 1. B 2. C (1) (3) (2) (2) (2) 11 10 9…
A: Proton NMR spectroscopy is a technique used for the identification of a compound.
Q: 1Compound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85…
A: Compound 1 molecular formula C7H16. 1H-NMR : one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm.…
Q: How many signals do you expect in the 1°C NMR spectrum of the following compound? O Seven O Three O…
A:
Q: How many signals will the following compound display in the carbon NMR spectrum? Compound:…
A: Number of signals in carbon NMR spectra is the total types of carbons present in the compound.
Q: Identify the carbon atoms that give rise to the signals in the 13C NMR spectrum of each compound.a.…
A: a)14ppm – sp3 carbon present next to H19ppm – sp3 carbon present next to H35ppm – sp3 carbon present…
Q: Predict the number of signals expected in the 'H-NMR spectrum. A 1 В 2 C 3 D 4
A: The number of signals in the 1H NMR spectrum is equal to the number of non-equivalent sets of…
Q: NO2 N. CH3 CH3 CH3 H3C
A: 13 C NMR spectroscopy is the application of NMR spectroscopy to carbon . 1H NMR spectroscopy is the…
Q: The following 1H NMR spectra are for four compounds, each with molecular formula of C6H12O2.…
A:
Q: 3. For each compound give the number of NMR signals and then determine how many peaks are present…
A: In 1HNMR , Number of signals are equal to number of chemically non-equivalent protons. And number of…
Q: How many signals are in its 13C NMR spectrum? H3C, CH3 CH3 H3C Express your answer as an integer.
A: signals in 13C NMR spectrum of given figure is :
Q: Predict the number of signals and the splitting pattern of each signal in the 1H-NMR spectrum of…
A: The number of signals and the splitting pattern of each signal in the 1H-NMR spectrum of a given…
Q: How many 1H signals would you expect to see in this compound's 1H NMR spectrum assuming all signals…
A:
Q: How many 13C-NMR signals does Toluene have? a) 2 b) 3 c) 4 d) 5
A: 13C-NMR (C-13 nuclear magnetic resonance) is a spectroscopic technique that is used to determine the…
Q: Consider the 13C NMR of benzamide. How many carbon signals would you expect to find for this…
A:
Q: (3) These natural compounds will have different numbers of 1H-NMR signals in different NMR solvents…
A: Deuterated chloroform (CDCl3) and methanol-d4 (CD3OD) both are widely used solvents in 1H-NMR…
Q: How many of the molecules below would have 3 signals in the ¹H NMR spectrum? Do not consider split…
A: In 1H NMR, each hydrogen atom present in a distinct chemical environment gives a peak in the…
Q: 3. How many signals would you expect in the 'H and 13C NMR spectrum of each molecule? H. ethyl…
A: The number of signals in 1H-NMR gives information about the number of different types of hydrogen…
Q: Draw the compound with molecular formula C9H100 that produces the following signals. Show your work…
A:
Q: 'H NMR 6 H 2H 2H 1H PPM "C NMR 20 PPM Each data set is specific to an individual compound. Use the…
A: A molecule can be identified on the basis of spectroscopy. It may be NMR, IR, 13C-NMR, or mass…
Q: Draw the structure of C3H80 and indicate which carbon atoms correspond to which signal in the C NMR…
A: The question is based on the concept of spectroscopy. we have to analyse the spectra provided and…
Q: 13. How many BC NMR signals are produced by each of the following compounds? (а) (b) CH3 CH3 O.…
A: We have to write 13C NMR signal which are produced by each of the following given compounds as…
Q: Identify each compound below from its molecular formula and its 13C NMR spectrum.
A: Calculation of double bond equivalent :
Q: Which compound is the correct structure for the given NMR? (3) (2) (2) 9 8 7 6. 3 1 PPM CN 2) 3)…
A:
Q: Chemical formula is C7H13Cl determine molecular structure using these 5 H-NMR characteristics.…
A: The double bond equivalence indicates the presence of one double bond in the compound.
Q: Predict the theoretical number of different NMR signals produced by each compound,and give…
A: The given compound is Ph-CHBr-CH2Br. From the above structure it is clear that the number of…
Q: The structure of citronellal is shown below, with the carbon atoms numbered. Also shown is a table…
A: Given: The given compound citronellal and its 13-C NMR peaks are,
Q: Identify the structures of each compound from the given data.
A:
Q: 5. Which of the following compounds displays besides other signals - a nonet (a signal that is split…
A:
Q: Cyclohexane-d1 (C6HD11) exhibits one signal in its "H NMR spectrum at room temperature. Two signals…
A: The structure of cyclohexane-d11 (C6HD11) is given as,
Q: The number of 13C NMR signals for this compound is 7. 8. 10 11 12 13 14 15 16 a. 1 17 18 19 b. 2 20…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- Identify the carbon atoms that give rise to the signals in the 13C NMR spectrum of each compound.a. CH3CH2CH2CH2OH; 13C NMR: 14, 19, 35, and 62 ppmb. (CH3)2CHCHO; 13C NMR: 16, 41, and 205 ppmc. CH2 = CHCH(OH)CH3; 13C NMR: 23, 69, 113, and 143 ppmIdentify the carbon atoms that give rise to the signals in the 13C NMR spectrum of each compound. a.CH3CH2CH2CH2OH; 13C NMR: 14, 19, 35, and 62 ppm b.(CH3)2CHCHO; 13C NMR: 16, 41, and 205 ppm c.CH2=CHCH(OH)CH3; 13C NMR: 23, 69, 113, and 143 ppm1H and 13C NMR spectrum Identify the multiplicity of each signal in the given compound.
- 1Compound 1 has molecular formula C7H16. It shows three signals in the 1H-NMR spectrum, one at 0.85 ppm, one at 1.02 ppm, and one at 1.62 ppm. The relative integrals of these three signals are 6, 1, and 1, respectively. Compound 2 has molecular formula C7H14. It shows three signals in the 1H-NMR spectrum, one at 0.98 ppm, one at 1.36 ppm, and one at 1.55 ppm. The relative integrals of these three signals are 3, 2, and 2, respectively. Propose structures for compounds 1 and 2, explaining how you reach your conclusion.How many 13C NMR signals does each compound exhibit ?How many 1H-NMR signals/peaks would you expect to see for the following molecule?
- Predict the number of signals and the splitting pattern of each signal in the 1H-NMR spectrum of each moleculeGiven the following NMR data, find the unknown compound. a. Identify the molecular formula; find IHDb. Identify the 1H and 13C NMR signalsDraw the structure of C4H8O2. Indicate which carbon atoms correspond to which signal in the C NMR spectrum/chart

