Q: Draw the product formed from the ring-closing metathesis of each compound. Then, devise a synthesis…
A: The given compound is diene. The ring closing metathesis (RCM) by Grubbs catalyst occurs, when the…
Q: Devise a synthesis of each compound from the indicated starting material.
A: a.
Q: Vitamin C is a stable enediol. Draw the structure of the two keto tautomers in equilibrium with the…
A: The two keto tautomers of vitamin C is shown below,
Q: Answer the following questions about benzphetamine, a habit-forming diet pill sold under the trade…
A: (a)
Q: What carbonyl compound and diol are needed to prepare each compound? b.
A: Given compounds, (a) (b)
Q: Devise a synthesis of each compound from the given starting materials. You may also use organic…
A: NOTE: Since, we only answer upto three sub-parts, we'll answer the first 3. Please resubmit the…
Q: What alkyl halides are needed to prepare each ketone using the acetoacetic ester synthesis?
A: Since you have posted a question with multiple questions, we will solve first question for you. To…
Q: What Grignard reagent and aldehyde (or ketone) are needed to prepare each alcohol? Show all possible…
A: Treatment of aldehydes with the Grignard reagent followed by its hydrolysis produces secondary…
Q: Safrole is a naturally occurring acetal isolated from sassafras plants. Once used as a common food…
A: To find: The hydrolysis product of safrole with aqueous acid?
Q: Devise a synthesis of each compound from the given starting material(s). Albuterol is a…
A: a. Synthesis of albuterol is given below, The first step involves the Friedel Crafts acylation…
Q: What product is formed when a solution of A and B is treated with mild base? This reaction is the…
A: Diseases, such as high-risk vascular disease and abnormal lipids are treated with the help of…
Q: Nabumetone is a pain reliever and anti-inammatory agent sold under the brand name of Relafen.a.…
A: a.
Q: Devise a synthesis of each compound from cyclohexene and organic alcohols. You may use any other…
A: (a) The synthesis of given target molecule involves three parts: First part: The treatment of…
Q: What products are formed when each acetal is hydrolyzed with aqueous acid?
A:
Q: Devise a synthesis of each compound from acetylene and any other required reagents.
A: NOTE: Since, we only answer upto three sub-parts, we'll answer the first 3. Please resubmit the…
Q: What product is formed when a solution of A to B is treated with mild base? This reaction is the…
A: The first step is the abstraction of an acidic proton by base from structure B as shown below : This…
Q: Devise a synthesis of each compound from benzene.
A: Since you have posted a question with multiple sub-parts, we will solve first three sub-parts for…
Q: Devise a synthesis of each compound using 1-bromobutane (CH3CH2CH2CH2Br) as the only organic…
A: Consider the given compound as a and b as shown below,
Q: What starting materials are needed to synthesize each compound by a Robinson annulation?
A: a). The starting materials that are needed to synthesize the given compound by using a Robinson…
Q: Synthesize each compound from ethyl acetoacetate. You may use any other organic compounds or…
A: (a)
Q: β-Vetivone is isolated from vetiver, a perennial grass that yields a variety ofcompounds used in…
A: Michael addition: The Michael addition begins with the removal of a proton from an ?,?-unsaturated…
Q: Devise a synthesis of each compound from cyclohex-2-enone and organic halides having one or two…
A: a) The name of the compound to be synthesized is shown below. The method of synthesis is shown…
Q: Draw a stepwise mechanism for the sulfonation of an alkyl benzene such as A to form a substituted…
A: Electrophilic substitution reactions are those reactions in which the electrophile usually displaces…
Q: Devise a synthesis of each compound from benzene.
A: Since we answer upto 3 sub-parts, we'll answer first three. Please resubmit the question specifying…
Q: Draw a stepwise mechanism for the conversion of lactone A to ester B using HCl in ethanol. B is…
A: In the first step of the mechanism the carbonyl group undergoes protonation followed by attack of…
Q: Devise a synthesis of each compound from the given starting material(s). Albuterol is a…
A: Given that,
Q: What carbonyl compound and diol are needed to prepare each compound?
A: When carbonyl compounds react with two equivalent alcohols in the presence of dry HCl gas, then…
Q: Identify A in the following reaction scheme, and draw a stepwise mechanism for the conversion of A…
A: Compound A can be drawn as
Q: Draw a stepwise mechanism for the intramolecular Friedel–Crafts acylation of compound A to form B. B…
A: Concept introduction: The Friedel-Craft acylation is a type of electrophilic substitution reaction.…
Q: (a) Give an acceptable name for each compound, (b) Draw the organic products formed when A or B is…
A:
Q: Devise a synthesis of each acetal from 1-bromo-2-methylhexane, alcohols (and diols) containing one…
A: The structure of 1-bromo-2-methylhexane is given as shown below. It consists of six carbon atoms in…
Q: Devise a synthesis of each alcohol from organic alcohols having one or two carbons and any required…
A:
Q: What alkyl halides are needed to prepare each ketone using the acetoacetic ester synthesis?
