How much energy (in ) is required to evaporate 25% of 4g of water currently at 100 °C? The answer (in fundamental Sl unit) is (type the numeric value only, dont use scientific notation)
How much energy (in ) is required to evaporate 25% of 4g of water currently at 100 °C? The answer (in fundamental Sl unit) is (type the numeric value only, dont use scientific notation)
Chapter2: The Kinetic Theory Of Gases
Section: Chapter Questions
Problem 48P: Nuclear fusion, the energy solute of Sun, hydrogen bombs, and fusion reactors, occurs much more...
Related questions
Question
Kindly add labels for the all correct answers. Thank you. 6
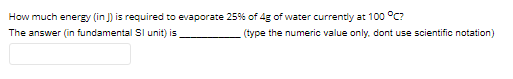
Transcribed Image Text:How much energy (in J) is required to evaporate 25% of 4g of water currently at 100 °C?
The answer (in fundamental SI unit) is
(type the numeric value only. dont use scientific notation)
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, physics and related others by exploring similar questions and additional content below.Recommended textbooks for you


Classical Dynamics of Particles and Systems
Physics
ISBN:
9780534408961
Author:
Stephen T. Thornton, Jerry B. Marion
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning


Classical Dynamics of Particles and Systems
Physics
ISBN:
9780534408961
Author:
Stephen T. Thornton, Jerry B. Marion
Publisher:
Cengage Learning

Principles of Physics: A Calculus-Based Text
Physics
ISBN:
9781133104261
Author:
Raymond A. Serway, John W. Jewett
Publisher:
Cengage Learning