Q: For most solid solutes, as temperature is increases, solubility
A: As the temperature of the solution increases, the average kinetic energy of solute molecules also…
Q: The mass of silver chromate that is dissolved in 175 mL of a saturated solution is
A: Consider the given information is as follows; Volume of solution = 175 mL = 0.175 L Mass of silver…
Q: ne molality of the
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: i) Define the term electrolyte. (ii) explain the difference between strong and weak electrolytes.…
A: Electrochemistry is the branch of chemistry in which we deal about electrical and chemical changes…
Q: A student has two different solutions of unknown organic compounds. obtain the following results.…
A: The presence of carbonyl compounds like methyl ketones can be identified by " idoform test " . In…
Q: So? (ag)→SO? (aq) (basic solution) 4 Express your answer as a chemical equation. Identify all of the…
A:
Q: Can you please answer sub question A and B and show all of the steps to the solution please and…
A: In the above problem the solutions are buffer solutions So we will use Henderson Hasselbalch…
Q: Compute for the molarity of a lution containing 100.35grams of dium hydroxide dissolved in water…
A: Molarity is defined as number of moles of solute divide by volume of solution
Q: Give the Van Slyke's Equation for: Weak electrolytes and their salt
A: Van slyke introduced the concept of buffer capacity according to him the buffer capacity equation…
Q: How many and what type of solutions do the following discriminants have? Positive Negative Zero…
A: If Discriminant is positive, There are two real and unequal solutions. If Discriminant is negative…
Q: Interconvert molar concentrations ofions and milliequivalents/liter.
A: Normality of a solution = Number of gram equivalent per liter = Number of milliequivalent per…
Q: The solubility of as temperature increases and the solubility of solids gases as temperature…
A: According to Henry law, Solubility if the gas is directly proportional to its partial pressure of…
Q: Let's start by finding the location on the periodic table for the atoms for substances that conduct…
A: In general ionic compounds formed between metal and non metal conducts electricity when dissolved in…
Q: How many milliliters of 0.250M HCl solution will exactly react with 10.5g of of solid NaHCO3?
A: Number of Mole- Ratio of given mass of substance to the molar mass of substance is called number of…
Q: If a solution can dissolve some more amount of solute if added to it, then the solution will be…
A: We have to identify the solution type
Q: Provide an example of a solution that contains a nonvolatile saw you dissolved in water. Statewood…
A: Most seen example for the solution that contains non volatile solute dissolved in water is Urea…
Q: Procedure Part A: Preparation of Solutions Sodium Bicarbonate Solution: Write procedures for…
A: Given : Concentration of sodium bicarbonate i.e NaHCO3 = 0.1 M And volume of solution = 50 mL =…
Q: e term "m" in the equation s of solute of solution
A: This calorimeter is used in to calculate the amount of heat released from the Solute when it's…
Q: Which one of the following can be classified as a weak electrolyte?
A: To identify which of the following given substance is a weak electrolyte. (i) HBr (ii) CaF2 (iii)…
Q: How do you solve for concentration of a solution?
A: The concentration of a solution is generally calculated as its molarity. The molarity of a solution…
Q: er:
A: The charge on the monoatomic ion is determined by its tendency to gain or lose electrons from its…
Q: molality of
A: Since you have asked multiple questions, we will solve the first question for you. If you want any…
Q: Example: What is the molali
A: Given, mass of NaOH = 10 g Mass of water = 500 g Molar mass of NaOH = 40 g/mol Molality =?
Q: Calculate the molar mass of NaF (with solution)
A: The molar mass of the compounds can be defined by the sum of the atomic masses of each atom making…
Q: The maximum concentration of solute that can be added is defined as Select one: a. degrees of…
A: Given:Maximum concentration of solute. To find : unknown term or undefined term. Solution : As we…
Q: Assuming the density of the phosphoric acid is 1.07 g/mL, what is the % of the solution?