A:
Q: Devise a synthesis of attached compound from the indicated starting material. You may use any other…
A: There are many mechanisms in organic chemistry such as nucleophilic substitution, nucleophilic…
Q: Devise a synthesis of attached compound from ethanol (CH3CH2OH) as theonly source of carbon atoms.…
A: Ethanol gives ethene when it undergoes dehydration in the presence of strong acid . On reaction with…
Q: Draw a stepwise mechanism for the following intramolecular reaction.
A: Since O in the reactant is electronegative element. hence it will take proton from the HCOOH forming…
Q: Devise a synthesis of attached compound from benzene, any organic alcohols having four or fewer…
A: A synthesis of the attached compound from benzene, any organic alcohols having four or fewer…
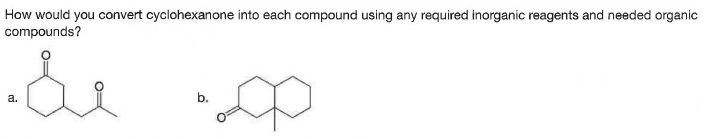
Q: Synthesize each compound from cyclohexanone and organic halides having ≤ 4 C's. You may use any…
A: (a) Treatment of cyclohexanone with a strong base, LDA, followed by the reaction with methyl bromide…
Q: Explain why acetanilide is less reactive toward electrophilic substitution than aniline.?
A:
Q: What Grignard reagent and carbonyl compound can be used to prepare the antidepressant venlafaxine…
A: The crabonyl compound and the grignard reagent that can be used to form venlafaxine is as follows:…
Q: Linalool and lavandulol are two of the major components of lavender oil. (a) What organolithium…
A: It is given that two major components of lavender oil are Linalool and Lavandulol and they need to…
Q: Devise a synthesis of each compound. You may use HC=CH, ethylene oxide, and alkyl halides as organic…
A: (a)
Q: Starting from benzene as the only organic reagent you have and use any other inorganic reagents to…
A: Synthesis of given compound from benzene: On following the below steps, 1,3-difluorobenzene can be…
Q: Devise a synthesis of attached compound from the given starting materials.You may also use organic…
A:
Q: Devise a synthesis of each compound from benzene and organic alcohols containing four or fewer…
A:
Trending now
This is a popular solution!
Step by step
Solved in 2 steps with 2 images

- What product is formed when a solution of A and B is treated with mild base? This reaction is the rst step in the synthesis of rosuvastatin (sold as a calcium salt under the trade name Crestor), a drug used to treat patients with high cholesterol.Draw a stepwise mechanism for the attached reaction, which involves two Friedel–Crafts reactions. B was an intermediate in the synthesis of the antidepressant sertralineDraw a stepwise mechanism for the sulfonation of an alkyl benzene such as A to form asubstituted benzenesulfonic acid B. Treatment of B with base forms a sodium salt C that canbe used as a synthetic detergent to clean away dirt.
- Draw a stepwise mechanism for the following reactions, two steps in R. B. Woodward’s classic synthesis of reserpine in 1958. Reserpine, which is isolated from the extracts of the Indian snakeroot Rauwolfia serpentina Benth, was used at one time to manage mild hypertension associated with anxiety.Draw a stepwise mechanism for the following reactions, two steps in R. B. Woodward’s classic synthesis of reserpine in 1958. Reserpine, which is isolated from the extracts of the Indian snakeroot Rauwola serpentina Benth, was used at one time to manage mild hypertension associated with anxiety.Devise a synthesis of each compound from benzene. You may also useany organic compounds having four or fewer carbons and any requiredinorganic reagents.
- Devise a synthesis of each compound from aniline (C6H5NH2) as starting material.Devise a synthesis of each compound from the given starting material(s). Albuterol is a bronchodilator and proparacaine is a local anesthetic.Show how you might use the Williamson ether synthesis to prepare each ether. (a) (b)
- Draw a stepwise mechanism for the sulfonation of an alkyl benzene such as A to form a substituted benzenesulfonic acid B. Treatment of B with base forms a sodium salt C that can be used as a synthetic detergent to clean away dirtDraw a stepwise mechanism for the following reaction, a key step in the synthesis of the anti-inflammatory drug celecoxib (trade name Celebrex).Draw a stepwise mechanism for the following reaction, a key step in the synthesis of ticlopidine, a drug that inhibits platelet aggregation. Ticlopidine has been used to reduce the risk of stroke in patients who cannot tolerate aspirin.

