Hydrophoblc Interactions between molecules In the cytoplasm are cruclal because O They provide the necessary atoms in reactions They facilitate the proper conformation of molecules O They allow covalent bond formation among reactants O They direct the rate of chemical reactions in cells
Hydrophoblc Interactions between molecules In the cytoplasm are cruclal because O They provide the necessary atoms in reactions They facilitate the proper conformation of molecules O They allow covalent bond formation among reactants O They direct the rate of chemical reactions in cells
Chapter5: Ground Rules Of Metabolism
Section: Chapter Questions
Problem 8SA
Related questions
Topic Video
Question
Answer the two questions! Will dislike if incomplete
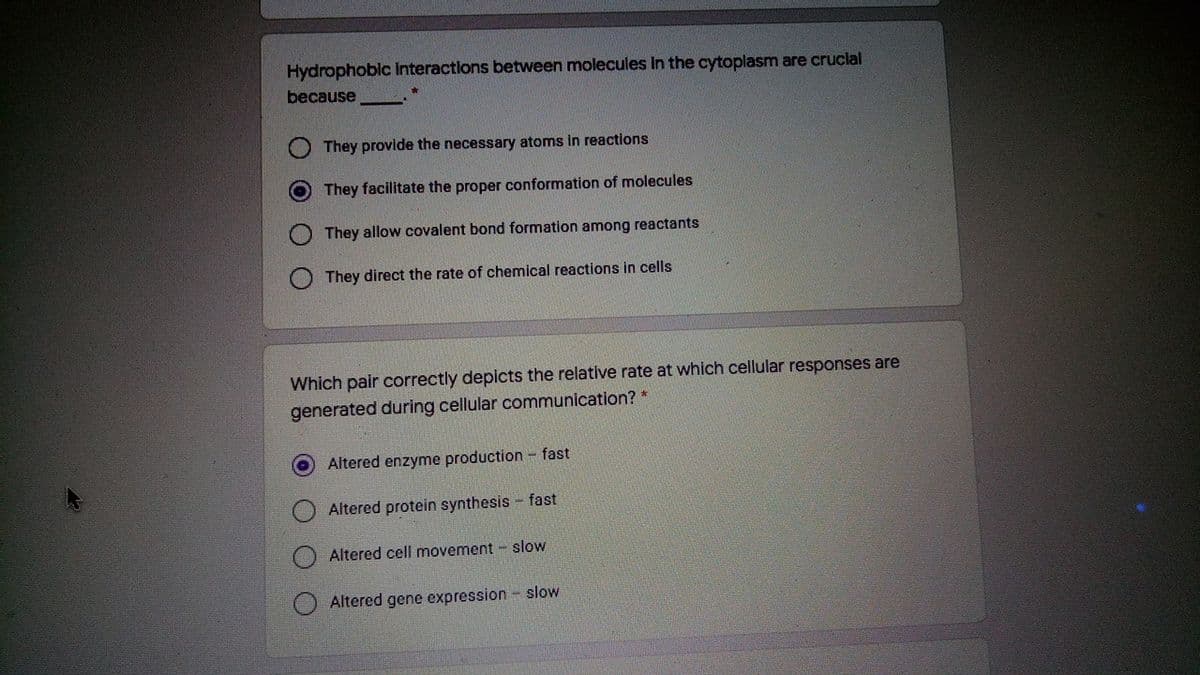
Transcribed Image Text:Hydrophoblc Interactions between molecules In the cytoplasm are cruclal
because
They provide the necessary atoms in reactions
O They facilitate the proper conformation of molecules
They allow covalent bond formation among reactants
They direct the rate of chemical reactions in cells
Which pair correctly depicts the relative rate at which cellular responses are
generated during cellular communication? *
Altered enzyme production- fast
Altered protein synthesis - fast
Altered cell movement - slow
Altered gene expression slow
Expert Solution
This question has been solved!
Explore an expertly crafted, step-by-step solution for a thorough understanding of key concepts.
This is a popular solution!
Trending now
This is a popular solution!
Step by step
Solved in 3 steps

Knowledge Booster
Learn more about
Need a deep-dive on the concept behind this application? Look no further. Learn more about this topic, biology and related others by exploring similar questions and additional content below.Recommended textbooks for you


Human Heredity: Principles and Issues (MindTap Co…
Biology
ISBN:
9781305251052
Author:
Michael Cummings
Publisher:
Cengage Learning


Human Heredity: Principles and Issues (MindTap Co…
Biology
ISBN:
9781305251052
Author:
Michael Cummings
Publisher:
Cengage Learning