A: Given: density of the phosphoric acid = 1.07 g/mL
Q: Calculate the volume percentage of solution F
A: Given :- volume of methyl alcohol = 50 ml Volume of water = 200 ml To calculate :- volume…
Q: beryllium oxide + strontium sulfate ---> Normal: Complete ionic: Net ionic:
A: A molecular equation represents molecules of reactants and products, a complete ionic equation…
Q: Match the following aqueous solutions with the appropriate letter from the column on the right.…
A:
Q: Standard solution is used to determine theconcentrations of ------ substances.( other, many, some)…
A: A Standard solution is a solution whose concentration is known accurately . It is prepared using a…
Q: Imagine that we have excess NaCl solid on the bottom of a saturated aqueous solution of NaCl. Please…
A: We have excess NaCl solid on the bottom of a saturated aqueous solution of NaCl.
Q: I need the answer of question attached. Please provide step by step solution.
A: Here is the answer
Q: What is the difference between strong and weak electrolytes? What is the difference between strong…
A: To find: The difference between strong and weak electrolytes and the difference between strong and…
Q: . According to Nernst', the molarity of the solution is the only factor that affect the value of…
A: NERNST EQUATION: In electrochemistry, he Nernst equation is used to find out the cell potential in…
Q: Which of the following would be classified as a strong electrolyte?
A: Covalent bonds are formed by sharing of electrons between two atoms. On the other hand, ionic bonds…
Q: Which of the following representations describes unsaturated, saturated, and supersaturated,…
A:
Q: Answer the following question and show your full solution.
A: Given: The concentration of sodium acetate, (CH3COONa) = 0.1 M. The concentration of acetic acid,…
Q: Calculate the concentration of the chemist's working solution.
A:
Q: How do you determine Moles, Molarity, and mass percent from titration results.
A: From titration results, we determine moles, molarity and mass percent. to find out the moles…
Q: Answer the following and show the complete solution and formula used:
A: The answer is 2400 litres
Q: Can you please answer question 17.43 and show all of the steps to the solution
A: In a galvanic cell, the conversion of chemical energy into electrical energy is observed. The cell…
Q: Q1: Gravimetric analysis refers ? 10 points Your answer Q2: Equivalence point calculate 10 points by…
A: Q. 1 Gravimetric analysis: It is a quantitative analytical technique. In this technique, we can…
Q: Please provide a step by step solution on how to compute for the volume of a 30% Brine Solution…
A: Given :- % w/v = 30% mass of solute = 10 kg To calculate :- volume of solution (in m3)
Q: Using Venn diagram compare and contrast the colligative properties of electrolyte and non…
A: A solution is formed by the combination of a solute and solvent. Even though the solution contains…
Q: Prepare 300ml of a 2M solution form 12M CuSO4 stock. Put into M1V1=M2V2 equation and solve.
A: Molarity is used to denote the concentration of the solution. The concentration of a particular type…
Q: An aqueous solution of silver rinate excess armmonia called A ammonical silver nitrate solution only…
A: An aqueous solution of silver nitrate in excess ammonia called
Q: Calculate the molarity of the KIO, solution.
A: Given :- mass of KIO3 = 549 g Volume of solution = 3.30 L Molar concentration is the ratio of…
Q: A TC glassware used to contain exact volume of prepared standard solution.
A: A standard solution is the solution of known concentration. 1) Erlenmeyer flask (or conical flask)…
Step by step
Solved in 2 steps

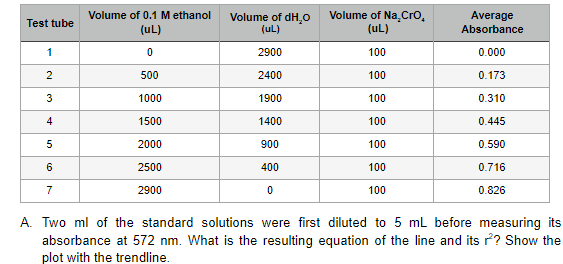
- Give the advantages and disadvantages of sequential injection analyzers compared to traditional flow Injection analyzers.analyte concentration(C)(mg/ml) injection volume (ul) elution time (time) peak DAD signal(mAU) caffeine 1 1 4.67 302.85 aspartame 5 1 7.53 15.83 benzoic acid 1 1 8.14 89.98 saccharin 1 1 1.91 84.86 mixture(add everything above with 1:1:1:1 ratio) 1 4.47 69.58 How to get the concentration of the mixture in this case?Compare and contrast the amount of caffeine you obtained from the single vs multipleextractions with Red Bull and Coca-Cola beverages. Explain why this has occured. Single Extractions: TEST SOLUTION NAME ABSORBANCE READING [CAFFEINE]mg/100ml 5ml Red Bull 2.412 57.9mg/100ml 5ml Coca-Cola 0.716 16.2mg/100ml Multiple Extractions: TEST SOLUTION NAME ABSORBANCE READING [CAFFEINE]mg/100ml 10ml Red Bull 3.253 78.453 10ml Coca-Cola 0.785 17.844
- A 0.0200 gram blood sample was decomposed by a microwave digestion technique followed by dilution to 100.0 mL in a volumetric flask. Aliquots of the sample solution were treated with a lead complexing reagent and water as follows: Solution 1: 10.0 ml blood sample + 20.0 mL complexing agent + 30.0 mL H20. Solution 2: 10.0 ml blood sample + 20.0 mL complexing agent + 26.0 mL H20 + 4.00 mL of 78 ppb Pb2+ standard. The resulting solutions were analyzed by UV/Vis at 375 nm. Absorbance for solution 1 = 0.155 and for solution 2 = 0.216. Calculate the concentration of lead (ppb) in the original sample.You have a concentrated sample solution of a new wine from a winery to analyze for malic acid content. once you have worked up the sample and done one 5x dilution you record an absorbance of .395 at the analytical wavelength of the malic acid. the molar absorptivity coefficient E of the malic acid for this method of measurement is 15.84 mM-1cm-1. What was the molarity of malic acid in original wine sample mM?A standard curve for glucose analysis was prepared.The slope (m) was found to be 1.341 while the y-intercept (b) was -0.333. Calculate the concentration (x) of glucose having an absorbance (y) of 0.151. 0.067 0.361 0.858 0.136
- A student weighed out 0.150 g of protein powder and dissolved it in 100 mL of water (Solution 1). The student then diluted this solution by transferring 1 mL into a 25 mL flask and diluting with water (Solution 2). Finally, 1 mL of that solution was transferred to a test tube and combined with 4 mL Bradford reagent. The absorbance of the solution in the test tube was 0.144. Assuming that the best fit linear line of the standard curve was y=0.04144x+0.01521 (μgmL), calculate the percent protein by mass in the original protein powder.The concentration of phenol in an aqueous solution is 10 ug/mL. The absorbance is found to be 0.209 when this solution is placed in a 1.00 cm cuvette and 258 nm radiation is passed through Calculate the specific absorptivity, including units, of phenol? What will be the absorbance if the solution is diluted to 5 ug/mL? What will be the absorbance if the path length of the original solution is increased to 5.00 cm?3 to the kit manual 200uL of plasma was mixed with 2.8 ml of the dilution buffer, mixed well then 200 uL of the dilution mixture was pipetted in 0.9 ml of solution A and 0.9 mL of solution B and measured using a spectrophotometer, the concentration was 30. What is the final concentration of the sample after adjusting the dilution factor
- Two components in an HPLC separation have retention times that differ by 15 s. The first peak elutes in 9.0 min, and the peak widths are approximately equal. The dead time to was 65 s. Use a spreadsheet to find the minimum number of theoretical plates needed to achieve the following resolution. R. values: 0.50 0.75, 0.90, 1.0, 1.10. 1.25, 1.50. L75. 2.0 and 2.5. How would the results change if peak 2 were twice as broad as peak 1?You obtained the following raw data when setting up a Bradford standard curve: BSA (mg/ml) Absorbancy 595nm 0 0.225 1 0.310 2 0.420 3 0.510 4 0.610 5 0.720 6 0.810 7 0.915 8 0.950 9 0.980 10 0.990 After blanking against a bradford-dH2O sample, the protein concentration of an unknown sample was determined using the same method and an absorbancy of 0.523 was obtained. Set up a standard curve, excluding outliers (experimental and statistical) and determine the protein concentration in the unknown sample in mg / ml (up to 3 significant figures).60 ml of standard hardness containing 1 mg of pure CaCO3 per ml consumed 22 ml of EDTA. 40 ml of water sample consumed 20 ml of EDTA solution using EBT indicator. 40 ml of water sample after boiling, filtering consumed 15 ml of EDTA. Calculate the temporary and permanent hardness of water sample.





